・Basic Research・・Current Issue・ ・Achieve・
・Search Articles・ ・Online
Submission・ ・About IJO・
Effect of cytokeratin
17 on retinal pigment epithelium degeneration and choroidal neovascularization
Yi Shen, Pei Zhuang, Tao Xiao, George
CY Chiou
Institute of Ocular Pharmacology,
College of Medicine, Texas A&M Health Science Center, College Station, TX 77843, USA
Correspondence to: George CY Chiou. Institute of Ocular Pharmacology,
College of Medicine, Texas A&M Health Science Center, College Station, TX
77843, USA. chiou@medicine.tamhsc.edu
Received: 2015-07-29
Accepted: 2015-11-26
Abstract
AIM: To study the effects of cytokeratin 17 (CK17) on sodium
iodate (NaIO3) induced rat retinal
pigment epithelium (RPE) degeneration, laser induced rat choroidal
neovascularization (CNV), and oxidative stress of human retinal pigment
epithelium cells (ARPE-19) and human umbilical vein
endothelial cell (HUVEC).
METHODS: Thirty 8-week-old
male Brown Norway rats were randomly divided into 3 groups, 10 rats
in control group treated with solvent
alone; 10 rats in NaIO3 group treated with
solvent and 35 mg/kg NaIO3 injection
through hypoglossal vein and 10 rats in CK17+NaIO3 group treated with 1% CK17 eye
drop 3 times a day for 1wk before and 4wk after NaIO3 injection. RPE function was
measured with c-wave of electroretinogram (ERG). Another 20 rats were randomly
divided into 2 groups. Of them 10 rats in CK17 group
were anesthetized to receive Nd:YAG laser and given 1% CK17
eye drop before same as above; 10 rats in control were received Nd:YAG and treated with solvent.
The development of choroidal neovascularization (CNV) was
determined by fundus fluorescein angiography (FFA) performed on
4wk
after laser. Methylthiazoly tetrazolium (MTT) assay was used to
study effect of CK17 on various oxidants induced injury in ARPE-19 and HUVEC in vitro.
RESULTS: Four weeks
after NaIO3 injection, the c-wave amplitude of ERG was 0.393±0.02 V
in the control group, 0.184±0.018 V in NaIO3 group and 0.3±0.01 V in CK17+NaIO3
group. There was a significant reversal of the c-wave by CK17 as compared to
NaIO3 group (P<0.01). Four
weeks after laser, the size of the CNV lesion was 2.57±0.27 mm2 in
control group and 1.64±0.08 mm2 in CK17 group. The lesion
size significantly diminished in CK17 group (P<0.01). The in vitro results showed CK17 also reversed the various
oxidants induced injuries in ARPE-19 at the dose of 100 μg/mL and enhanced the injury in
HUVECs at different concentrations.
CONCLUSION: CK17 can
significantly protect RPE from NaIO3 induced degeneration in vivo and in vitro and also could
reverse the various oxidants induced injuries in vitro. It inhibits the
development of CNV in rat model, interfered with vascular endothelial cell
proliferation in vitro.
KEYWORDS: cytokeratin
17; age-related macular degeneration; choroidal neovascularization; retinal
pigment epithelium; human umbilical
vein endothelial cells
DOI:10.18240/ijo.2016.03.06
Citation: Shen Y, Zhuang P, Xiao T, Chiou GCY. Effect of
cytokeratin 17 on retinal pigment epithelium degeneration and choroidal
neovascularization. Int J Ophthalmol 2016;9(3):363-368
INTRODUCTION
Retinal degenerative disease can lead
to blindness. Age-related macular degeneration (AMD) is the most
common retinal degenerative diseases[1]. A century ago, the AMD has been
described on the medical literature, but until the last century 70's, there has been a description
of the treatment of AMD. About 30% of adults aged 75y or older have
some signs of maculopathy, and 6% to 8% of these individuals are afflicted with
the advanced stages of AMD[2]. According to reports, in the United States AMD
accounted for 54% of the current blindness cases among the
Caucasian[3].
Single layer of retinal pigment
epithelium (RPE) separates the retinal nerve sensory layer and choroid. Its
main function is to provide nutrition and phagocytose the outer
segments of adjacent photoreceptors[4]. The RPE disable to remove the metabolic waste results
in the accumulation of drusen. RPE dysfunction causes the breakdown of the
Bruch's membrane. The breaks in Bruch’s membrane under the detached RPE
serve as an entrance for new and immature choroidal vessels into the subretinal
space that lead to the formation of choroidal neovascularization (CNV).
Furthermore, RPE loss may cause loss of choriocapillaris[5]. Therefore, RPE
can be used as a target for improving photoreceptor survival and treatment
related ocular diseases[6].
Cytokeratin has been used as a marker for
epithelial cells. They help in providing architecture and a definite cellular
organisation to the tissue as a whole, by controlling morphogenetic migration
of cells[7]. Cytokeratin
abnormal expression will result in cell dysfunction. This study is
to observe the effects of 1% cytokeratin 17 (CK17) on sodium iodate
(NaIO3) induced RPE degeneration, laser
induced choroidal neovascularization, and oxidative stress of RPE cells and human
umbilical vein endothelial cells (HUVECs).
MATERIALS AND
METHODS
Retinal
Pigment Epithelium Degeneration Model in Rat Eyes All animals
used in this study were bred in a colony at the University of Utah, and
maintained under a 12h light/dark cycle. They were housed and handled with the
authorization and supervision of the Institutional Animal Care and Use
Committee from the Texas A&M Health Science Center.
A total of 30 8-week-old male
Brown-Norway rats were randomly divided into 3 groups, 10 rats in control group, 10
rats in NaIO3 group and 10 rats in CK17+NaIO3 group.
Control group was treated with solvent (Dulbecco’s phosphate
buffered saline; DPBS) eye drop alone throughout the trial without NaIO3
(Sigma-Aldrich
Chemical Co., MO, USA) injection. NaIO3 group was treated with solvent
eye drop and 35 mg/kg NaIO3 hypoglossal
vein injection, whereas CK17+NaIO3 group was treated with 1% CK17 (Sigma-Aldrich
Chemical Co., MO, USA) eye drop and NaIO3
injection. All eye drops were instilled both eyes with 1 drop
for 3 times a day for 1wk before and 4wk after NaIO3
injection. At the end of 4wk, all rats were measured with c-wave of electroretinogram
(ERG).
Rats were dark adapted for 2h, and
then anesthetized with ketamine 35 mg/kg plus xylazine 5 mg/kg intramuscular injection.
Half of the initial dose was given each 1h thereafter.
Pupils of all rats were dilated with one drop of 1% atropine and 2.5% phenylephrine.
ERG recording methods developed by Peachey et al[8] were
followed. Briefly, Silver chloride electrode is arranged
on the surface of the binocular cornea, and the stainless steel needle
electrode was used as the reference electrode and the ground
electrode, which were respectively arranged on the same
side of the cheek and the tail. Responses were amplified (dc-100Hz; gain 1000×; DP-31,
Warner Instruments, Hamden, CT, USA). Data were analyzed by iWORX
LabScribe Data Recording Software (iWorxoCB Sciences,
Dover, NH, USA).
Choroidal
Neovascularization Model in Rat Eyes A total of 20
8-week-old male Brown-Norway rats were randomly divided into 2 groups, 10 rats
in control group, 10 rats in CK17 group. CK17 group was instilled with 1% CK17 eye drops and control group
was treated with solvent (DPBS) eye drop. Both eyes of all rats were
instilled with 1 drop for 3 times a day for 1wk before and 4wk after
laser.
Rats were anesthetized and the pupils were
dilated as mentioned above. The fundus was visualized with a VOLK super
pupil XL biomicroscopy lens (Keeler Instrument Inc., Broomall, PA,
USA). A double-frequency Nd:YAG laser (Laserex LP3532; Lumenis Inc., Salt Lake
City, UT, USA) was used at 532 nm wavelength.
Laser parameters used were 100 μm spot size, 0.15s exposure and
l50-200 mw powers. Five laser spots were made to the ocular
fundus at approximately equal distances around the optic nerves. Acute vapor
bubbles suggested rupture of Bruch’s membrane[9]. Only laser
spots with bubble formation were included in the study. Lesions with subretinal
hemorrhage were excluded.
Fundus fluorescein angiography (FFA) was performed
4wk after laser treatment with a digital fundus camera (TRC-50EX; TOPCON,
Tokyo, Japan). Ten percent fluorescein sodium salt was injected through
hypoglossal vein at 0.5 mL/kg. Both early (under 2min) and late
(over 7min) fluorescein phases were captured. Ten percent fluorescein
isothiocyanate-dextran was injected through hypoglossal vein at l.4 mg/kg after 3d
of fluorescein sodium salt injection. Fluorescein pictures were taken within
20min. The clearest pictures were chosen for the areas of CNV formation
measurement by IMAGEnet2000 digital imaging system (Topcon Medical Systems,
Inc., Paramus, NJ, USA).
Cell Culture Human retinal
pigment epithelium cells (ARPE-19; ATCC, Manassas, VA,
USA) were grown in Dulbecco’s modified Eagle’s medium/Ham’s F12
(DMEM/F12, 1:1; Sigma-Aldrich Chemical Co., MO, USA) supplemented with
10% fetal bovine serum (FBS; ATCC, Manassas, VA,
USA), 100 units/mL
penicillin G, and 100 µg/mL streptomycin sulfate. HUVECs (ATCC, Manassas, VA,
USA) were grown in vascular cell basal medium supplemented with endothelial
cell growth kit (ATCC, Manassas, VA,
USA). Cells were
incubated in a humidified incubator at 37℃ under 5% CO2
and 95% air.
Cells were allowed to attach
overnight, and then exposed to CK17 or vehicle solution under hypoxic condition
for 72h. Hypoxic conditions (1% O2, 5% CO2 and 94% N2)
were maintained by using a temperature and humidity controlled environmental
C-chamber by O2 and CO2 controllers (Proox Model 110 and
Pro CO2 Model 120, Bio Spherix Ltd., Redfield, NY, USA) with N2
and CO2 gas sources. Thiazolyl blue tetrazolium bromide (MTT) assay (Sigma-Aldrich
Chemical Co., MO, USA) was used to measure the viability of
ARPE-19 and HUVECs. Cells (1×105) were seeded
in 96-well plates (100 µL/well) and allowed to grow overnight. Negative control
was prepared by adding 100 µL medium without cells. The cells were then treated
with fresh medium with CK17 [CK17 was dissolved in 30%
2-hydroxypropyl-β-cyclodextrin (HP-β-CD), the final
concentration of HP-β-CD in cells is less than 0.3%] and/or
oxidizing agents (NaIO3/H2O2/NaN3/t-BHP) at the same
time for 12, 24, or 72h (200 µL/well). The vehicle control group was treated
with 30% HP-β-CD with fresh medium (the final concentration of HP-β-CD in cells
is less than 0.3%). MTT (5 mg/mL; 20 µL) was added to wells, and
incubated for another 4h. After incubation, the medium was discarded and 100 µL
DMSO was added to solubilize formazan produced from MTT by the viable cells.
Absorbance was measured at 570 nm using a microplate reader (Bio-Rad
Laboratories, Inc., CA, USA). Cells viability was calculated
according to the following formula: Viability of cells (%)=(absorbance in
tested sample-absorbance in
negative control)/(absorbance in vehicle control-absorbance in
negative control)×100%.
Statistical
Analysis All data were
presented as mean±SEM. A nonpaired Student's t-test was perfonned to analyze the significance between two groups at a certain
time point. Analysis of variance for comparison between three groups and more. The
differences were considered significant at P<0.05.
RESULTS
Electroretinogram
Recordings Four weeks
after NaIO3 injection, the amplitude of ERG c-wave was 0.393±0.02 V
in the control group, 0.184±0.018 V
in the NaIO3 group, 0.3±0.01 V in the CK17+NaIO3
group. There was a significant reversal of the ERG c-wave by CK17 as compared
to NaIO3 group (P<0.01) (Figure 1).
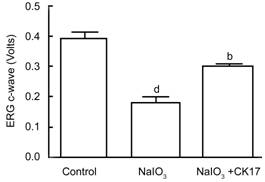
Figure 1 ERG outcomes of CK17 on NaIO3 induced RPE degeneration in rat eyes Data were
expressed as mean±SEM. dP<0.01 vs control group; bP<0.01 vs NaIO3 group.
Fundus
Fluorescein Angiography The angiograms
showed diminished lesion size in CK17 group 4wk after laser. The size of the
CNV lesion was 2.57±0.27 mm2 in control group, 1.64±0.08 mm2
in CK17 group (P<0.01) (Figure 2).
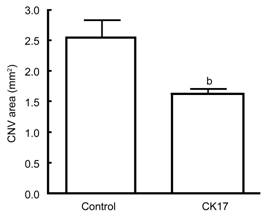
Figure 2 Effect of CK17 on laser induced
CNV rat model Data were
expressed as mean±SEM. bP<0.01 vs control group.
Cytotoxicity
of Cytokeratin 17 on Human Retinal Pigment Epithelium and
Umbilical Vein Endothelial Cells The results
showed that CK17 had no effect on ARPE-19 cells except at the concentration of
0.1 µg/mL it significantly increased the proliferation of ARPE-19 (P<0.05, Figure 3A). At the
concentration of 0.1, 0.3 and 3 µg/mL, CK17 significantly increased the
proliferation of HUVECs (P<0.01,
Figure 3B). However,
CK17 inhibited the proliferation of HUVECs at the concentration of 1, 10 and
100 µg/mL (P<0.01, Figure 3B).
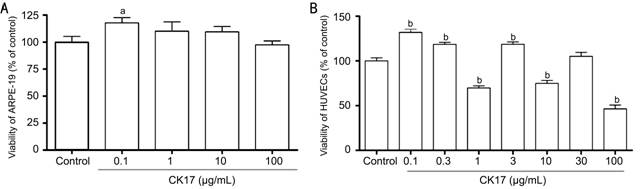
Figure 3 Cytotoxicity
of CK17 on ARPE-19 and HUVECs A: Effect of CK17 on proliferation of
ARPE-19; B: Effect of CK17 on proliferation of HUVECs. Data were
expressed as means±SEM; n=6. aP<0.05, bP<0.01 vs control group.
Effect of
Cytokeratin 17 on Hypoxia-induced Damage in Human Retinal Pigment Epithelium
and Umbilical Vein Endothelial Cells CK17
significantly increased the viability of ARPE-19 cells by 16% in hypoxic
condition at concentration of 100 µg/mL (P<0.01,
Figure 4A). It also
could increase the viability of HUVECs by 15% in hypoxic condition at
concentration of 0.1 µg/mL (P<0.05,
Figure 4B). However,
the other concentration of CK17 had no effect on both ARPE-19 and HUVECs.
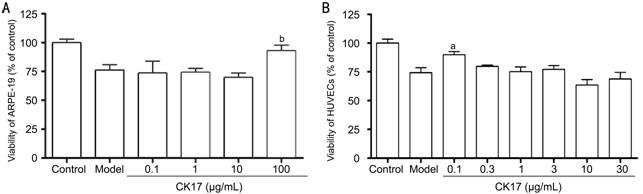
Figure 4 Effect of
CK17 on hypoxia-induced damage in ARPE-19 and HUVECs A: Effect of
CK17 on hypoxia-induced injury in ARPE-19; B: Effect of
CK17 on hypoxia-induced injury in HUVECs. Data were expressed as means±SEM; n=6. aP<0.05 and bP<0.01 vs model group.
Effect of
Cytokeratin 17 on NaIO3-induced Injury in Human Retinal
Pigment Epithelium and Umbilical Vein Endothelial Cells At the
concentration of 100 µg/mL, CK17 significantly increased the viability of 30,
100 and 300 µg/mL NaIO3-induced injury by 23%, 15% and 27% in
ARPE-19 cells, respectively (All P<0.05, Figure 5A). However,
all concentration of CK17 couldn’t reverse NaIO3-induced injury in
HUVECs. On the contrary, it enhanced NaIO3-induced injury in HUVECs especially the concentrations
1 and 10µg/mL (P<0.01,
Figure 5B).
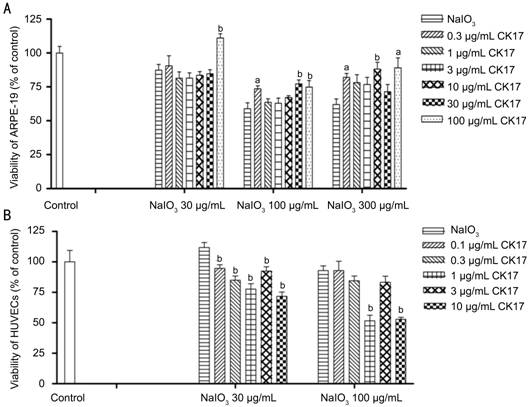
Figure 5 Effect of
CK17 on NaIO3-induced injury in ARPE-19 and HUVECs A: Effect of
CK17 on NaIO3-induced injury in ARPE-19; B: Effect of CK17 on NaIO3-induced
injury in HUVECs. Data were expressed as means±SEM; n=6. aP<0.05 and bP<0.01 vs NaIO3 group.
Effect of
Cytokeratin 17 on H2O2-induced Injury in Human Retinal
Pigment Epithelium and Umbilical Vein Endothelial Cells CK17 with 0.3, 30 and
100 µg/mL reversed 200 µmol/L H2O2-induced
injury by 27%, 39% and 27% in ARPE-19, respectively. However, 1 and 10 µg/mL
CK17 significantly enhanced 400 µmol/L H2O2-induced
injury in ARPE-19 (Figure 6A). CK17 with 1 and 10 µg/mL
significantly enhanced 200 µmol/L H2O2-induced
injury in HUVECs (Figure 6B).
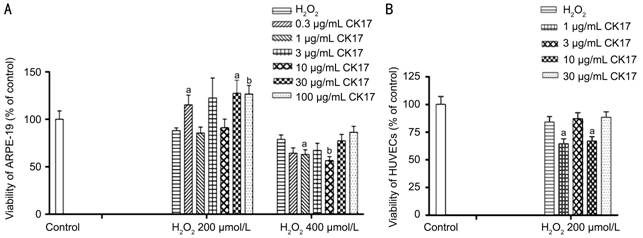
Figure 6 Effect of
CK17 on H2O2-induced injury in ARPE-19 and HUVECs A: Effect of
CK17 on H2O2-induced injury in ARPE-19; B: Effect of CK17
on H2O2-induced injury in HUVECs. Data were expressed as
means±SEM; n=6. aP<0.05, bP<0.01 vs H2O2 group.
Effect of
Cytokeratin 17 on NaN3-induced Injury in Human Retinal Pigment
Epithelium and Umbilical Vein Endothelial Cells CK17 reversed
0.3, 1 and 3 mmol/L NaN3-induced injury in ARPE-19 at the
concentration of 30 and 100 µg/mL (Figure 7A). On the
contrary, 1 and 10 µg/mL CK17 significantly enhanced 0.3, 1 and 3 mmol/L NaN3-induced
injury in HUVECs (P<0.01, Figure 7B).
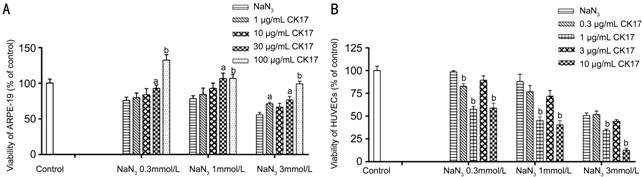
Figure 7 Effect of
CK17 on NaN3-induced injury in ARPE-19 and HUVECs A: Effect of
CK17 on NaN3-induced injury in ARPE-19; B: Effect of CK17 on NaN3-induced
injury in HUVECs. Data were expressed as means±SEM; n=6. aP<0.05
and bP<0.01 vs NaN3 group.
Effect of
Cytokeratin 17 on t-BHP-induced Injury in Human Retinal Pigment Epithelium and
Umbilical Vein Endothelial Cells CK17 reversed
the viability of 100 and 200 µmol/L t-BHP-induced injury in ARPE-19 at
the different concentration (Figure 8A). CK17 had
no effect on 50 µmol/L t-BHP-induced injury in HUVECs (Figure 8B).
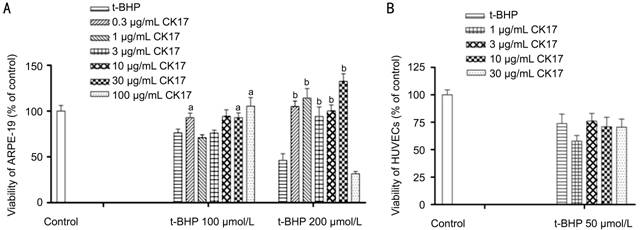
Figure 8 Effect
of CK17 on t-BHP-induced injury in ARPE-19 and HUVECs A: Effect of CK17 on
t-BHP-induced injury in ARPE-19; B: Effect of CK17 on t-BHP -induced injury in
HUVECs. Data were expressed as means±SEM; n=6.
aP<0.05, bP<0.01 vs t-BHP group.
DISCUSSION
There are two clinical types of AMD:
non-exudative or atrophic AMD (dry-AMD), which is characterized by the
degeneration of choriocapillaries, RPE and neurosensoty retina; and neovascular
or exudative AMD (wet-AMD), which is characterized by the development of
serious RPE detachments and/or choroidal neovascularization that can
lead to bleeding, exudation, and eventual scar formation. Although the form of
wet-AMD only accounts for 10%-20% of the
overall incidence of AMD, it is responsible for over 90% of cases with severe
visual loss[10-11]. Most of optical treatments are for the
wet-AMD, not for dry-AMD and for now there is no
effective treatment for the most prevalent dry-AMD[12]. Dry-AMD is
triggered by abnormalities in RPE that lies beneath the photoreceptor cells and
normally provides critical metabolic support to these light-sensing cells.
Secondary to RPE dysfunction, macular rods and cones degenerate leading to the
irreversible loss of vision. Oxidative stress, formation of drusen,
accumulation of lipofuscin, local inflammation and reactive gliosis represent
the pathologic processes implicated in pathogenesis of dry-AMD. The direct
toxic effect of NaIO3 on RPE cells with secondary effects on
photoreceptors and the choriocapillaries in
vivo is well known[13].The mechanisms of the toxicity of Na
'
IO3 to RPE cells are as follows: first, NaIO3
can increase the ability of melanin to convert glycine into glyoxylate, a
potential cell toxic compound[14]; second, NaIO3 could
denaturant retinal proteins by changes of -SH levels in retina[15]; third, NaIO3
could cause considerable structure changes by breakdown of RPE diffusion
barrier or by reduction of adhesion between RPE and photoreceptor cells [16-19]; finally,
NaIO3 inhibits various enzyme activities, such as noice phosphate
dehydragenase, succinodehydrogenase and lactate dehydrogenase[18,20].
The ERG results showed that CK17 can
significantly reverse NaIO3-induced injury in RPE cells. CK17
showed protective effect against NaIO3-induced RPE
degeneration in rat eyes. The NaIO3 intoxication causes death of RPE
cells and photoreceptor damage followed by marked phagocytic activity of
proliferating dedifferentiated pigmental cells. Further morphological study is
needed to reveal the CK17 role in RPE protection. FFA diagraphs
showed that CK17 could significantly decrease the intensity of fluorescein
leakage from the photocoagulated lesions and the size of CNV induced by laser
treatment on Brown Norway rats, and interfered with vascular endothelial cell
proliferation in vitro. Endothelial cells played an important
role in the process of CNV development. CNV is the result of angiogenesis,
which include endothelial cell proliferation, migration, and adhesion. Certain
concentration of CK17 could obviously inhibit the growth of HUVECs. So appropriate
dose of CK17 may inhibit the development of CNV through
regulation of the behavior of endothelial cells directly.
Mitochondria are the powerhouse of the
cell, and their primary function is to generate ATP through oxidative
phosphorylation via the electron
transport chain[21]. Any kind of oxidative stress can be inhibitor of
cytochrome oxidase and catalase, and will downregulate electron transport and O2 consumption to cause the death of cells[22]. Our
experiments showed the protection of CK17 on oxidative stress.
With the change of the concentration of CK, the
biological effects are obviously different. And the different degree of damage
can also lead to the different role of CK. For the next study, we will focus on
finding suitable CK treatment concentrations for different injury options.
In conclusion, CK17 might slow the
oxidative process of RPE cell layer which leads to RPE degeneration. CK17 might
also prevent the formation of CNV. According to the rational of the AMD, the
RPE abnonnalities or degeneration is the key point of both dry- and wet-AMD, so CK17
could be used to prevent and treat both dry- and wet-AMD in the
future.
ACKNOWLEDGEMENTS
Conflicts of Interest: Shen Y, None; Zhuang P, None; Xiao T, None; Chiou GCY, None.
REFERENCES[Top] [Top
1 Lin TC, Chang HM, Hsu CC, Hung KH, Chen YT, Chen SY, Chen SJ. Retinal
prostheses in degenerative retinal diseases. <ii>J Chin Med
Assoc</ii> 2015;78(9):501-505. [CrossRef]
[PubMed]
2 Complications of Age-Related Macular Degeneration Prevention Trial
Study Group. The Complications of Age-Related Macular Degeneration Prevention
Trial (CAPT): rationale, design and methodology. <ii>Clin</ii>
<ii>Trials</ii> 2004;1(1):91-107. [CrossRef]
[PubMed]
3 Eye Diseases Prevalence Research Group. Causes and prevalence of visual
impairment among adults in the United States. <ii>Arch
Ophthalmol</ii> 2004;122(4):477-485. [CrossRef]
[PubMed]
4 Engelmann K, Valtink M. RPE cell cultivation. <ii>Graefes Arch
Clin Exp Ophthalmol </ii> 2004;242(1):65-67. [CrossRef]
[PubMed]
5 Liu CF, Lin CH, Chen CF, Huang TC, Lin SC. Antioxidative effects of
tetramethylpyrazine on acute ethanol-induced lipid peroxidation. <ii>Am J
Chin Med</ii> 2005;33(6):981-988. [CrossRef]
[PubMed]
6 Ohtaka K, Machida S, Ohzeki T, Tanaka M, Kurosaka D, Masuda T, Ishii T.
Protective effect of hepatocyte growth factor against degeneration of the
retinal pigment epithelium and photoreceptor in sodium iodate-injected rats.
<ii>Curr Eye Res</ii> 2006;31(4):347-355. [CrossRef]
[PubMed]
7 Singh S, Gupta PD. Tampering with cytokeratin expression results in
cell dysfunction. <ii>Epithelial Cell Biol</ii> 1994;3(2):79-83. [PubMed]
8 Peachey NS, Stanton JB, Marmorstein AD. Noninvasive recording and
response characteristics of the rat dc-electroretinogram. <ii>Vis
Neurosci</ii> 2002;19(6):694-701. [CrossRef]
9 Zou Y, Xu X, Chiou, GC. Effect of interleukin-1 blockers, CK112, and
CK116 on rat experimental choroidal neovascularization <ii>in
vivo</ii> and endothelial cell cultures <ii>in
vitro</ii>.<ii> J Ocul Pharmacol Ther</ii> 2006;22(1):19-25. [CrossRef] [PubMed]
10 Bressler NM, Bressler SB, Fine SL. Age-related macular degeneration.
<ii>Surv Ophthahnol</ii> 1988;32(6):375-413. [CrossRef]
11 Votruba M, Gregor Z. Neovascnlar age-related macular degeneration:
present and future treatment options. <ii>Eye (Lond)</ii>
2001;l5(Pt 3):424-429.
12 Coleman HR, Chan CC, Ferris FL 3rd, Chew EY. Age-related macular
degeneration. <ii>Lancet</ii> 2008;372(9652):1835-1845. [CrossRef]
13 Kiuchi K, Yoshizawa K, Shikata N, Moriguchi K, Tsubura A. Morphologic
characteristics of retinal degeneration induced by sodium iodate in mice.
<ii>Curr Eye Res</ii> 2002;25(6):373-379. [CrossRef]
14 Baich A, Ziegler M. The effect of sodium iodate and melanin on the
formation of glyoxylate. <ii>Pigment Cell Res </ii>
1992;5(6):394-395. [CrossRef] [PubMed]
15 Sorsby A, Reading HW. Experimental degeneration of the retina. XI. The
effect of sodium iodate on retinal -SH levels. <ii>Vision Res</ii>
1964;4(10):511-514. [CrossRef]
16 Flage T, Ringvold A. The retinal pigment epithelium diffusion barrier
in the rabbit eye after sodium iodate injection. A light and electron
microscopic study using horseradish peroxidase as a tracer. <ii>Exp Eye
Res</ii> 1982;34(6):933-940. [CrossRef]
17 Sen HA, Berkowitz BA, Ando N, de Juan E Jr. In vivo imaging of
breakdown of the inner and outer blood-retinal barriers. <ii>Invest
Ophthalmol Vis Sci</ii> 1992;33(13):3507-3512. [PubMed]
18 Ashbum FS Jr, Pilkerton AR, Rao NA, Marak GE. The effects of iodate
and iodoacetate on the retinal adhesion. <ii>Invest Ophthalmol Vis
Sci</ii> 1980;19(12):1427-1432.
19 Stern WH, Ernest JT, Steinberg RH, Miller SS. Interrelationships
between the retinal pigment epithelium and the neurosensory retina.
<ii>Aust J Ophthalmol</ii> 1980;8(4):281-288. [CrossRef]
20 Enzmann V, Row BW, Yamauchi Y, Kheirandish L, Gozal D, Kaplan HJ,
McCall MA. Behavioral and anatomical abnonnalities in a sodium iodate-induced
model of retinal pigment epithelium degeneration. <ii>Exp Eye
Res</ii> 2006;82(3):441-448. [CrossRef]
[PubMed]
21 Wang ZJ, Liang CL, Li GM, Yu CY, Yin M. Stearic acid protects primaty
cultured cortical neurons against oxidative stress. <ii>Acta Pharmacol
Sin</ii> 2007;28(3):315-326. [CrossRef] [PubMed]
22 Thompson JG, McNaughton C, Gasparrini B, McGowan LT, Tervit HR. Effect
of inhibitors and uncouplers of oxidative phosphorylation during compaction and
blastulation of bovine embryos cultured <ii>in vitro</ii>.
<ii>J Reprod Fertil</ii> 2000;118(1):47-55. [CrossRef]
[Top]