·Clinical Research··Current Issue· ·Achieve· ·Search
Articles· ·Online
Submission· ·About IJO·
Real-time polymerase chain reaction for the
diagnosis of necrotizing herpes stromal
keratitis
Jun-Xin
Ma1, Lin-Nong Wang1, Ru-Xia Zhou1, Yang Yu2, Tong-Xin Du2
1Department of Ophthalmology,
Nanjing First Hospital, Nanjing Medical University, Nanjing 210006, Jiangsu
Province, China
2Department of Clinical
Nuclear Medicine Center, Nanjing First Hospital, Nanjing Medical University,
Nanjing 210006, Jiangsu Province, China
Correspondence
to: Lin-Nong
Wang. Department of Ophthalmology, Nanjing First Hospital, Nanjing Medical
University, No. 68 Changle Road Qinhuai District, Nanjing 210006, Jiangsu
Province, China. lndoccn@126.com
Received: 2015-02-15
Accepted: 2015-07-28
Abstract
AIM: To design, optimize and
validate a rapid, internally controlled real-time polymerase chain reaction
(RT-PCR) test for herpes simplex virus (HSV) in the diagnosis of necrotizing
herpes stromal keratitis.
METHODS: Tears alone or together
with corneal epithelium scrapings from 30 patients (30 eyes) suspected of
necrotizing herpes stromal keratitis were tested for HSV DNA by RT-PCR. The
samples were collected during the first visit and then on the subsequent 7, 14,
28, 42, and 56d. The symptoms of the patients were scored before treatment to
determine the correlation between HSV concentration in the corneal epithelium
scrapings and clinical scores.
RESULTS: The positive rate (46.4%)
in the corneal epithelium group before the therapy was significantly higher
than that (13.3%) in the tears group (P=0.006).
There were 13 positive HSV patients before the therapy, the concentration of
HSV DNA in corneal epithelium scrapings group was significantly higher than
that in the tears group (paired t-test,
P=0.0397). Multilevel mixed-effects
model analysis showed that the difference between the corneal epithelium
scrapings group and the tears group was statistically significant (P=0.0049). The Spearman rank correlation
analysis indicated a positive correlation between the HSV concentration in the
corneal epithelium scrapings and clinical scores before the treatment (r=0.844, P<0.0001).
CONCLUSION: RT-PCR appears to be a
powerful molecular tool for the diagnosis of necrotizing herpes stromal
keratitis.
KEYWORDS: necrotizing herpes stromal keratitis; real-time polymerase chain
reaction; corneal epithelium scrapings; tears
Citation: Ma JX, Wang LN, Zhou RX, Yu Y, Du TX. Real-time
polymerase chain reaction for the diagnosis of necrotizing herpes stromal
keratitis. Int J Ophthalmol 2016;9(5):682-686
INTRODUCTION
Herpes stromal
keratitis (HSK), which is subdivided into necrotizing herpes stromal keratitis
and immune herpes stromal keratitis[1], is a
leading cause of corneal blindness that accompanies herpes simplex virus (HSV)
infection of the eye[2]. Many
studies[3-5] have demonstrated
that HSV can establish latency in either the trigeminal ganglia or the cornea
after primary
infection and can eventually be reactivated by fever, exposure to ultraviolet
rays[6], general ill-health, emotional
stress, physical exhaustion, mild trauma, menstrual stress[7]
and so on[8].
Previously the
diagnosis of HSK relied on a history of recurrent keratitis, as
well as typical clinical manifestations in the infected eye[9].
However, after therapy, HSK does not have specific clinical features, so the
disease remains a diagnostic and therapeutic challenge to ophthalmologists.
Shimeld et al[10]
successfully
isolated HSV from the cornea in patients with chronic stromal keratitis, and
virus isolation is considered the ˇ°golden standardˇ± in laboratory diagnosis.
However, this technique is time-consuming, has low sensitivity, and requires a
special laboratory for viral processing. Techniques that rely on
immunofluorescence are adversely influenced by false-positive and false-negative
results, small sample size, and subjective variation in the interpretation of
data[11]. The polymerase
chain reaction (PCR), which is sensitive and has a relatively rapid processing time[12], can also be used to detect HSV
DNA. However, the theoretically high sensitivity of PCR is offset by a high
cost and the need for dedicated laboratory space (three separate areas) and
trained technicians[13].
Real-time polymerase chain reaction (RT-PCR), considered a powerful
molecular tool for the diagnosis of necrotizing herpes stromal keratitis, was
developed as an alternative approach. RT-PCR is a variant of PCR that is
performed in a closed system and does not require post-amplification sample
manipulation. Importantly, the ability to quantify the DNA by this method
allows the measurement of as little as several hundred DNA molecules to as much
as hundreds of millions of DNA molecules[14]. RT-PCR
overcomes the drawbacks of conventional PCR, reduces the risk for carry-over
contamination, and eliminates the time-consuming detection step.
Here we report the results of a study testing the use of RT-PCR in the
diagnosis of necrotizing
herpes stromal keratitis. We analyzed the variation trend of the positive rate
and concentration of HSV DNA in corneal epithelial scrapings and tears before
and after the therapy, and we also examined the correlation between the HSV
concentration and clinical scores before the treatment.
SUBJECTS AND METHODS
Subjects A total of
272 specimens (105 corneal
epithelium scrapings and 167 tears) were collected from 30 patients (30 eyes,
18 right and 12 left) with clinically diagnosed necrotizing herpes stromal
keratitis. These patients were treated at the Department of Ophthalmology,
Nanjing First Hospital between September 2012 and September 2013. The patients
included 13 males and 17 females, and their ages ranged from 20 to 56y, with a
mean age of 38.5y. Because the patients had normal kidney functions, were
neither pregnant nor breast feeding, or did not have severe heart, lung, liver,
or kidney dysfunctions or a history of diabetes and malignant tumors, the wide
range in the age of the subjects did not significantly influence the study.
This study was conducted in accordance with the Declaration of Helsinki. This
study was conducted with approval from the Ethics Committee of Nanjing
Medical University. Written informed consent was obtained from all
participants.
Inclusion
and Exclusion Criteria Selection
of patients was based on the following criteria[15]:
1) history of recurrent keratitis; 2) presence of deep stromal infiltration; 3)
typical dendritic or geographical configuration noted during one or more of the
previous attacks; 4) corneal anesthesia; and 5) negative for bacterial and
fungal ulcer (verified by cultivation). If one or more of the five criteria
were not fulfilled or a secondary bacterial or fungal infection was found, the
patient was excluded from the study. Patients recruited in this study were not
undergoing treatment with any drug, including systemic antiviral drugs, or had
stopped antiviral drugs treatment for at
least 1wk. Patients were strictly prohibited from taking other antiviral drugs during this trial, and they had no other eye
problems and had normally functioning kidneys (creatinine clearance rate ˇÝ70
mL/min). Patients were excluded from the study if they were pregnant or
breastfeeding or if they had severe heart, lung, liver, or kidney dysfunctions
or a history of diabetes or malignant tumors. Three patients were excluded without
normal functioning.
Treatment Patients were treated with 0.15% ganciclovir
(GCV) gel solution (one drop each time, 4 times per day, dripped into the
conjunctival sac of the eye) and 0.1% fluorometholone eye drops (1 drop each
time, 3 times a day, dripped into the conjunctival sac of the eye) until
complete recovery, in combination with oral GCV (1000 mg per dose, 3 times per
day for 8wk) followed by oral ACV at a dose of 400 mg twice per day for 6mo.
Specimen Collection Specimens were collected during the
first visit and then on the subsequent 7, 14, 28, 42, and 56d. Corneal
epithelium scrapings were collected[12] by debriding the edge of an ulcer with
sterile needles, then were stored in 100 µL of sterile saline at -70ˇăC until
processed. Tears (100 µL) were collected by stimulating the conjunctival fornix
with minuscule sterile glass capillaries, and then were stored in the same
conditions as the corneal epithelium scrapings.
There
were 28 samples collected from the corneal epithelium scrapings on the initial
visit, and 28, 23, 17, 8, and 1 (105 totally) collected on the respective
follow-up visits. Two of the 30 patients refused to submit to the corneal
epithelium scrapings during the first visit. Corneal epithelium scrapings
collection stopped when the corneal epithelium was intact. The numbers of
samples in the tears group were 30 from the first visit, and 30,
28, 27, 26, and 26 (167 totally) on the respective follow-up visits.
With treatment, the volume of the tears was too small to collect from four
patients.
DNA Extraction and Real-time Polymerase
Chain Reaction All samples obtained from the subjects were stored at -70ˇăC before DNA
extraction. RT-PCR [9] for
the detection of HSV DNA was carried out using the Artus HSV-1 QS-RGQ kit
(Qiagen, China). Reactions were set-up and performed according to the
manufacturerˇŻs instructions, and were executed by the vitro medical diagnostic
device intended for use on the Loche 480 instrument. All reactions were
performed in a total volume of 40 µL. The reaction conditions were as follows:
pre-denaturation at 37ˇć for 5min, followed by 40 cycles of denaturation at 94ˇć
for 1min, annealing at 95ˇć for 5s and extension at 60ˇć for 30s.
Scoring
of Symptoms Patients
were examined by slit lamp before and after the cornea was stained with
fluorescein and then were instructed to grade each of his/her symptoms [(visual
deterioration, redness, ophthalmalgia, lachrymation, photophobia, secretion,
conjunctiva injection, ciliary injection, folliculosis, cornea inflammation, Keratic
precipitate (kp), Tyndall phenomenon, posterior synechia of the iris,
uncomfortable)] by using the numbers 0-4 (0=lowest, 4=highest). The patients
were also asked to indicate the total scores in all follow-up windows. During
follow-up, all subjects were asked about whether any discomfort had occurred
during therapy. All patients orally administered with drugs underwent routine
blood and urine examinations as well as liver and kidney function examinations
to monitor adverse reactions of drugs.
Statistical Analysis All data were analyzed by the
SPSS19.0 and MLwiN2.28 software using the Chi-squared test, paired t-test, Spearman rank
correlation analysis and multilevel mixed-effects model. Values on this study are
reported as mean ˇŔSD or P50 (P25-P75). Results were
considered statistically significant for two-sided P<0.05.
RESULTS
Amplification Curves and Standard Curves of Herpes Simplex
Virus Transcripts Amplification
curves and the standard curve of HSV transcripts are shown in Figure 1. Values
of RT-PCR were normalized to calculate copy numbers of HSV transcripts in the
samples according to the standard curve with LightCycler Software, Version 3.5
(Roche Diagnostic, Inc.)[12].
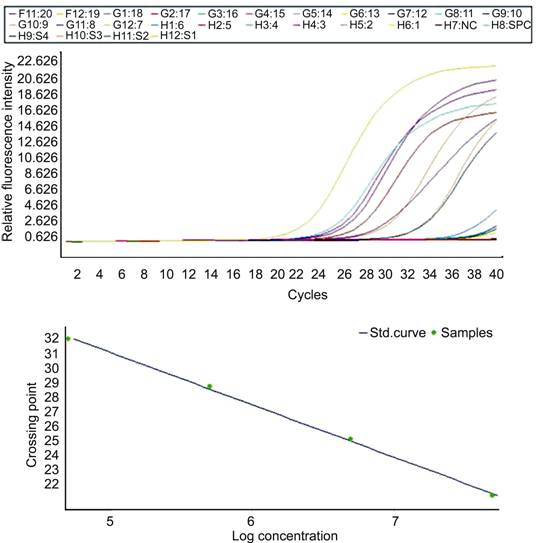
Figure 1 Amplification curves and standard curve of HSV transcripts.
Variation Trend of Positive Rate The comparison of the percentage of positive samples
in the corneal epithelial scrapings and in the tears after each follow-up visit
as detected by RT-PCR is shown in Table 1. The highest positive percentage was
obtained in the corneal epithelium scrapings on the first visit. The
positive percentage was significantly reduced in the corneal epithelium scrapings samples from day 1 to day
56 (8th week), while there was only marginally decline in the
positive percentage of tears samples at the same time. Corneal
epithelium scrapings yielded noticeably higher positive rate (46.4%) than in tears
(13.3%) before the treatment (P=0.006), but
there was no difference between corneal epithelium scrapings samples and tears
samples during the progression.
Table 1 The comparison of
positive rate in two groups with each follow-up point by RT-PCR
|
Group |
Time |
Pre-treatment |
1st week |
2nd week |
4th week |
6th week |
8th week |
|
Corneal epithelium
scrapings |
Positive samples |
13/28 |
2/28 |
2/23 |
1/17 |
0/8 |
0/1 |
|
1P |
|
0.001 |
0.003 |
0.004 |
0.032 |
1.000 |
|
|
Tears |
Positive samples |
4/30 |
2/30 |
1/28 |
0/27 |
0/26 |
0/26 |
|
1P |
|
0.671 |
0.354 |
0.114 |
0.115 |
0.115 |
|
|
P |
0.006 |
1.000 |
0.583 |
0.386 |
Ł |
Ł |
1P: Comparison of 1st
week, 2nd week, 4th week, 6th week and 8th
week to the pre-treatment (Chi-squared test).
Variation Trend of Concentration of
Herpes Simplex Virus DNA Prior to
treatment, 13 (43.3%) of the 30 subjects were positive for HSV either in
the corneal epithelium scrapings or the tears as measured by RT-PCR. Correlation of the
quantitative viral DNA analysis to the positive patients is
summarized in Table 2. The amount of HSV DNA in the corneal epithelium
scrapings was significantly higher than in tears on the first visit (paired t-test, P=0.0397). Multilevel mixed-effects model analysis indicated that
the difference between the corneal epithelium scrapings samples and the tears
samples was statistically significant (P=0.0049);
the concentration was undetectable at 28d after the treatment in the two
groups (P=0.0007) and the decline
rate of two groups was significantly different (P=0.0494).
Table 2 Results of the
quantitative viral DNA analysis to the positive patients
copies/mL
|
Groups |
Pre-treatment |
7th day |
14th day |
28th day |
|
|
Corneal epithelium scrapings |
Sample amount |
13 |
13 |
13 |
13 |
|
|
|
390420.77ˇŔ610715.88 |
529.23ˇŔ1291.86 |
176.54ˇŔ465.70 |
44.23ˇŔ159.48 |
|
|
P50 (P25-P75) |
2460 (2165-636500) |
0 (0-0) |
0 (0-0) |
0 (0-0) |
|
|
Min. |
2030 |
0 |
0 |
0 |
|
|
Max. |
2020000 |
3460 |
1580 |
575 |
|
|
Incidence No. (rate) |
13 (100%) |
2 (15.38%) |
2 (15.38%) |
1 (7.69%) |
|
Tears |
Sample amount |
13 |
13 |
13 |
13 |
|
|
|
893.85ˇŔ2201.78 |
140.15ˇŔ344.99 |
48.00ˇŔ173.07 |
0.00ˇŔ0.00 |
|
|
P50 (P25-P75) |
0 (0-1150) |
0 (0-0) |
0 (0-0) |
0 (0-0) |
|
|
Min. |
0 |
0 |
0 |
0 |
|
|
Max. |
8010 |
1020 |
624 |
0 |
|
|
Incidence No. (rate) |
4 (30.77%) |
2 (15.38%) |
1 (7.69%) |
0 (0.00%) |
Correlation Between Herpes
Simplex Virus Concentration and Clinical Scores Scatter plot of the HSV concentration in
the corneal epithelium scrapings (non-normal distribution) and clinical scores
before the treatment are shown in the Figure 2. Spearman rank correlation
analysis indicated that there was a positive correlation between HSV
concentration and clinical score (r=0.844,
P<0.0001). In all cases where
lower concentrations of HSV were detected, the clinical scores decreased as
well. The symptoms corresponded to the results obtained from the laboratory.
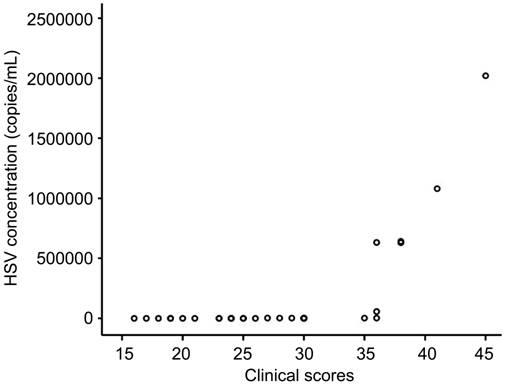
Figure 2 Scatter plot of the HSV concentration in
the corneal epithelium scrapings and before the treatment.
DISCUSSION
In recent years, the incidence of HSK, which has
already reached 31.5/105, has shown an increasing trend, both in
China and other countries. Recurrent cases account for the majority of HSK
cases, the incidence of which is 18.3/105[16].
Little statistical data is available on HSK in developing countries; however,
the prevalence and incidence in developing countries are higher than those in
developed countries. The people in developing countries tend to develop HSK at
an earlier age. Viruses are usually latent in the trigeminal ganglia[17] after primary infection. With
repeated reactivation cycles, viruses can also be found in corneal epithelial
scrapings, stroma, or tears[18].
Necrotizing stromal keratitis is a relatively serious type of HSK, recurrent
attacks of which may lead to blindness. Therefore, rapid and accurate
laboratory diagnosis is quite important. The modified PCR, RT-PCR[19-20], is more
specific and sensitive to viruses than normal PCR, and can be used to detect
the virus more quickly at lower concentration to help make diagnosis sooner.
A
multicenter, prospective, randomized, single-blind, and controlled clinical
trial was conducted by the EYE and ENT Hospital of Fudan University, Hangzhou
First PeopleˇŻs Hospital, and by our lab. We recently published the results from
that study, and here, we used the same criteria to select patients for this
study. In the previous studies, we only used RT-PCR to diagnose HSK, but we did
not monitor the variation in HSV concentration during the treatment. The objectives of this study were to
develop an optimum laboratory test for the diagnosis of necrotizing
herpes stromal keratitis and to determine if there is a correlation between HSV
concentration and clinical scores.
Before
the onset of therapy, the percentage of viral positive (46.4%) corneal
epithelial scrapings and the concentration of virus in those scrapings were
relatively high; however, patients occasionally experienced a mild trauma or
complained about foreign body sensation after sampling. The percentage of viral
positive (13.3%) tear samples and the concentration of virus in tears were
relatively low; however, the sampling of tears does not cause appreciable
discomfort in most patients. Some documents reported that positive rate of HSK
in tears of asymptomatic patients from the American areas (most of them are
white) was between 33.5% and 49%[21-22],
while others have shown that the positive rate of HSK in the tears of
asymptomatic patients in Japan was only 8.5%[23]. It
is possible that many of the asymptomatic patients in Japan were HSK carriers
after primary infection[21]. In our
study, the positive rate of HSK in the tears of patients was 13.3%. Therefore,
we infer that positive rate of HSK in the tears of patients is related to, not
only population demographics, but also to geographical position.
HSV was detected in the corneal epithelial scrapings or tears of 13 of 30
patients when first examined. Although HSV was detected in the tears of only
four of the 13 HSV positive patients, it was detected in the corneal epithelial
scrapings of all 13 patients. When the concentration of virus in the corneal
epithelial scrapings was lower than 105 copies/mL, HSV was not
detected in the tears. When concentration of virus in the corneal epithelial
scrapings was higher than 105 copies/mL, HSV was detected in the
tears of 80% of those patients and the concentration of HSK in the corneal
epithelial scrapings was significantly higher than in the tears.
Spearman rank correlation analysis indicated a positive correlation
between the HSV concentration in the corneal epithelium scrapings (non-normal
distribution) and clinical scores before the treatment. Patients with high
clinical scores also had high concentrations of HSV in the corneal epithelium
scrapings before the treatment, and vice versa. This result suggests that
clinical manifestation before the treatment correlates with laboratory results.
Our current research was done to understand on the relation between clinical
manifestations and viral concentration at the first visit, and to establish a
basis for diagnosis of herpes stromal keratitis.
Based
on these results, we propose that while it is easier to detect virus in the
corneal epithelial scrapings than in the tears, HSV becomes detectable in tears
after replication when the viral concentration is high enough in the corneal
epithelial scrapings to become sufficiently abundant in tears. We propose the
use of tears from patients that refuse to allow collection of corneal
epithelial scrapings or from whom collection of corneal epithelial scrapings
would not be technically feasible. Whenever collecting corneal epithelial
scrapings is feasible, tears cannot replace scrapings for diagnosis, but tears
may act as a substitute for definitive diagnosis.
In conclusion, RT-PCR is a new and effective laboratory diagnostic
technique that can be used to quantitatively test the concentration of HSV,
observe transformation of HSV and make correct diagnosis for herpes stromal
keratitis. The sites from where medical samples were collected before the
treatment clearly affect the percentage of positive results and the
concentration of HSV. Therefore, the accuracy of the diagnosis is improved if
the medical material is collected from the corneal epithelium scrapings. After
the treatment, the percentage of HSK positive samples is low in both corneal
epithelium scrapings and in tears; therefore, the site of sample collection
does not significantly affect the results. Using RT-PCR, we could monitor the
changes in HSV concentration, which enabled us to assess the development of the
disease.
ACKNOWLEDGEMENTS
Conflicts of Interest: Ma JX, None;
Wang LN, None; Zhou RX, None; Yu Y,
None; Du TX, None.
REFERENCES
1
Holland EJ, Schwartz GS. Classification of herpes simplex virus keratitis. Cornea 1999;18(2):144-154. [CrossRef] [PubMed]
2 Morris J, Stuart PM, Rogge M, Potter C, Gupta N, Yin
XT. Recurrent herpetic stromal keratitis in mice, a model for studying human
HSK. J Vis Exp 2012;18(70):e4276. [CrossRef]
3 Kaye SB, Lynas C, Patterson A, Risk JM, McCarthy K,
Hart CA. Evidence for herpes simplex viral latency in the human cornea. Br J Ophthalmol 1991;75(4):195-200. [CrossRef]
4 Cohrs RJ, Randall J, Smith J, Gilden DH, Dabrowski C,
van Der Keyl H, Tal-Singer R. Analysis of individual human trigeminal ganglia
for latent herpes simplex virus type 1 and varicella-zoster virus nucleic acids
using real-time PCR. J Virol
2000;74(24):11464-11471. [CrossRef]
5 Hill JM, Ball MJ, Neumann DM, Azcuy AM, Bhattacharjee
PS, Bouhanik S, Clement C, Lukiw WJ, Foster TP, Kumar M, Kaufman HE, Thompson
HW. The high prevalence of herpes simplex virus type 1 DNA in human trigeminal
ganglia is not a function of age or gender. J
Virol 2008;82(16):8230-8234. [CrossRef] [PubMed] [PMC free article]
6 Ludema C, Cole SR, Poole C, Smith JS, Schoenbach VJ,
Wilhelmus KR. Association between unprotected ultraviolet radiation exposure
and recurrence of ocular herpes simplex virus. Am J Epidemiol 2014;179(2):208-215. [CrossRef]
[PubMed] [PMC free article]
7 Tuokko H, Bloiqu R, Hukkanen V. Herpes simplex virus
type 1 genital herpes in young women: current trend in Northern Finland. Sex Transm Infect 2014;90(2):160. [CrossRef] [PubMed]
8 Farooq AV, Shukla D. Corneal latency and transmission
of herpes simplex virus-1. Future Virol 2011;6(1):101-108.
[CrossRef] [PubMed] [PMC free article]
9 Yoon KC, Im SK, Park HY. Recurrent herpes simplex
keratitis after verteporfin photodynamic therapy for corneal
neovascularization. Cornea
2010;29(4):465-467. [CrossRef]
10 Shimeld C, Tullo AB, Easty DL, Thomsitt J. Isolation
of herpes simplex virus from the cornea in chronic stromal keratitis. Br J Ophthalmol 1982;66(10):643-647. [CrossRef] [PubMed]
11 Elnifro EM, Cooper RJ, Klapper PE, Bailey AS, Tullo
AB. Diagnosis of viral and chlamydial keratoconjunctivitis: which laboratory
test? Br J Ophthalmol
1999;83(5):622-627. [CrossRef] [PubMed] [PMC free article]
12 Kakimaru-Hasegawa A, Kuo CH, Komatsu N, Komatsu K,
Miyazaki D, Inoue Y. Clinical application of real-time polymerase chain
reaction for diagnosis of herpetic diseases of the anterior segment of the eye.
Jpn J Ophthalmol 2008;52(1):24-31. [CrossRef] [PubMed]
13 Hlinomazov¨˘ Z, Loukotov¨˘ V, Hor¨˘ckov¨˘ M, Sery O. The
treatment of HSV1 ocular infections using quantitative real-time PCR results. Acta Ophthalmol 2012;90(5):456-460. [CrossRef] [PubMed]
14 Subhan S, Jose R J, Duggirala A, Hari R, Krishna P,
Reddy S, Sharma S. Diagnosis of herpes simplex virus-1 keratitis: comparison of
Giemsa stain, immunofluorescence assay and polymerase chain reaction. Current Eye Res 2004;29(2-3):209-213. [CrossRef] [PubMed]
15 Oosterhuis JA, van Ganswijk R, Versteeg J. Acyclovir
treatment in stromal herpetic keratitis. Doc
Ophthalmol 1983;56(1-2):81-88. [CrossRef]
[PubMed]
16 Liesegang TJ. Herpes simplex virus epidemiology and
ocular importance. Cornea
2001;20(1):1-13. [CrossRef]
17 Huang FF, Wang ZJ, Zhang CR. Tear HSV-specific
secretory IgA as a potential indicator for recurrent stromal herpes simplex
keratitis: a preliminary study. Cornea
2013;32(7):987-991. [CrossRef] [PubMed]
18 Kennedy DP, Clement C, Arceneaux RL, Bhattacharjee PS,
Huq TS, Hill JM. Ocular herpes simplex virus type 1: is the cornea a reservoir
for viral latency or a fast pit stop? Cornea
2011;30:251-259. [CrossRef] [PubMed] [PMC free article]
19 Marshall D S, Linfert D R, Draghi A, McCarter YS,
Tsongalis GJ. Identification of herpes simplex virus genital infection:
comparison of a multiplex PCR assay and traditional viral isolation techniques.
Mod Pathol 2001;14(3):152-156. [CrossRef] [PubMed]
20 Jazeron JF, Barbe C, Frobert E, Renois F, Talmud D,
Brixi-Benmansour H, Brodard V, Andr¨¦oletti L, Diebold MD, L¨¦v¨şque N.
Virological diagnosis of herpes simplex virus 1 esophagitis by quantitative
real-time PCR assay. J Clin Microbiol
2012;50(3):948-952. [CrossRef] [PubMed] [PMC free article]
21 Kaufman HE, Azcuy AM, Varnell ED, Sloop GD, Thompson
HW, Hill JM. HSV-1 DNA in tears and saliva of normal adults. Invest Ophthalmol Vis Sci 2005;46(1):241-247. [CrossRef] [PubMed] [PMC free article]
22 Kumar M, Hill JM, Clement C, Varnell ED, Thompson HW,
Kaufman HE. A double-blind placebo-controlled study to evaluate valacyclovir
alone and with aspirin for asymptomatic HSV-1 DNA shedding in human tears and
saliva. Invest Ophthalmol Vis Sci 2009;50(12):5601-5608. [CrossRef] [PubMed] [PMC free article]
23 Shimomura Y, Higaki S. The kinetics of herpes virus on
the ocular surface and suppression of its reactivation. Cornea 2011;30 Suppl:S3-7. [CrossRef] [PubMed]
[Top]