・Clinical Research・・Current Issue・ ・Achieve・ ・Search Articles・ ・Online
Submission・ ・About
IJO・
Selective laser trabeculoplasty
in patients with pseudoexfoliative glaucoma vs
primary open angle
glaucoma: a one-year comparative study
Arezoo
Miraftabi1,2, Naveed Nilforushan1, Nariman Nassiri2, Kouros Nouri-Mahdavi2
1Eye Research Center, Hazrat Rasoul Akram Hospital, Iran University of
Medical Sciences, Tehran 1445613131, Iran
2Jules Stein Eye Institute, University of California
Los Angeles, Los Angeles 90095, California, USA
Correspondence to: Arezoo Miraftabi. Eye Research Center, Hazrat Rasoul Akram
Hospital, Niayesh Street Satarkhan Avenue, Tehran 1445613131, Iran. arezoomiraftabi@yahoo.com;
miraftabi@jsei.ucla.edu
Received:
2015-03-11
Accepted: 2015-04-24
Abstract
AIM: To compare the efficacy of
single-session 360-degree selective laser trabeculoplasty (SLT) for reduction
of intraocular pressure (IOP) in patients with pseudoexfoliative glaucoma
(PXFG) and primary open angle glaucoma (POAG).
METHODS: This is a single-center, prospective,
nonrandomized comparative study. Patients older than 18 years of age with
uncontrolled PXFG or POAG eyes requiring additional therapy while on maximally
tolerated IOP-lowering medications were included. The primary outcome measure
changed
in IOP from baseline. Success was defined as IOP reduction ≥20% from baseline
without any additional IOP-lowering medication. All patients were examined at
1d, 1wk,
1, 3, 6,
9, 12mo after SLT.
RESULTS:
Nineteen patients (20 eyes) with PXFG and 27 patients (28 eyes) with
POAG were included in the study. In the visual fields mean deviation was -2.88
(±1.67) in the POAG and -3.1 (±1.69) in the PXFG groups (P=0.3). The mean (±SD) IOP was 22.9
(±3.7) mm Hg in the POAG group and 25.7 (±4.4) mm Hg in the PXFG group at
baseline and decreased to 18.4 (±3.2) and 18.0 (±3.9) mm Hg in the POAG group (P<0.001 and P=0.02), and to 17.9 (±4.0) and 21.0
(±6.6) mm Hg in the PXFG group (P<0.001 and P=0.47) at 6 and 12mo,
respectively. The number of medications was 2.6 (±0.8) in the POAG group and 2.5 (±0.8) in the
PXFG group at baseline, and did
not change at all follow-up visits in both groups (P=0.16 in POAG and 0.57 in PXFG). Based on Kaplan-Meier survival analysis, the success rate
was 75% in the POAG group compared to 94.1% in the PXFG group (P=0.08; log rank test) at 6mo, and 29.1% and 25.0% at 12mo, respectively (P=0.9; log rank).
CONCLUSION: The 360-degree SLT is an effective and
well-tolerated therapeutic modality in patients with POAG and PXFG by reducing
IOP without any change in number of medications. The response was more
pronounced early in the postoperative period in patients with PXFG whereas
there was no statistically significant difference at 12-month follow-up.
KEYWORDS: primary open angle glaucoma; pseudoexfoliative glaucoma; selective
laser trabeculoplasty
DOI:10.18240/ijo.2016.03.14
Citation: Miraftabi A, Nilforushan N, Nassiri N, Nouri-Mahdavi
K. Selective
laser trabeculoplasty in patients with pseudoexfoliative glaucoma vs primary open angle glaucoma: a
one-year comparative study. Int J Ophthalmol 2016;9(3):406-410
INTRODUCTION
Selective
laser trabeculoplasty (SLT) was introduced by Latina and Park in 1995 as an
option for the treatment of open angle glaucoma[1].
In this method, a Q-switched, frequency doubled laser with a wavelength of 532
nm and pulse duration of 3ns affects only the pigmented trabecular meshwork
cells while the non-pigmented meshwork cells remain intact[2-3].
SLT has been widely adopted for treatment
of glaucoma and is used by some clinicians early in the course of treatment of
the disease[4-5]. Results
of different studies have shown that SLT reduces the intraocular pressure (IOP)
between 11% to 40% in various types of glaucoma in short to intermediate-term
period[6]. The exact
mechanism remains uncertain by which SLT reduces IOP but in particular, cytokine secretion,
matrix metalloproteinase induction, increased cell division, repopulation of
burn sites and macrophage recruitment are responsible for IOP reduction[7].
Although the
efficacy of SLT in patients with primary open angle glaucoma (POAG) has been well
documented, there are few studies that have investigated its efficacy in other
types of glaucoma including pigmentary and pseudoexfoliative glaucoma (PXFG)[8-9]. In this
prospective study, we compared the efficacy of 360-degree SLT treatment in
patients with PXFG to those with POAG.
SUBJECTS
AND METHODS
Study Subjects This single-center, prospective, nonrandomized
comparative study was performed between March 2010 and March 2013 at the Hazrat
Rasoul Akram Hospital, Tehran,
Iran. The Ethics
Committee at Iran University of Medical Sciences, Tehran, Iran, approved the
study protocol. Written informed
consents were obtained from all patients and the study was carried out in
accordance with the principles of Declaration of Helsinki.
Patients aged
18 years or older with uncontrolled PXFG or POAG requiring additional therapy
while on maximally tolerated IOP-lowering medications were enrolled. Exclusion
criteria were eyes with prior history of laser or incisional surgery such as
phacoemulsification, glaucoma procedures or argon laser trabeculoplasty; ocular
trauma or any other preexisting corneal disease precluding the angle evaluation;
or if the trabecular meshwork could not be viewed for 360 degrees. Preoperative
exams included slit-lamp biomicroscopy, IOP measurement using Goldmann applanation
tonometry, gonioscopy, dilated fundoscopic examination with a 90 D lens, and
standard automated perimetry with 24-2 Swedish interactive threshold Algorithm
(SITA; Carl Zeiss Meditec Inc. Dublin, CA, USA).
Outcome
Measures The primary outcome measure was IOP (mm Hg) before
and after 360-degree SLT in both groups at 1d, 1wk and then every 3mo after
surgery. All of IOP measurements performed at morning in sitting position.
Other outcome measures included success
rate, changes in the number of IOP-lowering medications, and complications.
Success was defined as IOP reduction ≥20% from the baseline without additional
IOP-lowering medications. Failure was defined as IOP reduction <20% from the
baseline at two consecutive visits and/or addition of IOP-lowering medications.
Laser Procedure All the study eyes underwent 360-degree SLT for the
first time. All the procedures were performed by two surgeons (Miraftabi A or
Nilforushan N) with the same protocol, which included Q-switched
frequency-doubled 532 nm Nd:YAG laser (Ellex Tango SLT/Photodistruptor
Combination Laser, Ellex Medical Lasers Ltd., Adelaide, Australia) with pulse
duration of 3 ns and a spot size of 400 μm for 360 degrees. No pretreatment with IOP-lowering
medications was used. After topical anesthesia with 0.5% tetracaine
hydrochloride, a laser goniolens (Ocular Latina SLT Gonio Laser Lens, Ocular
Instruments, USA) was placed on the eye and a series of 70-90 non-overlapping
shots was evenly carried out for 360 degrees in mid-trabecular meshwork. The
laser energy was initially selected based on the intensity of angle pigmentation:
0.4 mJ for eyes with densely pigmented trabecular meshwork, 0.6 mJ for eyes
with moderate pigmentation, and 0.8 mJ for lower grades of trabecular meshwork
pigmentation, and increased by 0.1-0.2 mJ until small “champagne bubbles” were observed and then decreased by
0.1 mJ. All patients were given 0.5% bethametasone drops 4 times daily for 7d
after the procedure, and instructed to continue routine IOP-lowering
medications. The medications were modified at follow-up visits according to IOP
measurements as needed.
Statistical Analysis The sample size was calculated based on
a difference of at least 1.3 mm Hg in IOP between two groups, standard
deviation of 3 mm Hg and a confidence interval of 95%. Kolmogorov-Smirnov
test and Q-Q plots were used to evaluate the normality of IOP measurements. To
compare the two study groups, we used t-test,
Mann-Whitney test, Chi-square and Fisher exact
test, whenever appropriate. Log-rank test was used to compare survival curves.
Univariate and multivariate Cox’s regression analyses were used to assess
factors associated with time to failure. All statistical analyses were
performed with SPSS (version 21.0, Chicago, IL, USA).
P-values less than 0.05 were
considered statistically significant.
RESULTS
Forty-eight
eyes of 46 patients (27 females and 19 males) who met the eligibility criteria
were consecutively enrolled in the study. There were 27 patients with POAG and 19 patients
with PXFG. In each group, one patient had both eyes included in the study. The baseline characteristics of the
study subjects are described in Table 1. There were no significant differences
between the two study groups in baseline characteristics except for gender
(with a higher ratio of female in the POAG group), and IOP, which was higher in
the PXFG group (P=0.025). In the
visual fields the average mean deviation (MD) was -2.88 (±1.67) in the POAG and
-3.1 (±1.69) in the PXFG groups (P=0.3).
The number of patients at each
follow-up visit, and changes in IOP and number of medications from baseline in
each study group are described in Tables 2 and 3.
Table 1
The baseline characteristics of the
study patients
![]() , n (%)
, n (%)
|
Variables |
Total |
POAG |
PXFG |
P |
|
No. of eyes |
48 |
28 |
20 |
|
|
No. of patients |
46 |
27 |
19 |
|
|
Age (a) |
64.0±9.0 |
62.2±8.9 |
68.6±7.5 |
0.2 |
|
Sex |
|
|
|
|
|
Male |
19 (41.7) |
6 |
13 |
0.01 |
|
Female |
27 (58.3) |
21 |
6 |
0.01 |
|
Laterality |
|
|
|
|
|
Right eye |
20 (41.7) |
13 (46.4) |
7 (35.0) |
|
|
Left eye |
28 (58.3) |
15 (53.6) |
13 (65.0) |
|
|
Diabetes mellitus |
|
|
|
|
|
No |
35 (72.9) |
20 (71.4) |
15 (75) |
|
|
Yes |
13 (27.1) |
8 (28.6) |
5 (25) |
|
|
Central corneal thickness |
537±31 |
531.6±31.84 |
537.6 ±29.9 |
0.5 |
|
Cup to disc ratio |
0.6±0.2 |
0.570±0.14 |
0.591±0.17 |
0.78 |
|
Baseline IOP (mm Hg) |
24±4.2 |
22.9±3.7 |
25.7±4.4 |
0.025 |
|
Baseline number of medications |
3.0±0.8 |
2.6±0.8 |
2.5±0.8 |
0.84 |
|
Baseline MD (dB) |
-3.0 ±1.67 |
-2.88±1.67 |
-3.1±1.69 |
0.3 |
POAG:
Primary open angle glaucoma; PXFG: Pseudoexfoliative glaucoma.
Table 2
IOP measurements in the study groups at different
follow-up visits ![]()
|
Time |
POAG |
PXFG |
1P |
|
Baseline |
|
|
|
|
No. of eyes |
28 |
20 |
|
|
Value |
22.9±3.7 |
25.7±4.4 |
0.02 |
|
1d |
|
|
|
|
No. of eyes |
28 |
20 |
|
|
Value |
17.8±5.1 |
14.7±4.3 |
0.03 |
|
Change |
-5.2±5.0 |
-11±5.3 |
<0.001 |
|
Change % |
-22±20 |
-42±18 |
<0.001 |
|
1wk |
|
|
|
|
No. of eyes |
23 |
20 |
|
|
Value |
19.8±3.6 |
18.7±4.2 |
0.33 |
|
Change |
-3.2±3.2 |
-7.1±6.2 |
0.01 |
|
Change % |
-13±13 |
-26±21 |
0.02 |
|
1mo |
|
|
|
|
No. of eyes |
27 |
20 |
|
|
Value |
18.4±4.9 |
17.7±4.3 |
0.61 |
|
Change |
-4.5±4.0 |
-7.9±5.6 |
0.02 |
|
Change % |
-20±18 |
-30±19 |
0.04 |
|
3mo |
|
|
|
|
No. of eyes |
28 |
20 |
|
|
Value |
18.1±2.7 |
17.0±3.4 |
0.22 |
|
Change |
-4.8±3.0 |
-8.7±5.8 |
0.005 |
|
Change % |
-20±11 |
-32±19 |
0.009 |
|
6mo |
|
|
|
|
No. of eyes |
28 |
20 |
|
|
Value |
18.4±3.2 |
17.9±4.0 |
0.62 |
|
Change |
-4.6±3.0 |
-7.8±5.5 |
0.01 |
|
Change % |
-19±11 |
-29±17 |
0.02 |
|
9mo |
|
|
|
|
No. of eyes |
23 |
17 |
|
|
Value |
17.9±3.3 |
16.3±1.0 |
0.96 |
|
Change |
-4.9±3.5 |
-6.7±2.9 |
0.30 |
|
Change % |
-21±13 |
-28±11 |
0.22 |
|
12mo |
|
|
|
|
No. of eyes |
18 |
15 |
|
|
Value |
18.0±3.9 |
21.0±6.6 |
0.32 |
|
Change |
-3.5±4.2 |
-4.7±9.3 |
0.75 |
|
Change % |
-16±17 |
-16±32 |
0.9 |
POAG: Primary open angle glaucoma; PXFG: Pseudoexfoliative glaucoma.
1t-test.
Table 3 Number of IOP-lowering medications in
the study groups at different follow-up visits ![]()
|
Time |
POAG |
PXFG |
1P |
|
Baseline |
2.6±0.8 |
2.5±0.8 |
0.84 |
|
1d |
2.5±0.8 |
2.3±0.9 |
0.49 |
|
1wk |
3.0±3.0 |
2.3±0.7 |
0.41 |
|
1mo |
3.0±2.3 |
2.3±0.8 |
0.10 |
|
3mo |
2.4±0.8 |
2.3±0.7 |
0.72 |
|
6mo |
2.4±0.7T |
2.4±0.8 |
1.00 |
|
9mo |
2.6±0.9 |
2.5±1.0 |
0.75 |
|
12mo |
2.4±1.1 |
2.7±0.6 |
0.74 |
POAG:
Primary open angle glaucoma; PXFG: Pseudoexfoliative glaucoma.
1Mann-Whitney test.
The mean±SD IOP at baseline and the last visit was 22.9±3.7 and 18.0±3.9 mm Hg in
the POAG group (P=0.02), and 25.7±4.4 and 21.0±6.6 mm Hg in the PXFG group (P=0.47), respectively. Mean number of IOP-lowering
medications at baseline was 2.6±0.8 in the POAG group and 2.5±0.8 in the PXFG
group, which decreased to 2.4±1.1 and increased to 2.7±0.6 at the last
follow-up visit, respectively. The differences were not statistically
significant in both groups (P=0.16,
0.57). On Kaplan-Meier survival analysis, the success
rate was 75.0% in the POAG group compared to 94.1% in the PXFG group (P=0.08; Log-rank test) at 6mo, and 29.1% in
the POAG group and 25.0% in the PXFG group at 12mo (P=0.9; Log-rank test; Figure 1). The median
time to failure was 6mo in both groups. The percentage of IOP reduction was
statistically significantly (P=0.02) in the PXFG group during the
first 6mo of follow-up (Table 2) that might be related to higher baseline IOP in
PXFG group (Figure
2).
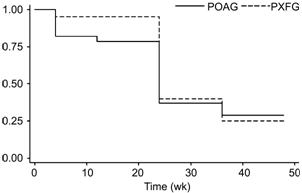
Figure 1 Kaplan-Meier survival curve analysis of event-free
rates between the POAG and PXFG groups.
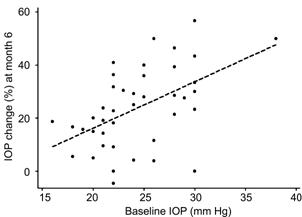
Figure 2 Graph showing the relationship between baseline
IOP and changes of IOP at month 6 from baseline.
On univariate analyses, there was no statistically
significant association between time to failure and the following factors: age,
gender, glaucoma type, laterality, diabetes, central corneal thickness, cup to
disc ratio, preoperative IOP and number of medications, and the percentage of
IOP reduction at postoperative week 1. The only observed complication was redness without
intraocular inflammation in one eye in each group one day after SLT, which
resolved spontaneously after 3d.
DISCUSSION
In
this prospective comparative study, we investigated intermediate-term outcomes
of 360-degree SLT in patients with mild to moderate POAG and PXFG with
uncontrolled IOP despite maximally tolerated IOP-lowering medications. Although
the absolute post-laser IOP measurements were similar in both study groups at
all follow-up visits (except 1d), the percent IOP reduction was statistically
significantly more pronounced in the PXFG group during the first 6mo of
follow-up; this might be attributed to higher baseline IOP in the PXFG group.
The mean number of IOP-lowering medications was statistically similar in both
groups at baseline and SLT did not significantly reduce the number of
medications at any of the follow-up visits in both groups. In a retrospective
study, Kara et al [10] similarly reported that the PXFG patients had
significantly higher percentage of IOP reduction compared to POAG patients over
one year of follow-up[10].
They also found that SLT did not reduce the number of medications at any
follow-up visits in both POAG and PXFG groups[10]. In another prospective study, Ayala and Chen[11] found that SLT reduced
IOP by about 6-7 mm Hg in both POAG and PXFG patients one month after the
procedure. Although the reduction was slightly better in their PXFG patients,
the difference was not statistically significant[11]. In our study, the efficacy of SLT
decreased over time in both groups, and this was more pronounced in the PXFG
group. Our results are similar to Gracner findings in PXFG patients[12]. This
study showed that SLT is an effective procedure for lowering IOP in PXFG and
POAG eyes, although the effect seems to last less in PXFG eyes. In another study, Kara et al [10] showed that SLT was more effective in reducing IOP in
PXFG compared to POAG, and the effect did not decrease over one year of
follow-up[10]. They found that IOP
significantly decreased by 4.4±2.1 mm Hg in the POAG group and by 6.1±3.6 mm Hg
in the PXFG group at the 12-month follow up[10].
A recent Meta-analysis showed that IOP
reduction at 12mo post-SLT in 35 studies ranged from 6.9% to 35.9%[13].
Although few studies have
included other types of glaucoma than POAG, the efficacy of SLT appears to vary
according to type of glaucoma[8,14].
Overall, it seems that the IOP-lowering effect of SLT is less in patients with
normal tension glaucoma (range: 14% to 16%), and relatively greater in PXFG
patients (range: 11% to 32%)[13,15-16]
. The result of this Meta-analysis also showed that
medications reduced IOP more than SLT (0.9 mm Hg more with multi therapy and
0.6 mm Hg more with mono-therapy); however, the difference was not
statistically significant[13].
The effect of SLT on reducing the number of medications varies in different
studies. While some studies have shown that the number of medications did not
change after SLT, others have reported that SLT reduced the number for
medications[13,15,17-18]. In
our study, the mean
number of IOP-lowering medications was statistically similar in both groups at
baseline and SLT did not significantly reduce the number of medications at any
of the follow-up visits in both groups. Lee et
al[19] studied different predictors of success/failure of
SLT in 111 eyes (65 patients) with normal tension glaucoma and POAG[19]. They reported that higher baseline IOP, use of
carbonic anhydrase inhibitors, thinner retinal nerve fiber layer, and lower day
1 IOP were predictors of success[19].
Their definition for success was IOP reduction ≥20%. Martow et al[20] also found
that the only predictive factor of SLT success was the pretreatment IOP and
that glaucoma type including PXF was not a predictor[20].
Tzimis et al [21] showed that
only baseline IOP was the most powerful variable in predicting outcome of SLT[21]. In contrast, no
baseline factors were found to be a predictor for failure after SLT in our
study. This may be a factor of the fairly small sample size of our study since
the sample size was determined for a different outcome measure.
The most common side effects of SLT are IOP spike,
anterior chamber inflammation, eye pain or discomfort, photophobia, and
conjunctival hyperemia[13]. These
side effects are generally transient and minor. The reported incidence of IOP spike varied from 0
to 62% in different studies[13]. In studies where prophylactic IOP-lowering medications were used, the reported
incidence was lower (between 0 and 29%)[13]. In our study, we did not
observe any significant complications during and after the procedure although
we did not use any prophylactic medication.
This study has some shortcomings. The fairly small
number of patients in our study limited the power of our study for finding
predictors of failure after SLT as mentioned above. Furthermore, the
degree of angle pigmentation was not recorded in our study and therefore, the
potential role of this variable could not be explored.
In conclusion, we found that 360-degree SLT is an
effective and well-tolerated therapeutic modality in patients with POAG and
PXFG. We found that SLT was more effective early in the postoperative period in
patients with PXFG compared with patients with POAG. However, the efficacy of
SLT decreased over time and this was more pronounced in PXFG patients.
ACKNOWLEDGEMENTS
Conflicts of Interest:
Miraftabi A, None; Nilforushan N, None;
Nassiri N, None;
Nouri-Mahdavi K, None.
REFERENCES
1 Park CH, Latina MA, Schuman JS. Developments in laser
trabeculoplasty. <ii>Ophthalmic Surg Lasers
</ii>2000;31(4):315-322. [PubMed]
2
Kramer TR, Noecker RJ. Comparison of the morphologic changes after selective
laser trabeculoplasty and argon laser trabeculoplasty in human eye bank eyes.
<ii>Ophthalmology</ii> 2001;108(4):773-779. [CrossRef]
3
Cvenkel B, Hvala A, Drnovsek-Olup B, Gale N. Acute ultrastructural changes of
the trabecular meshwork after selective laser trabeculoplasty and low power
argon laser trabeculoplasty. <ii>Lasers Surg Med </ii>
2003;33(3):204-208. [CrossRef]
[PubMed]
4
Melamed S, Ben Simon GJ, Levkovitch-Verbin H. Selective laser trabeculoplasty
as primary treatment for open-angle glaucoma: a prospective, nonrandomized
pilot study. <ii>Arch Ophthalmol</ii> 2003;121(7):957-960. [CrossRef] [PubMed]
5
Samples JR, Singh K, Lin SC, Francis BA, Hodapp E, Jampel HD, Smith SD. Laser trabeculoplasty
for open-angle glaucoma: a report by the american academy of ophthalmology.
<ii>Ophthalmology</ii> 2011;118(11):2296-2302. [CrossRef] [PubMed]
6
Stein JD, Challa P. Mechanisms of action and efficacy of argon laser
trabeculoplasty and selective laser trabeculoplasty. <ii>Curr Opin
Ophthalmol </ii> 2007;18(2):140-145. [CrossRef] [PubMed]
7
Kagan DB, Gorfinkel NS, Hutnik CM. Mechanisms of selective laser
trabeculoplasty: a review. <ii>Clin</ii> <ii>Experiment
Ophthalmol</ii> 2014;42(7):675-681. [CrossRef] [PubMed]
8
Ayala M. Long-term outcomes of selective laser trabeculoplasty (SLT) treatment
in pigmentary glaucoma patients. <ii>J Glaucoma</ii>
2014;23(9):616-619. [CrossRef]
[PubMed]
9
Shazly TA, Smith J, Latina MA. Long-term safety and efficacy of selective laser
trabeculoplasty as primary therapy for the treatment of pseudoexfoliation
glaucoma compared with primary open-angle glaucoma. <ii>Clin Ophthalmol
</ii>2011;5:5-10.
10
Kara N, Altan C, Yuksel K, Tetikoglu M. Comparison of the efficacy and safety
of selective laser trabeculoplasty in cases with primary open-angle glaucoma
and pseudoexfoliative glaucoma. <ii>Kaohsiung J Med Sci</ii>
2013;29(9):500-504. [CrossRef]
[PubMed]
11
Ayala M, Chen E. Comparison of selective laser trabeculoplasty (SLT) in primary
open angle glaucoma and pseudoexfoliation glaucoma. <ii>Clin
Ophthalmol</ii> 2011;5:1469-1473. [CrossRef] [PubMed] [PMC free article]
12
Grancner T. Intraocular pressure response of capsular glaucoma and primary
open-angle glaucoma to selective Nd:YAG laser trabeculoplasty: a prospective,
comparative clinical trial. <ii>Eur J Ophthalmol</ii>
2002;12(4):287-292.
13
Wong MO, Lee JW, Choy BN, Chan JC, Lai JS. Systematic review and meta-analysis
on the efficacy of selective laser trabeculoplasty in open-angle glaucoma.
<ii>Surv Ophthalmol</ii> 2015;60(1):36-50. [CrossRef] [PubMed]
14
Tokuda N, Inoue J, Yamazaki I, Matsuzawa A, Munemasa Y, Kitaoka Y, Takagi H,
Ueno S. Effects of selective laser trabeculoplasty treatment in steroid-induced
glaucoma. <ii>Nippon Ganka Gakkai Zasshi</ii> 2012;116(8):751-757.
[PubMed]
15
Lee JW, Ho WL, Chan JC, Lai JS. Efficacy of selective laser trabeculoplsty for
normal tension glaucoma: 1 year results. <ii>BMC Ophthalmol </ii>
2015;15:1 [CrossRef] [PubMed] [PMC free article]
16
Goldenfeld M, Geyer O, Segev E, Kaplan-Messas A, Melamed S. Selective laser
trabeculoplasty in uncontrolled pseudoexfoliation glaucoma.
<ii>Ophthalmic Surg Lasers Imaging</ii> 2011;42(5):390-393. [CrossRef] [PubMed]
17
Katz LJ, Steinmann WC, Kabir A, Molineaux J, Wizov SS, Marcellino G, SLT/Med
Study Group. Selective laser trabeculoplasty versus medical therapy as initial
treatment of glaucoma: a prospective, randomized trial. <ii>J
Glaucoma</ii> 2012;21(7):460-468. [CrossRef] [PubMed]
18
Abdelrahman AM, Eltanamly RM. Selective laser trabeculoplasty in egyptian
patients with primary open-angle glaucoma. <ii>Middle East Afr J
Ophthalmol</ii> 2012;19(3):299-303. [CrossRef] [PubMed] [PMC free article]
19
Lee JW, Liu CC, Chan JC, Lai JS. Predictors of success in selective laser
trabeculoplasty for Chinese open-angle glaucoma. <ii>J Glaucoma
</ii> 2014;23(5):321-325. [CrossRef] [PubMed]
20
Martow E, Hutnik CM, Mao A. SLT and adjunctive medical therapy: a prediction
rule analysis. <ii>J Glaucoma</ii> 2011;20(4):266-270. [CrossRef] [PubMed]
21
Tzimis V, Tze L, Ganesh J, Muhsen S, Kiss A, Kranemann C, Birth CM. Laser
trabeculoplasty: an investigation into factors that might influence outcomes.
<ii>Can J Ophthalmol</ii> 2011;46(4):305-309. [CrossRef] [PubMed]