[26]. After dissolving the purple reaction product in DMSO, the
optical density of each well was measured using an automatic plate reader at
490 nm wavelength (Bio-tek, USA). The rate of cell inhibition was calculated
using the following equation to establish the concentration of olmesartan to
use for further experiments.
Inhibition
rate = (ODC−ODO)/ ODCˇÁ100%
(ODC: OD of the blank control; ODO: OD of olmesartan
treated)[27].
Real-time Polymerase Chain Reaction A concentration of olmesartan of 0.75 ¦Ěmol/mL (inhibition
rate of 11% in MTT) was selected for PCR, to choose an inhibition rate approximate 10% has reduced the
cell numbers related influence to the results. Six flasks of cells newly seeded
at the same density were divided into 2 groups, 3 as the olmesartan-treated
group, and the rest as control. After incubating the cells with dulbecco's modified
eagle medium (DMEM) without FBS for 24h to synchronize their growth, the medium
was replaced with fresh complete medium (control) or complete medium with 0.75 ¦Ěmol/mL
of olmesartan as the experimental group. After an incubation for 48h, total RNA
was isolated from the cells using RNAiso (Takara Bio, Kyoto, Japan), and its
quality and concentration was measured using Thermo Scientific NanoDrop. One microgram of total RNA
from each samples was used for reverse transcription using a real time (RT) reagent kit with a gDNA Eraser,
followed by amplification using the Applied Biosystems step one detection
system (Applied Biosystems STEP ONE, NY, USA) and SYBR Remix Ex Taq
(Takara Bio, Kyoto, Japan). A ROX reference dye
was applied to correct the fluorescence signal. The thermal cycle protocol
consisted of: 30s at 95ˇć for 1 cycle, 5s at 95ˇć and 30s at 60ˇć with for 40 cycles, 15s at 95ˇć, 1min at 60ˇć, 15s at 95ˇć. The sequences of the PCR primers used were
as follows: ¦Â-actin upper: CCCTGAAGTACCCCATCG, ¦Â-actin lower:
GCTGGGTGTTTGAAGGTC; MMP-2 upper: AGTGGATGATGCCTTTGCTC, MMP-2 lower: GAGTCCGTCCTTACCGTCA; TIMP-1
upper: TCTGGCATCCTGTTGTTG, TIMP-1 lower: GGTCTGGTTGACTTCTGG; TIMP-2
upper: TCTGTGACTTCATCGTGCC, TIMP-2 lower: TGACCCAGTCCATCCAGAG; PCNA
upper: GGCACTCAAGGACCTCAT, PCNA lower: CATACTGGTGAGGTTCACG.
Ct values were used for further analysis, and differences in the total amount of RNA
present in each sample were normalized to ¦Â-actin. The ∆∆Ct method was used to calculate
the levels of gene expression relative to the expression levels of ¦Â-actin.
Rabbit
Conjunctival Flap Model Thirty New Zealand albino rabbits, weighting 2.2-2.5 kg each, of
either sex were housed and fed separately and were randomly divided into 5
groups (3rd, 7th, 14th, 21st, and 28th day group which correspond to the day the
rabbits' eyes would be harvest after the surgery), with 6 animals in each
group. For each rabbit, one eye was used as the control eye while the other eye
defined as the experimental eye. After anesthetized rabbit with phenobarbital
(30 mg/kg, IV), a rabbit conjunctival flap model was created in both eyes
through the incision of the conjunctiva between 10 oˇŻclock and 2 oˇŻclock in the
upper limbus of the cornea with the fornix as the base, as previously described[28]. The scleral tissue under the
conjunctiva flap was exposed thoroughly first and then pressed with a cotton
swab to stop bleeding. Four to six stitches with 8-0 nylon threads were applied
to suture the wound and restore the consistency of the eye. A 500 µL solution of 1% sterilized olmesartan (olmesartan
acid-active metabolite, later referred to as olmesartan) in PBS was injected to subconjunctival (at 12
o' clock to the base of the conjunctiva flap) to the experimental eye, while
only PBS was injected to subconjunctival to the other eye (control) in exactly
the same location by the end of the surgery. Topical antibiotic eye drops were
applied to both eyes and then during the following 3d. The same subconjunctival
injection of olmesartan and PBS was repeated on the 5th postoperative day. On the 7th day postoperatively, the stitches were removed under topical
anesthetic eye drops. On the 3rd, 7th, 14th, 21st
and 28th days after surgery, all 6 rabbits from each group were
firstly observed under slit-lamp for anterior segment of eyes, fundoscopy for
retina , and then euthanized, and the eyes were enucleated and washed with icy
PBS. All of the animal procedures
complied with the ARVO Statement and were approved by our Institutional Review
Board.
Hematoxylin and Eosin, MassonˇŻs Trichrome
Stain and Immunohistochemistry Biopsies of the conjunctiva, subconjunctiva and sclera were
obtained from the surgical site and the adjacent area within 5 mm. Maintain the
integrity of the structure for the cross-section of the tissues from the
outermost surface of conjunctiva to the innermost sclera to guarantee the
density of conjunctiva fixed. Tissues were fixed in a 4% paraformaldehyde
solution in PBS, and sections were routinely stained with hematoxylin and
eosin. MassonˇŻs
trichrome staining was performed
only on the day 7 group. For the immunohistochemistry stains, each 5-¦Ěm thick
section was treated sequentially with PBS, 3% H2O2 in distilled water and a bovine serum albumin (BSA)
solution were and incubated with primary antibodies to either MMP-2 (Santa Cruz
Biotechnology, Santa Cruz, CA, USA, 1:200 dilution) or PCNA ( Santa Cruz
Biotechnology, 1:400 dilution) for 24h in a humidified chamber at 4ˇăC. After
washing with PBS, the sections were incubated with a secondary antibody for
40min, were washed, and were then reacted with diaminobenzidine (DAB) until a
brownish stain developed. The sections were rinsed in distilled water,
dehydrated with ascending
concentrations of alcohol, cleared in xylene and mounted in Permount.
Observation and Quantitation of
Histology-stained Sections All of the sections were
observed by light microscopy and photographed with a digital camera (Nikon
Eclipse, Tokyo, Japan). Pictures were taken at 100ˇÁ, 200ˇÁ and 400ˇÁ magnification. The relative abundance and morphology of
cells (including fibroblasts, inflammatory cells, epithelial cells) were observed
on the HE-stained sections. The presence of new collagen and neovascularization
were examined on MassonˇŻs trichrome stained sections (only the 7th day
group eyes were chosen for this Masson's trichrome stain as mentioned before).
The expression of MMP-2 by immunohistochemistry was analyzed using the
Image-Pro Plus analysis system. For each section, 5 visual fields at 400ˇÁ magnification were selected, and the mean density of
the immunostain signal was recorded. The numbers of PCNA positive cells were calculated
using Image-Pro Plus. The areas analyzed for either
MMP-2 or PCNA expression from different experimental groups were from areas of
comparable sections.
Statistical
Analysis Relative quantification of mRNA
expression rectified by control and ¦Â-actin gene with ∆∆Ct method (n=6). MTT and real-time PCR data were expressed as the meanˇŔSD.
Data from the different groups were compared using a paired t-test or a one way ANOVA. P<0.05
was with significant difference.
The
results of immunofluorescence were expressed as the meanˇŔSEM; the mean density
of MMP-2 analyzed by Image-Pro Plus software.
Positive PCNA expression cell nuclear numbers were analyzed by Image-Pro Plus
software, data expressed as meanˇŔSEM, paired t-test applied, P<0.05 as with
significant difference .
RESULTS
Cell Culture In primary cultures of human TenonˇŻs
fibroblast in vitro 7d after seeding,
cells started to migrate from the tissue and grew vigorously (Figure 1). Two to
three days later, the cells reached 80% confluence.
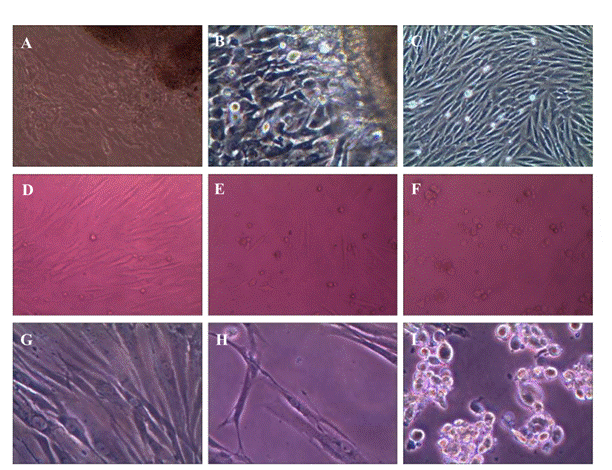
Figure 1 Fibroblast culture
with different olmesartan concentration interference A,B,C: Cultured human
TenonˇŻs fibroblasts growing out of the primary tenon's tissue under 100ˇÁ magnification; D,E,F: Fibroblasts treated with increased
concentration of olmesartan (0.75 µmol/mL,
1.5 µmol/mL, 2 µmol/mL) in MTT with 100ˇÁ
magnification; G,H,I: Fibroblasts in control, 1.25 µmol/mL, 1.5 µmol/mL olmesartan treated
fibroblasts in MTT with 200ˇÁ magnification.
Methyl Thiazol
Tetrazolium Assay of Cell Inhibition and Drug Concentration In the MTT assay (Figure
2), the calculated inhibition rate was as reported previously[28]. Olmesartan (2.0 µmol/mL) generated a
strong suppression of cells; lesser inhibition was observed at lower
concentrations. Olmesartan (0.75 µmol/mL) with an 11% inhibition rate was
chosen for subsequent experiments with cultured fibroblasts.
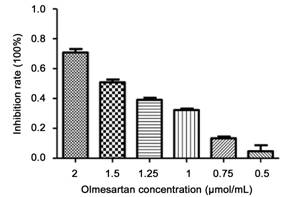
Figure 2 MTT result for
inhibition rate of olmesartan with different concentration from 0.5 to 2 µmol/mL,
increasing inhibition rate of fibroblasts with higher olmesartan
concentration n=6 wells for each concentration.
Real-time Polymerase
Chain Reaction The inhibition effect of olmesartan on
fibroblast cultures in vitro were
analyzed by real-time PCR. The ∆∆Ct method was used to calculate
the relative gene expression for MMP-2, TIMP-1,
TIMP-2, and PCNA (Figure 3). The expression of TIMP-1 and TIMP-2 increased in
the presence of olmesartan, while MMP-2 and PCNA decreased compared with the
control.
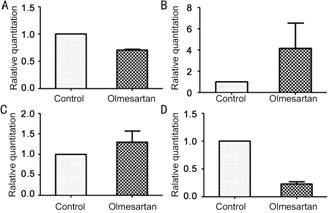
Figure 3 PCR of mRNA
expression in vitro A: MMP-2 mRNA; B: TIMP-2 mRNA; C: TIMP-1mRNA; D: PCNA mRNA.
Observation of
Rabbits' Eyes with Slit-lamp and Fundoscopy Rabbits' eyes were observed under
slit-lamp and fundoscopy before euthanasia. The corneal epithelia was intact
with no spotting lesions, the anterior chambers were clear
without obvious inflammatory reaction, the lenses were intact and translucent, and no abnormalities of the retina were detected by
fundoscopy.
Haematoxylin and Eosin, MassonˇŻs Staining and Immunohistochemistry for Matrix
Metalloproteinase-2 and Proliferating Cell Nuclear Antigen Expression The subconjunctival injection of
olmesartan in the surgical area induced
less fibroblasts proliferation (Figure 4), neovascularization (Figure 5), ECM
deposition (Figure 5) and epithelial proliferation (Figures 4 and 5) in
conjunctiva of
the rabbits eyes at each time group as compared with the control eye.
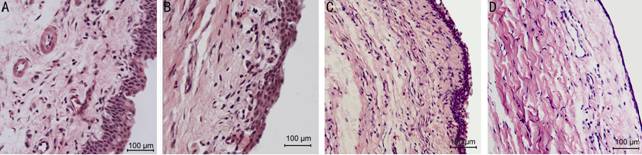
Figure 4 Hemotoxylin and eosin
stain of rabbits conjunctiva, subconjunctiva with 10ˇÁ40
magnification A: 7d group control eye with abundant epithelial
cells proliferation and inflammatory cells migration in subconjunctiva; B: 7d
group olmesartan eye with active proliferation both epithelial cells and
fibroblasts in subconjunctiva; C: 14d group control eye with more fibroblasts;
D: 14d group olmesartan eye.
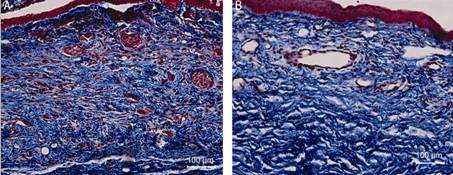
Figure 5 Masson's trichrome
stain of rabbits conjunctiva, subconjunctiva and sclera tissue on the 7d group A: Control eye treated with
PBS only, tight sclera tissue and very crowed neovasculature and fibroblasts in
red; B: Olmesartan treated eye with less crowded neovasculature.
The inhibitive effects of olmesartan on
the conjunctival and subconjunctival tissue proliferation were examined. Via haematoxylin and eosin staining
(Figure 4), more conjunctival epithelial cells
were observed in the control eye compared with the olmesartan-treated eye in
day 3 group; more inflammatory cells migrated to or proliferated in the
subconjunctival tissue of the control group. In the day 7 group, abundant
fibroblast cells were observed in the subconjunctiva tissue in the control eye,
while less fibroblasts were observed in the olmesartan-treated eye. From day 14
to day 21, cells either in the conjunctiva epithelium or subconjunctival tissue
decreased; by day 28, the cells returned to baseline both in the control and
olmesartan-treated eyes.
MassonˇŻs staining in the day 7 group
(Figure 5) showed that neovascularization was
suppressed by olmesartan in the treated eyes compared with the control eyes;
also, collagen fibers and fibroblasts showed reduced density in the olmesartan
group.
The analysis of the expression of MMP-2
by immunohistochemistry with the Image-Pro Plus software showed that the mean
density of MMP-2 decreased with injections of olmesartan in the day 3, 7, 14
and 21 groups; however, no significant difference in the day 28 group was
observed (Figures 6 and 7).
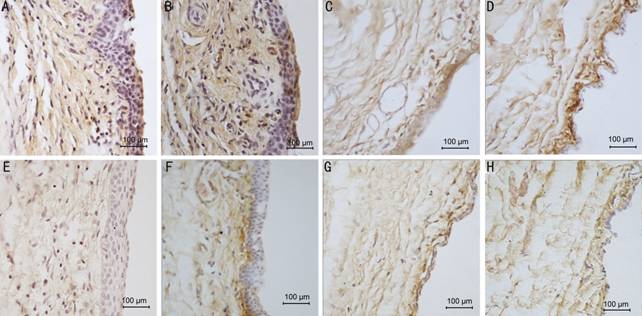
Figure
6 The immunohistochemical
pictures show
the expression of MMP-2 in
conjunctiva and subconjunctival tissue of 10ˇÁ40
magnification A: 3d group control eye, over-proliferative epithelial cells and fibroblasts in
subconjunctiva, brown granules show the MMP-2 expression in all layers of
tissues; B: 7d group control eye, brown staining in all layers; C: 21d
group control eye, darker brown in subconjunctiva; D: 28d
group control eye; E: 3d group
olmesartan eye with milder brown in the same areas as the control; F: 7d
group olmesartan eye, lighter
brown mostly in the subconjunctival area right beneath the epithelial; G: 21d
group olmesartan eye, lighter brown in all layers; H: 28d
group olmesartan eye.
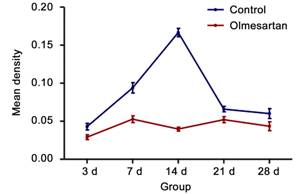
Figure 7 Olmesartan
treated-eye presents with lower MMP-2 expression by means of mean density in
each group comparing with the controlled eye.
Given that PCNA were expressed in the
cell nucleus, the number of positive cells decreased significantly with
olmesartan injections in the day 3, 7, 14 and 21 groups but not in day 28 group
(Figures 8 and 9).

Figure 8
Immunohistochemistry staining of PCNA, 10ˇÁ40
magnification A: 3d group control eye, dark brown granules deposits in
nuclear of cells either in epithelial or subconjunctiva fibroblasts; B: 14d
group control eye, dark brown scattered in different layers cell nuclear; C: 21d
group control eye with dark brown nuclear mainly in epithelial cells; D: 28d
group control eye; E: 3d
group olmesartan treated eye with less brown granules in nuclear; F: 14d
group olmesartan eye with less PCNA positive cell nuclear; G: 21d
group olmesartan eye with milder brown and less positive cells; H: 28d
group olmesartan eye lighter brown granules.

Figure 9 Positive
PCNA expression cell nuclear number Olmesartan-treated eye with lower PCNA
expression in day 3, 7, 14 and 21 group, day 28 group has no significant difference.
DISCUSSION
Studies of MMPs in the cardiovascular
system demonstrated their roles as a pro-proliferation factor and a member of
signaling pathways, inducing cascades of tissue remodeling and contraction[29]. MMP-2 has a pivotal role in the MMP
family through mutual regulation with the other members in the family[30]. In TenonˇŻs fibroblasts, MMP-2 may be
involved, as a pro-proliferation factor, in the molecular signaling to enhance
cell proliferation activity. This is in accordance with the study of Yang et alˇŻs[31], which showed a higher MMP-2 expression
in the fibroblasts of the more active stage of the pterygium. Moreover,
targeting in MMP-2 inhibition can induce tumor
cell apoptosis and reduce scar formation[32-33]. No matter olmesartan suppressed MMP-2
expression through fibroblasts inhibition or the effects on cross-talking of
cell molecules it participates can restrain fibroblast proliferation,
fibroblast proliferation and MMP-2 expression altered in parallel.
Simultaneously, with the increased expression of TIMP-1 and TIMP-2, the
endogenous inhibitors of MMPs, further suppressed the activity of MMPs.
In vivo, olmesartan suppressed the inflammatory
reaction in the rabbit eye surgical model. The number of inflammatory cells
decreased with the subconjunctival injection of olmesartan. These cells include
fibroblasts macrophages and neutrophils. Excessively fibroblast proliferation
and activated macrophage migration can interact with multiple inflammatory
factors, which strengthens the inflammatory effects and induces fibrosis[34]. Therefore, as a key role in
inflammatory reaction, fibroblasts are targeted for inflammation suppression.
Decreasing fibroblasts either in vivo
or in vitro has been demonstrated by
our studies with the administration of olmesartan.
Neovascularization also plays an
important role in inflammatory reaction, neo-vessels generated in the course of
reaction bridge the inflammatory factors cascade. Through in vivo study of morphology on HE staining of Tenon's capsule in
different groups, it is observed that neovascularization changed dynamically.
They appear from 3d postoperatively, after day 7, less neo-vessels could be
observed. Therefore, the 7d group was selected for Masson's trichrome stain for
neovascularization comparison. Reduced angiogenesis in the presence of
olmesartan may account for the suppression of the inflammatory response. As
angiogenesis is closely associated with MMPs and VEGF, in our MassonˇŻs stained
sections, we observed decreased neovascularization corresponding to weaker
expression of MMP-2, as observed by immunohistochemistry, after treatment with
olmesartan, which could be explained by the alteration of MMP-2 and
angiogenesis related cytokines[35-37].
Because endothelial cells of blood
vessels adapt to both physiological and pathological (inflammation and
tumorigenesis) environments continuously, tissue repair processes begin with
the injury-activated acute inflammation reaction, during this process,
cytokines related to angiogenesis are released from the vessels adjacent to the
injury area, which induces vascular sprouting and the production of granulation
tissue that is highly vascularized. Without
appropriate balance mechanisms, newly formed blood vessels do not regress. On
the contrary, a positive feedback loop is formed to enhance further
inflammatory responses. With the participation of MMP-2, membrane-type matrix
metalloproteinase (MT1-MMP), the ECM was degraded, enabling new vascular
sprouts to spread and progress. After GFS, similar processes occurred, as
mediated by VEGF, inducing scar formation[38-39]. Therefore, olmesartan dampened this
course via the inhibition of MMP-2
and correspondingly decreases angiogenesis.
Another proliferative related
factor-PCNA was also examined during the study. Its expression was suppressed
by olmesartan both in vivo and in vitro.
PCNA, a protein present in eukaryotic cells, acts as a DNA clamp, encircles
DNA, and promotes DNA synthesis and repair; meanwhile, PCNA can bind to
CDK/cyclin and modulate the CDK2 and substrate reaction, with a negative effect
on cell apoptosis. Therefore, PCNA also inhibits cell apoptosis. In the studies
of tumor cells and fibroblasts, PCNA was widely used to monitor cell
proliferation, especially the status of the proliferative activity of
fibroblasts[40-41].
Although detailed molecular talk and
cross-linking mechanisms require further study, we present evidence that
olmesartan can effectively inhibit fibroblast proliferation in the conjunctiva
flap model, which approximates the effects of GFS to the conjunctiva and
subconjunctival tissue. Fibroblast proliferation and angiogenesis have been
suppressed with olmesartan, resulting in decreased fibrosis and scarring, which
should improve the results of surgery.
As with the first evaluation of
olmesartan in TenonˇŻs capsule in the
eye, we observed no obvious toxic effect of olmesartan to the rabbit eye; its inhibitory
effect on the fibroblasts and the related scarring has been shown in this study. We hope to offer further options for safer adjuvant
medications for GFS surgery.
ACKNOWLEDGEMENTS
The authors thank the Scientific Research and Laboratory
Center of the Second
Affiliated
Hospital of Xi'an
Jiaotong University for the technical support.
Conflicts of
Interest: Wang X, None; Fan YZ, None; Yao L, None; Wang JM, None.
REFERENCES [Top]
1
Eren K, Turgut B, Akin MM, Demir T. The suppression of wound healing response
with sirolimus and sunitinib following experimental trabeculectomy in a rabbit
model. Curr Eye Res 2015:1-10. [CrossRef] [PubMed]
2
Xue H, McCauley RL, Zhang W , Martin DK. Altered interleukin-6 expression in
fibroblasts from hypertrophic burn scars. J
Burn Care Rehabil 2000;21(2):142-146. [CrossRef] [PubMed]
3
Nakamura-Shibasaki M, Ko JA, Takenaka J, Chikama T, Sonoda KH, Kiuchi Y. Matrix
metalloproteinase and cytokine expression in Tenon fibroblasts during scar
formation after glaucoma filtration or implant surgery in rats. Cell Biochem Funct 2013;31(6):482-488. [CrossRef]
[PubMed]
4
Bourboulia D, Stetler-Stevenson WG. Matrix metalloproteinases (MMPs) and tissue
inhibitors of metalloproteinases (TIMPs): positive and negative regulators in
tumor cell adhesion. Semin Cancer Biol 2010;20(3):161-168. [CrossRef] [PubMed]
[PMC free article]
5
Fujiwara M, Muragaki Y, Ooshima A. Keloid-derived fibroblasts show increased
secretion of factors involved in collagen turnover and depend on matrix
metalloproteinase for migration. Br J
Dermatol 2005;153(2):295-300. [CrossRef] [PubMed]
6
Lanigan L, Stuirmer J, Baez KA, Hitchings RA, Khaw PT. Single intraoperative
applications of 5-fluorouracil during filtration surgery: early results. Br J Ophthalmol 1994;78(1):33-37. [CrossRef]
7
Casson R, Rahman R, Salmon JF. Long term results and complications of
trabeculectomy augmented with low dose mitomycin C in patients at risk for
filtration failure. Br J Ophthalmol 2001;85(6):686-688. [CrossRef]
8
Bindlish R, Condon GP, Schlosser JD, D'Antonio J, Lauer KB, Lehrer R. Efficacy
and safety of mitomycin-C in primary trabeculectomy: five-year follow-up. Ophthalmology 2002;109(7):1336-1341. [CrossRef]
9
Ball KJ, Williams PA, Stumpe KO. Relative efficacy of an angiotensin II
antagonist compared with other antihypertensive agents. Olmesartan medoxomil
versus antihypertensives. J Hypertens
Suppl 2001;19(1):S49-56. [CrossRef] [PubMed]
10
Anand N, Arora S, Clowes M. Mitomycin C augmented glaucoma surgery: evolution
of filtering bleb avascularity, transconjunctival oozing, and leaks. Br J Ophthalmol 2006;90(2):175-180. [CrossRef]
[PubMed] [PMC free article]
11
De Giusti VC, Garciarena CD, Aiello EA. Role of reactive oxygen species (ROS)
in angiotensin II-induced stimulation of the cardiac Na+/HCO3- cotransport. J Mol Cell Cardiol 2009;47(5):716-722. [CrossRef] [PubMed]
12
Pacurari M, Kafoury R, Tchounwou PB, Ndebele K. The
Renin-Angiotensin-Aldosterone System in vascular inflammation and remodeling. Int J Inflam 2014;2014:689360. [CrossRef]
[PubMed] [PMC free article]
13
Harada K, Sugaya T, Murakami K, Yazaki Y, Komuro I. Angiotensin II type 1A
receptor knockout mice display less left ventricu- lar remodeling and improved
survival after myocardial infarction. Circulation
1999;100(20):2093-2099. [CrossRef]
14
Kim S, Ohta K, Hamaguchi A, et al.
Contribution of renal angiotensin II type I receptor to gene expressions in
hypertension-induced renal injury.Kidney
Int 1994, 46(5):1346-1358. [CrossRef] [PubMed]
15
Yuko W,Waseda Y, Yasui M, Nishizawa Y, Inuzuka K, Takato H, Ichikawa Y, Tagami
A, Fujimura M, Nakao S. Angiotensin II type 2 receptor antagonist reduces
bleomycin-induced pulmonary fibrosis in mice. Respir Res 2008;9:43. [CrossRef]
[PubMed] [PMC free article]
16
Escobar E, Rodr¨Şguez-Reyna TS, Arrieta O, Sotelo J. Angiotensin II, cell
proliferation and angiogenesis regulator: biologic and therapeutic implications
in cancer. Curr Vas Pharmacol 2004;2(4):385-399. [CrossRef]
17
Li L, Fan D,Wang C, Wang JY, Cui XB, Wu D, Zhou Y, Wu LL. Angiotensin II
increases periostin expression via Ras/p38 MAPK/CREB and ERK1/2/TGF-¦Â1 pathways
in cardiac fibroblasts. Cardiovasc ReS 2011;91(1):80-89. [CrossRef]
[PubMed]
18
Arenas IA, Xu Y, Lopez-Jaramillo P, Davidge ST. Angiotensin II-induced MMP-2
release from endothelial cells is mediated by TNF-¦Á. Am J Physiol 2004;286(4):C779-784. [CrossRef] [PubMed]
19
Hirose A, Ono M, Saibara T, Nozaki Y, Masuda K, Yoshioka A, Takahashi M,
Akisawa N, Iwasaki S, Oben JA, Onishi S. Angiotensin II type 1 receptor blocker
inhibits fibrosis in rat nonalcoholic steatohepatitis. Hepatology 2007;45(6):1375-1381. [CrossRef]
[PubMed]
20
Shaaban AA, Shaker ME, Zalata KR, El-kashef HA, Ibrahim TM. Modulation of
carbon tetrachloride-induced hepatic oxidative stress, injury and fibrosis by
olmesartan and omega-3. Chem Biol
Interact 2014;207:81-91. [CrossRef] [PubMed]
21
Kanamori H, Takemura G, Li Y, Okada H, Maruyama R, Aoyama T, Miyata S, Esaki M,
Ogino A, Nakagawa M, Ushikoshi H, Kawasaki M, Minatoguchi S, Fujiwara H.
Inhibition of Fas-associated apoptosis in granulation tissue cells accompanies
attenuation of postinfarction left ventricular remodeling by olmesartan. Am J Physiol Heart Circ Physiol 2007;292(5):H2184-2194. [CrossRef] [PubMed]
22
Danser AH, Derkx FH, Admiraal PJ, Deinum J, de Jong PT, Schalekamp MA.
Angiotensin levels in the eye. Invest
Ophthalmol Vis Sci 1994;35(3):1008-1018. [PubMed]
23
Vaajanen A, Vapaatalo H. Local ocular renin-angiotensin system-a target for
glaucoma therapy? Basic Clin Pharmacol
Toxicol 2011;109(4),217-224. [CrossRef] [PubMed]
24
Chen J, Runyan SA, Robinson MR. Novel ocular antihypertensive compounds in
clinical trials. Clin Ophthalmol
2011;5:667-677. [CrossRef] [PubMed]
[PMC free article]
25
Seet LF, Su R, Toh LZ, Wong TT. In vitro analyses of the anti-fibrotic effect
of SPARC silencing in human TenonˇŻs fibroblasts: comparisons with mitomycin C. J Cell Mol Med 2012;16(6):1245-1259. [CrossRef] [PubMed]
[PMC free article]
26
Cetin Y, Bullerman LB. Cytotoxicity of Fusarium mycotoxins to mammalian cell
cultures as determined by the MTT bioassay. Food
and Chem Toxicol 2005;43(5):755-764. [CrossRef] [PubMed]
27 Ren JH, Lin JS, He WS, Sun XM, Li PY,
Chang Y, Liu Y, Li C, Gao XS. RASSF2 induces activated ras-dependent cell
growth inhibition and apoptosis in human pancreatic cancer cells. J Cancer Mol 2006;2(3):117-122.
28
Maruichi M, Takai S, Sugiyama T, Ueki M, Oku H, Sakaguchi M, Okamoto Y,
Muramatsu M, Ikeda T, Miyazaki M. Role of chymase on growth of cultured canine
TenonˇŻs capsule fibroblasts and scarring in a canine conjunctival flap model. Exp Eye Res 2004;79(1):111-118. [CrossRef] [PubMed]
29
Bosonea AM, Wang X, Odenbach J, Fernandez-Patron C. Metalloproteinases in
hypertension and cardiac disease: differential expression and mutual
regulation. Drug Discov Today Dis Models 2011;8(1):29-35.
[CrossRef] [PubMed]
[PMC free article]
30
Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix
metalloproteinases: an overview. Mol Cell
Biochem 2003;253(1-2):269-285. [CrossRef]
[PubMed]
31
Yang SF, Lin CY, Yang PY, Chao SC, Ye YZ, Hu DN. Increased expression of
gelatinase (mmp-2 and mmp-9) in pterygia and pterygium fibroblasts with disease
progression and activation of protein kinase C. Invest Ophthalmol Vis Sci 2009;50(10):4588-4596. [CrossRef]
[PubMed]
32
Kargiotis O, Chetty C, Gondi CS, Tsung AJ, Dinh DH, Gujrati M, Lakka SS,
Kyritsis AP, Rao JS. Adenovirus-mediated transfer of siRNA against MMP-2 mRNA
results in impaired invasion and tumor-induced angiogenesis, induces apoptosis
in vitro and inhibits tumor growth in vivo in glioblastoma. Oncogene 2008;27(35):4830-4840. [CrossRef]
[PubMed] [PMC free article]
33
Wong TT, Mead AL, Khaw PT. Matrix metalloproteinase inhibition modulates
postoperative scarring after experimental glaucoma filtration surgery. Invest Ophthalmol Vis Sci 2003;44(3):1097-1103. [CrossRef]
34
Adegunsoye A, Balachandran J. inflammatory response mechanisms exacerbating
hypoxemia in coexistent pulmonary fibrosis and sleep apnea. Mediators Inflamm 2015;2015:510105. [CrossRef]
[PubMed] [PMC free article]
35
Sang QX. Complex role of matrix metalloproteinase in angiogenesis. Cell Res 1998;8(3):171-177. [CrossRef]
[PubMed]
36
Chetty C, Lakka SS, Bhoopathi P, Rao JS. MMP-2 alters VEGF expression via
alphaVbeta3 integrin-mediated PI3K/AKT signaling in A549 lung cancer cells. Int J Cancer 2010;127(5):1081-1095. [CrossRef]
[PubMed] [PMC free article]
37
Song H, Pan D, Sun W, Gu C, Zhang Y, Zhao P, Qi Z, Zhao S. SiRNA directed
against annexin II receptor inhibits angiogenesis via suppressing MMP2 and MMP9
expression. Cell Physiol Biochem 2015;35(3):875-884. [CrossRef]
[PubMed]
38
Arroyo AG, Iruela-Arispe ML. Extracellular matrix,inflammation,a nd the
angiogenic response. Cardiovascular Res
2010;86(2):226-235. [CrossRef] [PubMed]
[PMC free article]
39
Van Bergen T, Vandewalle E, Van de Veire S, Dewerchin M, Stassen JM, Moons L,
Stalmans I. The role of different VEGF isoforms in scar formation after
glaucoma filtration surgery. Exp Eye Res 2011;93(5):689-699.
[CrossRef] [PubMed]
40
Savio M, Stivala LA, Bianchi L Vannini V, Prosper E. Involvement of the
proliferating cell nuclear antigen (PCNA) in DNA repair induced by alkylating
agents and oxidative damage in human fibroblasts. Carcinogenesis 1998;19(4):591-596. [CrossRef]
41
Kojima S, Sugiyama T, Takai S, Jin D, Ueki M, Oku H, Tabata Y, Ikeda T. Effects
of gelatin hydrogel loading mitomycin C on conjunctival scarring in a canine
filtration surgery model. Invest Ophthalmol
Vis Sci 2015;56(4):2601-2605. [CrossRef]
[PubMed]
[Top]