・Basic Research・・Current Issue・ ・Achieve・ ・Search Articles・ ・Online
Submission・
・About IJO・
Acellular ostrich corneal stroma used as scaffold for
construction of tissue-engineered cornea
Xian-Ning
Liu1,2,3, Xiu-Ping Zhu1,2,3, Jie Wu3,
Zheng-Jie Wu4, Yong Yin1,2,3, Xiang-Hua Xiao1,2,3,
Xin Su4, Bin Kong4,Shi-Yin Pan1,2,3, Hua Yang1,2,3,
Yan Cheng3, Na An1,2,3, Sheng-Li Mi4
1Shaanxi Institute of
Ophthalmology, Xi’an 710002, Shaanxi Province, China
2Shaanxi Key Laboratory of Eye,
Xi’an 710002, Shaanxi Province, China
3Xi’an First Hospital, Xi’an
710002, Shaanxi Province, China
4Biomanufacturing Engineering Laboratory,
Graduate School at Shenzhen, Tsinghua University, Shenzhen 518055, Guangdong
Province, China
Correspondence to: Sheng-Li
Mi. Biomanufacturing engineering laboratory, Graduate School at Shenzhen,
Tsinghua University, Shenzhen 518055,
Guangdong Province, China.
mi.shengli@sz.tsinghua.edu.cn
Received: 2015-06-19 Accepted:2016-02-15
Abstract
AIM: To assess acellular ostrich corneal matrix used as a scaffold to reconstruct a
damaged cornea.
METHODS: A hypertonic
saline solution combined with a digestion method was used to decellularize the
ostrich cornea. The microstructure of the acellular corneal matrix was observed
by transmission electron microscopy (TEM) and hematoxylin and eosin (H&E)
staining. The mechanical properties were detected by a rheometer and a tension
machine. The acellular corneal matrix was also transplanted into a rabbit
cornea and cytokeratin 3 was used to check the immune phenotype.
RESULTS: The
microstructure and mechanical properties of the ostrich cornea were well
preserved after the decellularization process. In vitro, the methyl
thiazolyl tetrazolium results revealed that extracts of the acellular ostrich corneas (AOCs) had no inhibitory effects on the
proliferation of the corneal epithelial or endothelial cells or on the
keratocytes. The rabbit lamellar keratoplasty showed that the
transplanted AOCs were transparent and completely incorporated into the host
cornea while corneal turbidity and graft dissolution occurred in the acellular
porcine cornea (APC) transplantation. The phenotype of the reconstructed cornea
was similar to a normal rabbit cornea with a high expression of cytokeratin 3
in the superficial epithelial cell layer.
CONCLUSION: We first used AOCs as scaffolds to
reconstruct damaged corneas. Compared with porcine corneas, the anatomical
structures of ostrich corneas are closer to those of human corneas. In
accordance with the principle that structure determines function, a xenograft
lamellar keratoplasty also confirmed that the AOC transplantation generated a
superior outcome compared to that of the APC graft.
KEYWORDS: ostrich; acellular corneal stroma; tissue
engineering; cornea
DOI:10.18240/ijo.2016.03.01
Citation: Liu XN, Zhu XP, Wu J, Wu ZJ, Yin Y, Xiao XH, Su X,
Kong B, Pan SY, Yang H, Cheng Y, An N, Mi SL. Acellular ostrich corneal stroma
used as scaffold for construction of tissue-engineered cornea. Int J
Ophthalmol 2016;9(3):325-331
INTRODUCTION
Corneal
transplantation is presently the only effective method for the visual
rehabilitation of patients with corneal blindness. However, there is an
increasing need for human donor corneal tissue and a
shortage of suitable cornea donors. Therefore, many researchers have
attempted to fabricate alternatives to donor corneas for the treatment of corneal
blindness[1-4].
Recently,
new scaffolds for tissue engineering based on native tissues have become an
attractive option. The primary objectives of preparing a decellularized extracellular
matrix (ECM) are to eliminate tissue immunogenicity and retain the
three-dimensional spatial structure of the ECM of native tissues[5]. Acellular porcine
corneas (APCs) are composed of natural stromal proteins that exhibit reasonable
structural characteristics. Several research groups have succeeded in preparing
a porcine acellular corneal stroma using detergent and/or several enzymes[6-11].
The
five largest eyes in the vertebrate kingdom are those of the whale, elephant,
zebra, giraffe and ostrich. The axial length of the eye in these species ranges
from 54 mm in the baleen whale to 39 mm in the ostrich[12]. The ostrich cornea is large enough to be trimmed
to fit the human eye and ostrich corneas are an abundant resource. The goal of
this study was to use an acellular ostrich cornea (AOC) stroma to replace an
APC as a new scaffold to construct a tissue-engineered cornea (TEC). We hope
that the AOCs will prove to be a potential solution to the short supply of
donor corneas.
MATERIALS AND METHODS
Animals Whole ostrich
eyes (either gender, 12 months old, weighing 60-70 kg) and Yorkshire Landrace
pig eyes (either gender, 6 months old, weighing 120-150 kg) were obtained
within 1-3h of postmortem and subjected to a decellularization procedure within
2h of receipt. The native ostrich corneas/porcine corneas with 2 mm scleral
rings were removed with a pair of curved scissors. Young adult New Zealand
white rabbits (either gender, 10 weeks old, weighing 2-3 kg) were used as
animal transplant models. All animal experiments conformed to the Association
for Research in Vision and Ophthalmology statement for the use of animals in
ophthalmic and vision research.
Preparation of Acellular
Ostrich Corneas The above
corneoscleral tissues were rinsed three times with phosphate buffered saline
(PBS). Then, a lamellar cornea stroma with a diameter of 12 mm ring and
thickness of 400 microns was acquired by scaled trephine under an
ophthalmologic microscope (Olympus, Japan). Subsequently, the lamellar cornea
was soaked in hypertonic saline solution with 20% NaCl (w/v) for 48h at 37℃. Next, the corneal grafts were immersed in 0.13% trypsin
solution (GIBCO,USA) or trypLE™ Express (1×) solution (GIBCO) for 48h at 37℃ and then washed in ultrapure water 3
times for 30min each time. Finally, the grafts were put into a sealed dry
container and dehydrated with calcium chloride for 1-2d at room temperature.
The prepared AOCs were sealed in sterile plastic envelopes, sterilized by
g-irradiation (25 kGy) and stored at 4℃ until used.
Hematoxylin and Eosin Staining Native corneas
(ostrich, human and porcine) and transplanted corneas were collected and
examined with hematoxylin and eosin (H&E) staining. The tissues were
wax-embedded, processed routinely and examined using light microscopy after
H&E staining.
Transmission Electron Microscopy Native ostrich,
human and porcine corneas were collected and examined with transmission
electron microscopy (TEM). Specimens were fixed in 2.5% glutaraldehyde in PBS,
washed three times in PBS for 15min each time, and post-fixed for 2h in 2%
aqueous osmium tetroxide. They were washed three more times in PBS before being
passed through a graded ethanol series. For the purposes of TEM, the specimens
were embedded in an epoxy resin (Agar Scientific, Ltd., Stansted, UK).
Ultrathin (70 nm) sections were collected on copper grids and stained for 1h
with uranyl acetate and 1% phosphotungstic acid and then for 20min with
Reynolds’ lead citrate before being examined with a transmission electron
microscope (Philips CM10).
Rheology
Measurements of the rheological properties were carried
out for natural ostrich corneas (n=5) and AOCs (n=5). A
controlled shear stress rheometer (Anton Paar, MCR302, Austria) was used for
the measurement. The real part of the rigidity modulus, G’ is the elastic or storage modulus and its imaginary part, G’’ is the viscous or loss modulus. Two
types of oscillatory measurements were performed: 1)frequency oscillatory
measurement: a declining frequency from 100 to 0.1 Hz was applied on the
rotation. As the applied frequency declines, the stress increases; 2) strain
oscillatory measurement: a sinusoidal strain from 0.01 to 100 Hz was applied on
the rotation. As the applied strain increases, the stress increases.
Assessment of Mechanical Properties of Different
Grafts According
to a previously reported method[13],
the maximum static tension was measured using an Instron electromechanical
universal tester (Instron, UK) equipped with Bluehill 2.35 software (n=8).
The specimens were kept wet using PBS and cut into 10×4 mm2 rectangular strips. The load range was
set to 0-100 mol/L, and the crosshead speed was 0.3 mm/min. Native ostrich
corneas were used as a control (n=8).
Cytotoxicity of Extractable Materials To determine
whether extracts from the AOCs would cause cytotoxicity, each scaffold (10 mm
diameter) was extracted using a 5 mL 1:1 mixture of Dulbecco’s minimal
essential medium and Ham’s F12 medium containing 10% fetal bovine serum
(culture medium) at 37°C for 48h.
Primary rabbit corneal keratocytes (1×103),
epithelial (2×103)
and endothelial (2×103) cells were separately seeded into
each well of 96-well plates then cultured with extracts (the experimental
group, n=5) and above culture medium (control group, n=5). The
proliferation activity of the cells was quantitatively determined at 1, 3, 5,
and 7d by an MTT assay. The optical density (OD) value of the absorbance at 490
nm was measured with a microplate reader (Rayto, RT2100). The differences in
the OD values between the experimental and control groups were subjected to
statistical analysis.
Xenograft
Lamellar Keratoplasty Lamellar
keratoplasty was used to evaluate the therapeutic effects. To perform lamellar
keratoplasty, a circular incision with a depth of approximately 300 mm was made
in the right eye. The anterior lamellar stroma was then dissected using a fine
operating knife. AOCs were fixed into the recipient bed with interrupted 10-0
sutures. The control group of five rabbits was implanted with APCs.
Immunofluorescence For histological
analysis, the corneas were harvested from the transplanted rabbits and examined
by immunofluorescence. Prior to immunocytochemistry, each section (5 µm thick)
was incubated overnight at 4°C with primary
antibodies against Cytokeratin 3 (CK3, 1:50; Millipore). Fluorescein
isothiocyanate (FITC)-labeled secondary antibodies (1:50; Sigma) were used for
1h at room temperature. Sections were co-stained with
4',6-diamidino-2-phenylindole (DAPI, Sigma) and observed by fluorescence
confocal microscopy (Xcellence-DSU, Olympus, Japan).
Statistical Analysis All data are
expressed as the mean±SD. A Student’s t-test (unpaired) was performed
with Microsoft Excel to analyze the data. The results are presented as the mean
of three individual experiments with the standard error of the mean and a P-value
less than 0.05 was considered significant.
RESULTS
Anatomical Structure of the Cornea Comparison of
the corneal anatomical structure of the ostrich (cell layers of the central
epithelium, the thickness of the central cornea, refractive power) to that of a
human or pig showed that the ostrich cornea is most similar to that of the
human. Another advantage is that the ostrich corneal area is larger and is more
readily available (Table 1).
Table 1 The comparison of
anatomical considerations of the cornea
|
Contents |
Ostrich (n=10) |
Human (n=10) |
Porcine (n=10) |
|
Cell layers of
central corneal epithelium |
4-6 |
5 |
7-9 |
|
The full thickness of central
cornea (μm) |
550±35 |
525±25 |
689±68 |
|
Refractive power (Diopter) |
44.1 |
43.0 |
40.3 |
|
The axial
length of eye (mm) |
38.7 |
23.7 |
21.7 |
Comparing
the corneal anatomical structure of ostrich, human and porcine, the ostrich is
closer to human.
Histological Structure of the Acellular Corneal
Stroma and Natural Human Cornea The
histological structure examination showed that all the corneas had similar
lamellar structures. The thickness of the ostrich cornea was closest to that of
a human (Figure 1A, 1E), and the thickest was the porcine cornea (Figure 1I).
The ostrich and human corneas had well-developed anterior corneal elastic
layers (Figure 1C, 1G, indicated with asterisks, its thickness shown with
double arrows) and base membranes (single arrows). The anterior corneal elastic
layer of porcine corneas was hypogenic (Figure 1K, shown by asterisks, its
thickness was very thin), while the base membrane was completely developed
(single arrows). Corneal stromal collagen fibers of ostrich and human corneas
were arranged regularly and formed oriented lamellar structures (Figure 1D,
1H). The anterior collagen fibers of the porcine corneal stroma were disordered
(Figure 1L), while the posterior collagen fibers were relatively ordered.

Figure
1 The histological structure examination
Ostrich cornea (A and B is H&E staining, C and D is
TEM); human cornea (E and F is H&E staining, G and H is TEM); porcine
cornea (I and J is H&E staining, K and L is TEM).
Preparation
of the Acellular Ostrich Corneas and
Histological Examination The
AOCs were decellularized using a hypertonic saline solution combined with an
enzyme/trypLE™ Express digestion method. The H&E staining showed that the
gaps between the collagen fibers after trypsin digestion (Figure 2B, denoted by
the star symbol) were larger than the gaps when digested by trypLE™ Express
(Figure 2A, shown by stars). The trypsin-digested scaffolds also retained much
more cell debris (Figure 2B, shown with single arrows) compared to the trypLE™
Express-digested group (Figure 2A).

Figure 2 The H&E staining of AOCs
after trypLE™ Express digestion (A) and trypsin digestion (B) Magnification ×400.
Rheology
The represented the storage
modulus, which characterized the elastic property of the scaffolds. The is the
loss modulus, which characterized the viscosity of the scaffolds. The rheology
results of the scaffolds revealed that there were no significant differences
before and after decellularization. Representative experimental results were
shown in Figure 3.
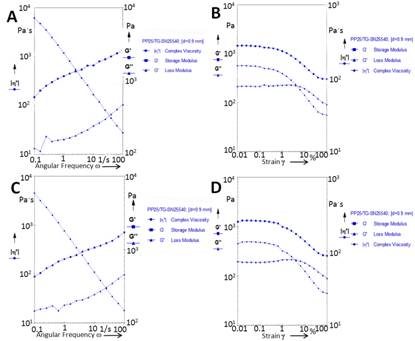
Figure
3 The rheology test of the natural ostrich cornea (A&B) and AOCs (C&D) Fixed amplitude and frequency swing
(A&C), fixed frequency and amplitude swing (B&D). The rheology results
of the scaffolds revealed that there were no significant differences before and
after decellularization.
Assessment of Mechanical Properties of the
Different Grafts The
maximum static tension of the natural ostrich corneal grafts was 14.1±2.3 mol/L
(n=8), and the maximum static tension of the ostrich acellular grafts was
13.9±2.5 mol/L (n=8). There was no significant difference between these
two groups (P>0.05). Representative experimental results are shown in
Figure 4.
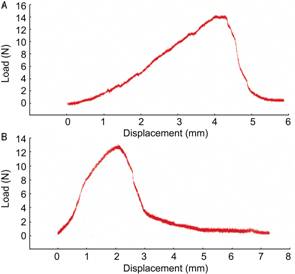
Figure 4 The
typical experimental results of the maximum static tension of natural ostrich
corneas (A) and the AOCs (B).
Cytotoxicity of Extractable Materials There were no significant differences in the proliferation of rabbit
corneal epithelial and endothelial cells or keratocytes between the
experimental and control groups as determined by MTT assay (n=5, P>0.05)
(Figure 5).
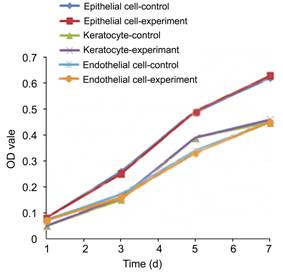
Figure 5 The effect of AOC extracts on the
proliferation of corneal cells.
Xenograft
Lamellar Keratoplasty Six months after the ostrich lamellar keratoplasty,
the grafts remained completely transparent, they did not dissolve and there
were no obvious immune rejection reactions, infections, interlayer hematoceles,
interlamellar dropsies or broken line situations (Figure 6C). Seven to fourteen
days following the porcine lamellar keratoplasty, corneal turbidity occurred in
a few cases (Figure 6D, 6E). After 30d, some grafts began to dissolve, which
was revealed by fluorescein staining (Figure 6F).
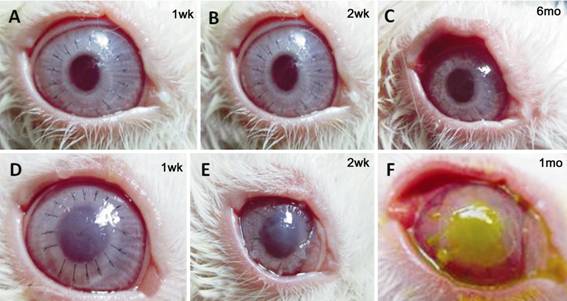
Figure 6 Representative images of the
process of the restoration of transparency after transplantation The AOCs transplantation group (A-C) and
APCs transplantation group (D-F). Six months following the ostrich
lamellar keratoplasty, the graft remains completely transparent (C). Seven to
fourteen days following the porcine lamellar keratoplasty, corneal turbidity
occurred in a few cases (D, E). After 30d, some grafts began to dissolve which
was revealed by fluorescein staining (F).
Immunofluorescence Cytokeratin 3 (CK3), often used as a specific marker in corneal
epithelial cells, was highly expressed in the superficial epithelial cells but
was not found in the basal epithelial cells (Figure 7C). This type of CK3
expression pattern is similar to the normal corneal immune phenotype.
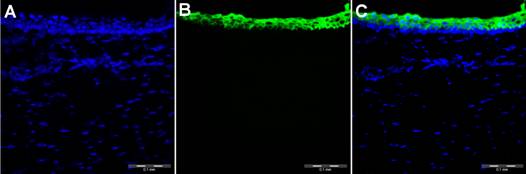
Figure 7 Immunofluorescent staining and laser scanning
confocal microscopy of a corneal epithelium A:
4',6-diamidino-2-phenylindole stained the nucleus; B: CK3 stained the
cytoplasm; C: The merged color image. CK3 was highly expressed in the
superficial epithelial cells but was not found in the basal epithelial cells.
This type of CK3 expression pattern is similar to the normal corneal immune phenotype.
DISCUSSION [Top]
Recently,
there have been many reports on acellular porcine corneal grafts[6-11,14-19]. Compared with
porcine eyes, the ostrich eye is larger and has better eyesight. More
importantly, the anatomical structures and the diopter of ostrich eyes are
closer to those of human eyes (Table 1, Figure 1). Firstly, the basic structure
of corneas is a lamellar structure but the porcine cornea is thicker and has
more epithelial cell layers and the thickness and epithelial cell layers of
ostrich corneas are closer to those of human corneas. Secondly, the porcine
corneal Bowman’s layer structure is dysplastic, while both the ostrich and
human corneas have fully developed Bowman’s layers (Figure 1). The Bowman’s
layer may play a role in maintaining the corneal surface shape and structural
stability[20]. Thirdly, the
human and ostrich anterior corneal collagen fibers are regularly arranged but
the porcine collagen fibers of the anterior corneal stroma are irregularly arranged.
The orientation degree of the corneal collagen fibers is positively correlated
to the corneal transparency[21-23]
and the lamellar corneal transplantation mainly uses the anterior corneal
stroma. Using the principle that structure determines function, the potential
application prospects of ostrich acellular corneal scaffolds in
tissue-engineered corneas are better than those of porcine acellular corneal
scaffolds.
Corneal
transplant surgeries also revealed that the mechanical properties of APCs became
weak after rehydration; in some cases, the APCs were unable to tolerate the
suture operation. Even after transplantation, the cornea initially retained
transparency but approximately 10d after the operation, corneal turbidity
occurred and after 30d, part of the scaffold had dissolved (Figure 6). In
contrast with the rehydrated APCs, the rehydration of AOCs acquired a high
transparency and there was no significant difference in the mechanical
properties (elasticity, viscosity and maximum static tension) before and after
decellularization (Figures 3, 4). The rehydrated AOCs were able to withstand
the suture operation. Six months after the operation, the cornea maintained
complete transparency and integrity (neither neovascularization nor degradation
was observed), suggesting a very low immunogenicity (Figure 6).
In previous
studies, many proteases and chemical reagents (trypsin, dispase, sodium dodecyl
sulfate and sodium hydroxide)[6-8,15]
were used to decellularize the corneal cells. Although the corneal cells were
removed completely and good biocompatibility was observed[14], the decellularization reagents inevitably altered
the extracellular matrix (ECM) composition (the process may remove laminin,
fibronectin and glycosaminoglycans[5]
and cause a certain degree of ultra-structural disruption) [15]. In this study, we used a
gentle decellularization method (hypertonic saline solution combined with
trypLE™ Express digestion) to prepare the acellular corneal scaffolds. This
study chose trypLE™ Express to replace traditional trypsin because 1) trypLE™
Express is non-animal-derived and of a high purity and the degradation of the
ECM is minimal. Trypsin is derived from animals, thus, there are differences
between batches and the collapsing force of the ECM is strong; 2) trypLE™
Express is mild and does not require an enzyme inhibitor; 3) the digestion
kinetics and shear specificity between trypsin and trypLE™ Express are similar
but the stability of the latter is significantly higher than that of the
former. Trypsin-treated scaffolds easily incur differences between batches.
ACKNOWLEDGEMENTS [Top]
Foundations: Supported by National Natural Science
Foundation of China (No. 31200724); Key Innovation Project of Shaanxi Science
and Technology Plan (No. 2012KTCQ03-11); Shenzhen Peacock Plan (No.
KQCX20130628155525051); Projects of Basic Research of Shenzhen (No.
JCYJ20120614193611639, No. JCYJ 20140509172959988).
Conflicts of Interest: Liu XN, None; Zhu XP,
None; Wu J, None; Wu ZJ, None; Yin Y, None; Xiao XH,
None; Su X, None; Kong B, None; Pan SY, None; Yang H,
None; Cheng Y, None; An N, None; Mi SL, None.
REFERENCES [Top]
1 Levis
HJ, Kureshi AK, Massie I, Morgan L, Vernon AJ, Daniels JT.
Tissue engineering the cornea:
the evolution of RAFT.<ii> J Funct Biomater</ii>
2015;6(1):50-65. [CrossRef]
2 Harkin DG, George KA, Madden PW, Schwab IR, Hutmacher DW, Chirila TV.
Silk fibroin in ocular tissue reconstruction.
<ii>Biomaterials</ii> 2011;32(10):2445-2458. [CrossRef] [PubMed]
3 Mi S, Chen B, Wright B, Connon CJ. Ex vivo construction of an
artificial ocular surface by combination of corneal limbal epithelial cells and
a compressed collagen scaffold containing keratocytes. <ii>Tissue
Eng Part A</ii> 2010;16(6): 2091-2100. [CrossRef] [PubMed]
4 Feng Y, Foster J, Mi S, Chen B, Connon C. Influence
of substrate on corneal epithelial cell viability within ocular surface models.
<ii>Exp Eye Res</ii> 2012;101:97-103. [CrossRef] [PubMed]
5 Sanchez PL, Fernandez-Santos ME, Costanza S, <ii>et
al</ii>. Acellular human heart matrix: A critical step toward whole
heart grafts. <ii>Biomaterials</ii> 2015;61:279-289. [CrossRef] [PubMed]
6 Wu Z, Zhou Y, Li N, Huang M, Duan H, Ge
J, Xiang P, Wang Z. The use of phospholipase A2 to prepare acellular
porcine corneal stroma as a tissue engineering scaffold.
<ii>Biomaterials</ii> 2009;30(21):3513-3522. [CrossRef] [PubMed]
7 Hashimoto Y, Funamoto S, Sasaki S, Honda T, Hattori S, Nam K, Kimura
T, Mochizuki M, Fujisato T, Kobayashi H, Kishida A.
Preparation and characterization of decellularized cornea using high-hydrostatic
pressurization for corneal tissue engineering.
<ii>Biomaterials</ii> 2010;31(14):3941-3948. [CrossRef] [PubMed]
8 Pang K, Du L, Wu X. A rabbit anterior cornea replacement derived from
acellular porcine cornea matrix, epithelial cells and keratocytes.
<ii>Biomaterials </ii> 2010;31(28):7257-7265. [CrossRef] [PubMed]
9 Lynch AP, Ahearne M. Strategies for
developing decellularized corneal scaffolds.Exp Eye Res. 2013;
108: 42-7. [CrossRef]
10 Diao JM, Pang X, Qiu Y, Miao Y, Yu MM, Fan TJ.
Construction of a human corneal stromal equivalent with non-transfected human
corneal stromal cells andacellular porcine corneal stromata.
<ii>Exp Eye Res</ii> 2015; 132:216-224. [CrossRef] [PubMed]
11 Xu YG, Xu YS, Huang C, Feng Y, Li Y, Wang W. Development of a rabbit
corneal equivalent using an acellular corneal matrix of a porcine substrate.
<ii>Mol Vis</ii> 2008;14:2180-2189. [PMC free article] [PubMed]
12 Kiladze AB. Structural organization of anterior corneal epithelium of
the African ostrich eye.<ii> Morfologiia</ii> 2013;143(1):32-36. [PubMed]
13 Mi S, Dooley EP, Albon J, Boulton ME, Meek
KM, Kamma-Lorger CS. Adhesion of laser in situ keratomileusis-like flaps
in the cornea: Effects of crosslinking, stromal fibroblasts, and cytokine
treatment. <ii>J Cataract Refract Surg</ii> 2011;37(1):166-172. [CrossRef] [PubMed]
14 Luo H, Lu Y, Wu T, Zhang M, Zhang Y, Jin Y. Construction of tissue
engineered cornea composed of amniotic epithelial cells
and acellularporcine cornea for treating corneal alkali burn.
<ii>Biomaterials </ii> 2013;34 (28):6748-6759. [CrossRef] [PubMed]
15 Zhu J, Zhang K, Sun Y, Gao X, Li Y, Chen Z, Wu X. Reconstruction of
functional ocular surface
by acellular porcine cornea matrix scaffold and limbal stem
cells derived from human embryonic stem cells. <ii>Tissue Eng Part
A</ii> 2013;19(21-22):2412-2425. [CrossRef] [PubMed]
16 Huang M, Li N, Wu Z, Wan P, Liang X, Zhang W, Wang X, Li C, Xiao J,
Zhou Q, Liu Z, Wang Z. Using acellular porcine limbal stroma for rabbit limbal
stem cell microenvironment reconstruction. <ii>Biomaterials</ii>
2011; 32(31):7812-7821. [CrossRef] [PubMed]
17 Xiao J, Duan H, Liu Z, Wu Z, Lan Y, Zhang
W, Li C, Chen F, Zhou Q, Wang X, Huang J, Wang Z.
Construction of the recellularized corneal stroma using porous acellular
corneal scaffold. <ii>Biomaterials </ii> 2011;
32(29):6962-6971. [CrossRef] [PubMed]
18 Zhang MC, Liu X, Jin Y, Jiang DL, Wei XS, Xie
HT. Lamellar keratoplasty treatment of fungal corneal ulcers
with acellular porcine corneal stroma. <ii>Am J
Transplant</ii> 2015;15(4):1068-1075. [CrossRef]
19 Shao Y, Yu Y, Pei CG, Zhou Q, Liu QP, Tan G, Li JM, Gao GP, Yang L.
Evaluation of novel decellularizing corneal stroma for cornea tissue
engineering applications. <ii>Int J Ophthalmol</ii> 2012;5(4):415-418.
[PMC free article] [PubMed]
20 Wang Q, Li W. Advancement of Bowmanps membrane of cornea. <ii>Int
J Ophthalmol </ii> 2009;9 (12):2353-2356.
21 Chen S, Mienaltowski MJ, Birk DE. Regulation of corneal
stroma extracellular matrix assembly. <ii>Exp Eye Res</ii>
2015;133:69-80. [CrossRef] [PubMed]
22 Hassell JR, Birk DE.
The molecular basis of corneal transparency.
<ii>Exp Eye Res </ii> 2010;91(3):326-335. [CrossRef]
23 Qazi Y, Wong G, Monson B, Stringham J, Ambati BK.
Corneal transparency: genesis, maintenance and dysfunction.
<ii>Brain Res Bull</ii> 2010;81(2-3):198-210. [CrossRef] [PubMed] [PMC free article]
[Top]