・Clinical Research・・Current Issue・ ・Achieve・ ・Search Articles・ ・Online
Submission・ ・About
IJO・
Comparison of Diaton
transpalpebral tonometer with applanation tonometry in keratoconus
Robert PL Wisse, Natalie Peeters, Saskia
M Imhof, Allegonda van der Lelij
Utrecht
Corneal Research Group Department of Ophthalmology, University Medical
Center Utrecht, Utrecht
3508 GA, The Netherlands
Correspondence to:
Robert PL Wisse.
Office E.03.136, Department of Ophthalmology, University
Medical Center Utrecht, PO
Box 85500, Utrecht 3508 GA, The Netherlands. r.p.l.wisse@umcutrecht.nl
Received: 2015-01-23
Accepted:
2015-06-10
Abstract
AIM: To
investigate the added value of using a Diaton transpalpebral tonometer (DT) to
measure IOP in keratoconus. Most type of tonometers use corneal applanation or
biomechanical resistance to measure intraocular pressure (IOP); however, these
factors can be altered by keratoconus. Specifically, we examined whether DT can
detect false-negative low Goldmann applanation tonometry (AT) measurements.
METHODS: Patients
with keratoconus were recruited from our tertiary academic treatment center.
Measurements included AT and DT (in random order) and Scheimpflug imaging. An
age- and gender-matched group of control subjects with no history of corneal
disease or glaucoma was also recruited.
RESULTS:
In total, 130 eyes from 66 participants were assessed. In the keratoconus
group, mean AT was 11.0 ± 2.6, mean DT 11.2±5.5 (P=0.729), and the two measures were correlated significantly (P=0.006, R=0.323). However, a Bland-Altman plot revealed a wide distribution
and poor agreement between both measurements. Previous corneal crosslinking,
corneal pachymetry, and Krumeich classification had no effect on measured IOP.
CONCLUSION:
Measurements obtained using a Diaton tonometer are not affected by corneal
biomechanics; however, its poor agreement with Goldmann AT values calls into
question the added value of using a Diaton tonometer to measure IOP in
keratoconus.
KEYWORDS: Diaton; Goldmann applanation tonometry;
transpalpebral tonometry; keratoconus;
Bland-Altman plot
Citation: Wisse RPL, Peeters N, Imhof SM, van der Lelij A. Comparison of Diaton
transpalpebral tonometer with applanation tonometry in keratoconus. Int J Ophthalmol
2016;9(3):395-398
INTRODUCTION
The
presence of corneal pathology can potentially affect measurements of
intraocular pressure (IOP) and several methods for measuring IOP in corneal
pathology have been described[1-3].
For example, Rosentreter et al[1]
compared rebound tonometry, applanation tonometry, and dynamic contour
tonometry in pathological corneas. However, all of these devices depend upon
corneal applanation and/or biomechanical resistance. Both factors can be
altered by keratoconus, a progressive condition with thinning of the cornea,
irregular astigmatism, and decreased biomechanical resistance[4-7].
In particular, the thinning of the cornea can be extremely severe; applanation
of such a thin cornea potentially requires much less pressure and can therefore
result in an underestimation of the actual IOP[8].
This effect has been observed when measuring IOP in healthy corneas with
varying corneal pachymetry measurements[9],
and this phenomenon was proposed as a factor in normal-tension glaucoma[10].
Specifically, the irregular shape of the cornea might prevent the Goldmann
applanation tip from aligning properly, thus preventing uniform contact; this
problem is not an issue with other methods (for example, rebound tonometry).
Corneal rigidity can further be altered by corneal crosslinking, a widely used
procedure for preventing the progression of keratoconus[11].
The effect of crosslinking on various IOP measuring methods has been studied,
and these studies revealed increased IOP readings after crosslinking. It is
important to note that all devices depend on corneal rigidity for their
accuracy.
To
circumvent this problem, the Diaton tonometer (DT, manufactured by Ryazan State
Instrumental-Making Enterprise, Ryazan, Russia, http://www.diaton-tonometer.com)
uses an alternate method to measure IOP; the movement pattern of a small rod
falling freely onto the eyelid surface is measured and individual measurements
are displayed digitally. The DT is a portable, hand-held device that measures
transpalpebral IOP through the patient’s upper eyelid while the patient is in a
reclined or supine position. The DT has been promoted as a suitable alternative
method of tonometry for patients with conjunctivitis and/or corneal disease, or
following corneal surgery[12].
Previous research found that the DT is reliable in patients without corneal
disease and provides measurements that are similar to Goldmann applanation
tonometry (AT); however, DT yields results with wider variation and lower
correlation with repeated measurements[13-15].
Thus, the value of using DT for glaucoma screening has been questioned.
Because
applanation IOP measurements in keratoconus patients can underestimate the
actual IOP, and because of the claims made by the manufacturer, we investigated
the added value of measuring transpalpebral IOP using DT compared to Goldmann AT in patients with
keratoconus. Specifically, we examined whether false-negative (i.e. low) AT measurements in keratoconus
can be detected using DT.
SUBJECTS
AND METHODS
Patients
were recruited from the cornea outpatient clinic in our tertiary academic
center from October 2013 through January 2014. The inclusion criteria included
a current diagnosis of keratoconus and no gross anatomical eyelid abnormalities
hampering DT measurement; patients of all ages were eligible for inclusion.
Corneal scarring and/or previous crosslinking treatment did not preclude
patients from participating. An age- and gender-matched control group was
recruited and consisted of healthy volunteers with no history of corneal
disease, ocular hypertension, or glaucoma.
All
measurements were collected by one examiner (Peeters N) under standardized
conditions; DT measurements were taken in the supine position in accordance
with the manufacturer’s instructions. The DT indicates the number of
measurements necessary for each eye and provides a single reading. AT was
measured using standard procedures. The order of IOP measurements (i.e. DT followed by AT versus AT
followed by DT) was randomized. All patients underwent a slit-lamp evaluation
and Scheimpflug corneal imaging (Pentacam HR type 70900, Oculus GmbH) prior to
the IOP measurements. All keratoconus eyes were diagnosed and graded using the
Krumeich classification system[16]
by one corneal specialist (Wisse RPL).
Statistical
analyses were performed using SPSS version 20.0 (IBM). Box plots, scatter
plots, and Bland-Altman plots were used to visualize the outcomes[17].
Differences in AT and DT readings were analyzed using the Student’s t-test. A linear regression model using
a generalized estimating equation (correcting for patients with two affected
eyes) was used to assess the relationship between the difference in IOP and
pachymetry and Krumeich classification. Normality was tested based on skewness
and kurtosis, with a cut-off value of 3.29 (P<0.001).
This
study was approved by our Institution’s Ethics Review Board and was performed
in accordance with the Declaration of Helsinki. None of the eligible
participants refused to participate, and all subjects provided informed
consent.
RESULTS
One hundred and thirty eyes from 66
participants were initially enrolled; 36 keratoconus patients had 70 eyes with
keratoconus. Two eyes from one patient in the keratoconus group were excluded
from the analysis due to missing AT measurements. The mean age (±SD) of the
subjects in the keratoconus and control groups was 25.8±9.3 and 33.1±9.8y,
respectively; 62% and 56% of the subjects were male in the keratoconus and
control groups, respectively. Baseline characteristics did not differ significantly
between the two groups. Among the eyes with keratoconus, 40 (57%) previously
underwent corneal crosslinking (CXL). The grading of the keratoconus eyes
(based on the Krumeich classification system[16])
was as follows: 23% were grade I, 56% were grade II, 10% were grade III, and
11% were grade IV. The mean value for thinnest corneal pachymetry was 451±57
µm. None of the patients had a history of glaucoma or ocular hypertension.
Applanation
vs Diaton Intraocular Pressure
Measurements In the
keratoconus group, mean IOP measured using AT and DT was 11.0±2.6 mm Hg and 11.2±5.5
mm Hg, respectively
(P=0.729). In the healthy control
group, mean IOP measured using AT and DT was 12.7±2.7 mm Hg and 7.3±2.5
mm Hg, respectively
(P<0.001). Thus, the mean
difference between the AT and DT measurements in the keratoconus and control
groups was -0.2±5.2 mm Hg and 5.5±3.5
mm Hg, respectively
(P<0.001). The IOP measurements of
keratoconus eyes that received CXL did not
differ significantly from their untreated counterparts: AT measurements were
10.8 mm Hg vs 11.5 mm Hg (P=0.295), and DT
measurements 11.9 mm Hg vs 10.2 mm Hg (P=0.194). The mean
difference between AT and DT measurements after CXL changed from 1.3±5.4 mm Hg to -1.1±4.8
mm Hg (P=0.057). Similar results were obtained
regardless of whether the AT or DT measurement was taken first (data not
shown). The AT and DT measurements in the two groups are summarized in Figure
1.
Correlation
Between Diaton Tonometer and Applanation Tonometry Intraocular Pressure Measurements The correlation
between the DT and AT measurements was low but significant in the keratoconus
group (R2=0.104 P=0.006), but not in the healthy control
group (R2=0.017, P=0.316). The measurements and their
correlation coefficients are shown in Figure 2; R2 is given for absolute IOP measurements. Trend lines
are added to highlight the lack of agreement; perfect agreement would result in
a trend with a 45° slope through the origin.
Figure
3 shows a Bland-Altman plot of the AT and DT measurements in the keratoconus
group. Although the mean difference is extremely small (-0.21 mm Hg), a big variation of
measurements is visualized. This variation exists at low mean IOP levels (left
side of the plot) as well as at higher mean IOP levels (right side of the
plot). The SD of the difference between the AT and DT measurements is 5.2 mm Hg, which means that
27% of the DT measurements differed from their corresponding AT measurement by
>1 SD. Only 16% of the measurements are within 2 mm Hg range of agreement.
Effect of Pachymetry and Keratoconus Staging on
Outcomes Linear
regression analysis revealed a small, non-significant effect of pachymetry on
the difference between the AT and DT measurements (B: -0.011; 95% CI: -0.032 to
0.010; χ2: 1.022; P=0.312), which means that a difference
in pachymetry of 100 µm
estimates a lower difference between AT and DT of 1.1 mm Hg. Krumeich
classification had no effect on the difference between the AT and DT
measurements (χ2: 1.331; P=0.722).
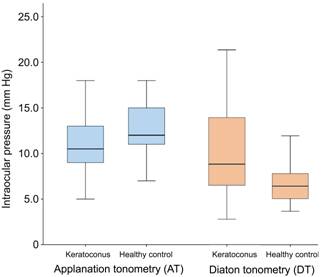
Figure 1
IOP measurements with applanation
tonometry (AT) vs Diaton tonometry
(DT) in keratoconus and healthy controls The
mean IOP was comparable in the AT-group (P=0.729),
and significantly lower for healthy controls in the DT-group (P<0.001).
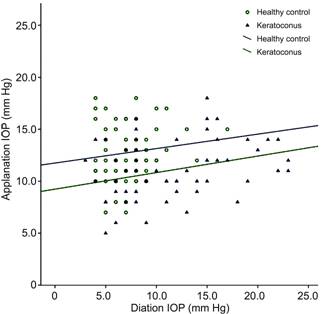
Figure 2
Correlation of applanation tonometry
(AT) vs Diaton tonometry (DT) IOP measurements
for the keratoconus group (R2=0.104
P=0.006) and healthy controls (R2=0.017, P=0.316) Trend
lines are given for both groups.
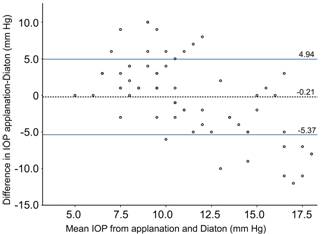
Figure 3
Bland-Altman plot of the agreement of
applanation tonometry (AT) vs Diaton
tonometry (DT) in keratoconus patients (n=70) The dashed line represents the mean
difference (-0.21 mm Hg); The solid lines
represent the ±1 SD
of the mean difference (±5.2 mm Hg). Note the high
spread number of measurements; 16% of measurements are within a 2 mm Hg range of agreement.
DISCUSSION [Top]
In
this study, we investigated the added value of performing transpalpebral
tonometry versus Goldmann AT to measure IOP in keratoconus. The
small mean difference of IOP measurements in keratoconus between both
instruments suggest that DT could be an alternative for AT. However, the wider
variability of DT measurements and their poor correlation to AT renders the use
of the Diaton tonometer in keratoconus debatable.
These
findings are consistent with two large studies in which Diaton tonometry was
used to measure IOP in eyes without corneal disease[13-14].
Both studies reported remarkably poor agreement between DT and AT measurements
and concluded that DT is not a feasible substitute for AT in routine clinical
practice. However, patients generally favor DT over AT, particularly young
patients[13-14].
Nevertheless, Goldmann AT remains the gold standard for measuring
IOP, although other devices have been studied extensively and are considered
suitable alternatives[2,18-20]. The ocular response analyzer in
particular combines IOP measurements with information on central corneal
thickness and corneal hysteresis[20].
It
is important to note that all IOP measurements were within the normal range;
the highest recorded IOP was 23 mm Hg. We cannot draw conclusions for
higher IOP ranges. In our measurements, we did not account for eyelid
abnormalities due to allergic papillary conjunctivitis, which is a potential
confounding factor for transpalpebral tonometry in keratoconus. All patients
were treated for concomitant ocular allergy; however, eyelid eversion was not
performed routinely. Another consideration regarding DT is that the
measurements are rather cumbersome to perform, as the patient must be in a
supine or reclined position. In addition, the Diaton device has a steep
learning curve; however, this was not likely to have affected the outcome, as
the examiner in this study (Peeters N) had extensive experience performing DT prior to the start of
the study. The significant difference between DT measurements in keratoconus
and healthy eyes (with a mean difference of -5.5±3.5 mm Hg) could not be
explained and is not consistent with previous studies[13-15].
A quarter of the DT measurements in healthy eyes were <5 mm Hg, which is not
compatible with the distribution of IOPs in a normal population[21].
The initial patient records and the study database were checked for erroneous
data entries, but these were not found. We can only hypothesize on the origin
of this difference; statistical chance is highly unlikely based on the solid
significance. A calibration deficit might have clouded the measurements, though
the apparatus was calibrated before every measurement according to the
manufacturers instruction. Regardless of the origin of this deficit we state
that these data do not support our hypothesis that DT can potentially identify
false-negative IOP measurements in keratoconus eyes.
The
prevalence of glaucoma increases in eyes following penetrating keratoplasty
(PK), and AT
can be difficult to perform in these cases[22]. Although no post-PK eyes were
included in this study, we recommend using a device that has been shown to be
reliable for measuring IOP in keratoconus and/or post-PK eyes.
The
Diaton device is specifically advertised for use in patients with corneal
disease; however, although the device is portable, well tolerated by patients,
and not influenced by corneal biomechanics, our results suggest that it does
not measure IOP reliably in patients with keratoconus.
ACKNOWLEDGEMENTS [Top]
Conflicts
of Interest: Wisse
RPL, None; Peeters N, None; Imhof SM, None; van der Lelij A, None.
REFERENCES[Top]
1 Rosentreter A, Athanasopoulos A, Schild AM, Lappas A,
Cursiefen C, Dietlein TS. Rebound, applanation, and dynamic contour tonometry
in pathologic corneas. <ii>Cornea</ii> 2013;32(3):313-318. [CrossRef]
[PubMed]
2 Smedowski A, Weglarz B,
Tarnawska D, Kaarniranta K, Wylegala E. Comparison of three intraocular
pressure measurement methods including biomechanical properties of the cornea.
<ii>Invest Ophthalmol Vis Sci</ii> 2014;55(2):666-673. [CrossRef]
[PubMed]
3 Klamann MK, Maier AK,
Gonnermann J, Ruokonen P, Bertelmann E, Torun N. Influence of corneal thickness
in keratoconic corneas on IOP measurement with IOPen, iCare, dynamic contour
tonometry and Goldmann applanation tonometry. <ii>Klin Monbl
Augenheilkd</ii> 2013;230(7):697-700. [PubMed]
4 Edmund C. Corneal topography
and elasticity in normal and keratoconic eyes. A methodological study
concerning the pathogenesis of keratoconus. <ii>Acta Ophthalmol
Suppl</ii> 1989;193:1-36. [PubMed]
5 Morishige N, Wahlert AJ,
Kenney MC, Brown DJ, Kawamoto K, Chikama T, Nishida T, Jester JV.
Second-harmonic imaging microscopy of normal human and keratoconus cornea.
<ii>Invest Ophthalmol Vis Sci</ii> 2007;48(3):1087-1094. [CrossRef]
[PubMed]
[PMC free article]
6 Ruiseñor Vázquez PR, Delrivo
M, Bonthoux FF, Pförtner T, Galletti JG. Combining ocular response analyzer
metrics for corneal biomechanical diagnosis. <ii>J Refract
Surg</ii> 2013;29(9):596-602. [CrossRef]
[PubMed]
7 Johnson RD, Nguyen MT, Lee N,
Hamilton DR. Corneal biomechanical properties in normal, forme fruste
keratoconus, and manifest keratoconus after statistical correction for
potentially confounding factors. <ii>Cornea</ii>
2011;30(5):516-523. [PubMed]
8 Herndon LW, Choudhri SA, Cox
T, Damji KF, Shields MB, Allingham RR. Central corneal thickness in normal,
glaucomatous, and ocular hypertensive eyes. <ii>Arch
Ophthalmol</ii> 1997;115(9):1137-1141. [CrossRef]
9<ii> </ii>Salvetat
ML, Zeppieri M, Tosoni C, Brusini P. Repeatability and accuracy of applanation
resonance tonometry in healthy subjects and patients with glaucoma.
<ii>Acta Ophthalmol</ii> 2014;92(1):e66-73. [CrossRef]
[PubMed]
10 Cohen EJ, Myers JS.
Keratoconus and normal-tension glaucoma: a study of the possible association
with abnormal biomechanical properties as measured by corneal
hysteresis.<ii> Cornea</ii> 2010;29(9):955-970. [CrossRef]
[PubMed]
11 Terai N, Raiskup F, Haustein
M, Pillunat LE, Spoerl E. Identification of biomechanical properties of the
cornea: the ocular response analyzer. <ii>Curr Eye Res </ii>
2012;37(7):553-562. [CrossRef] [PubMed]
12 Nesterov AP, Dzhafarli TB,
Illarionova AR. Use of transpalpebral tonometry in the estimation of
intraocular pressure in patients with refractory anomaly before and after
keratophotorefraction interventions. <ii>Vestn Oftalmol</ii>
2007;123(6):41-43. [PubMed]
13 Doherty MD, Carrim ZI,
O’Neill DP. Diaton tonometry: an assessment of validity and preference against
Goldmann tonometry. <ii>Clin Experiment Ophthalmol</ii> 2012
40(4):e171-175. [CrossRef] [PubMed]
14 Toker MI, Vural A, Erdogan H,
Topalkara A, Arici MK. Central corneal thickness and Diaton transpalpebral
tonometry. <ii>Graefes Arch Clin Exp Ophthalmol </ii>
2008;246(6):881-889. [CrossRef] [PubMed]
15 Schlote T, Landenberger H.
Intraocular pressure difference in Goldmann applanation tonometry versus a
transpalpebral tonometer TGDc-01“PRA” in glaucoma patients. <ii>Klin
Monbl Augenheilkd</ii> 2005;222(2):123-131. [CrossRef]
[PubMed]
16 Krumeich JH, Daniel J, Knülle
A. Live-epikeratophakia for keratoconus. <ii>J Cataract Refract
Surg</ii> 1998;24(4):456-463. [CrossRef]
17 Bland JM, Altman DG.
Statistical methods for assessing agreement between two methods of clinical
measurement. <ii>Lancet</ii> 1986;1(8476):307-310. [CrossRef]
18 Kim KN, Jeoung JW, Park KH,
Yang MK, Kim DM. Comparison of the new rebound tonometer with Goldmann
applanation tonometer in a clinical setting. <ii>Acta
Ophthalmol</ii> 2013;91(5):e392-396. [CrossRef]
[PubMed]
19 Kotecha A, White E,
Schlottmann PG, Garway-Heath DF. Intraocular pressure measurement precision
with the Goldmann applanation, dynamic contour, and ocular response analyzer
tonometers. <ii>Ophthalmology</ii> 2010;117(4):730-737. [CrossRef]
[PubMed]
20 Ouyang PB, Li CY, Zhu XH,
Duan XC. Assessment of intraocular pressure measured by Reichert Ocular
Response Analyzer, Goldmann Applanation Tonometry, and Dynamic Contour
Tonometry in healthy individuals. <ii>Int J Ophthalmol </ii>
2012;5(1):102-107. [PMC free article] [PubMed]
21 Li Y, Shi J, Duan X, Fan F.
Transpalpebral measurement of intraocular pressure using the Diaton tonometer
versus standard Goldmann applanation tonometry. <ii>Graefes Arch Clin Exp
Ophthalmol</ii> 2010;248(12):1765-1770. [CrossRef]
[PubMed]
22 Huber KK, Maier AK, Klamann
MK, Rottler J, Özlügedik S, Rosenbaum K, Gonnermann J, Winterhalter S, Joussen
AM. Glaucoma in penetrating keratoplasty: risk factors, management and outcome.
<ii>Graefes Arch Clin Exp Ophthalmol </ii> 2013;251(1):105-116. [CrossRef]
[PubMed]