・Basic
Research・・Current Issue・ ・Achieve・ ・Search
Articles・ ・Online
Submission・ ・About IJO・ PMC
The role of LOX-1 on innate immunity against Aspergillus keratitis in mice
Kun He, Li-Hui Yue, Gui-Qiu Zhao, Cui Li, Jing Lin,
Nan Jiang, Qian Wang, Qiang Xu, Xu-Dong Peng, Li-Ting Hu, Jie Zhang
Department of
Ophthalmology, the Affiliated Hospital of Qingdao University, Qingdao 266003,
Shandong Province, China
Correspondence to: Gui-Qiu Zhao. Department of
Ophthalmology, the Affiliated Hospital of Qingdao University, No.16 of Jiangsu
Road, Shinan District, Qingdao 266003, Shandong Province, China.
yankeyuelihui@126.com
Received: 2016-01-28
Accepted: 2016-04-26
Abstract
AIM: To explore the effects of lectin-like ox-LDL receptor (LOX-1) on innate immunity
against Aspergillus fumigatus (A. fumigatus ) in mice cornea.
METHODS: The mRNA levels of LOX-1 were
tested in normal and A. fumigatus infected corneas of
C57BL/6 and BALB/c mice. The expression of LOX-1, pro-inflammatory cytokines
TNF-α, CXCL1 and IL-6, anti-inflammatory cytokines IL-10, and matrix
metalloproteinase 9 (MMP9) were tested with
treatment with LOX-1 neutralizing antibody
or control IgG in A. fumigatus infected
corneas of C57BL/6. Macrophages and neutrophils were extracted from susceptible C57BL/6
mice, and pretreated with LOX-1 neutralizing antibody or IgG, then stimulated
with A. fumigatus. The mRNA levels of LOX-1, TNF-α,
CXCL1, IL-6, IL-10 and MMP9 were evaluated by polymerase
chain reaction.
RESULTS: The expression of LOX-1 was
significantly increased in C57BL/6 mice corneas after A. fumigatus infection compared with BABL/c
mice. After treatment with LOX-1 neutralizing antibody, the expression of
LOX-1, TNF-α, CXCL1, IL-6, MMP9 and IL-10 in C57BL/6 corneas were significantly
decreased compared with treatment with control IgG; the expression of LOX-1,
CXCL1, IL-6 and IL-10 were significantly decreased in macrophages, while TNF-α
and MMP9 expressions had no change; LOX-1, TNF-α, CXCL1, IL-6, MMP9 and IL-10
expressions were significantly decreased in neutrophils.
CONCLUSION: The expression of LOX-1 can
affect the expression of pro-inflammatory and anti-inflammatory cytokines in
fungal infected corneas, macrophages and neutrophils of C57BL/6. LOX-1 inhibition rebalances the inflammatory
response of fungal keratitis in mice.
KEYWORDS: lectin-like
ox-LDL receptor; fungal keratitis; macrophages; neutrophils; mice
DOI:10.18240/ijo.2016.09.01
Citation: He K, Yue LH, Zhao GQ, Li C, Lin J, Jiang N, Wang Q,
Xu Q, Peng XD, Hu LT, Zhang J. The role of LOX-1 on innate immunity against Aspergillus keratitis in mice. Int J Ophthalmol 2016;9(9):1245-1250
INTRODUCTION
Fungal keratitis (FK) is caused by a variety of pathogenic
fungi[1], and Aspergillus fumigatus (A. fumigatus)
is the most common pathogenic fungi[2-3]. Recently the incidence of FK has been increased because
of eye trauma, long-term usage of antibiotic or corticosteroid,
extended usage of contact lens[2]. As a serious infectious corneal disease, FK induced
higher blindness rate than other corneal diseases. Innate immune system is the
first line to defense against fungal infection, and participates in the defense
through pattern-recognition receptors (PRRs). PRRs mainly include C-type lectin
receptors (CLRs), Toll-like receptors (TLRs) and nucleotide-binding oligomerization
domain-like receptors (NLRs)[4].
Lectin-like ox-LDL receptor (LOX-1) is one of type Ⅱ membrane protein which belongs to the C-type lectin
family, and includes a short N-terminal
cytoplasmic domain, a trans-membrane domain and a C-terminal extra-cellular
domain. In 1997,
Sawamara et al[5] found that LOX-1
is the key receptor of ox-LDL in
endothelial cells. LOX-1 is also expressed in monocytes, platelets, smooth
muscle cells, and macrophages[6]. LOX-1 can be induced by lots of pathological stimulus. According to the study of Honjo et al[7], LOX-1 inhibition can reduce the inflammatory response,
and LOX-1 plays an important role in inflammation. LOX-1 can trigger the
oxidative stress response (ROS) which associated with many of the inflammatory signals
activation, such as MAPK and nuclear factor-kappa B (NF-κB)[8]. There’s no study showed the role of LOX-1 in fungal
infected corneas of C57BL/6 mice and in fungal infected macrophages and
neutrophils. Therefore, this study explored the role of LOX-1 in inflammatory
response of A. fumigatus keratitis and A. fumigatus stimulated macrophages and
neutrophils.
MATERIALS AND METHODS
Mice and Reagents
This
study was supported by Qingdao University and the experiment
conformed to the ARVO Statement for the Use of Animals in Ophthalmic and Vision
Research. Eight-week-old female C57BL/6 mice was purchased from the Chang Zhou
Cavens Laboratory Animal Ltd. A.
fumigatus strains (NO3.0772) were purchased from China General
Microbiological Culture Collection Center; Sabouroud medium was purchased from
Babio biotech (Jinan, Shandong Province, China); Delbeccon’s modified Eagle’s
medium (DMEM) and fetal bovine serum (FBS) were purchased from Gibco (San
Diego, California, USA); thioglycollate medium was purchased from Hope
Bio-Technology Co., Ltd. (Qingdao, Shandong Province, China); sodium
thioglycollate was purchased from Xiya Chemical Industry Co., Ltd. (Shanghai,
China); goat anti-mouse LOX-1 neutralizing antibody and goat IgG were purchased
from R&D Systems (Minneapolis,
MN, USA); RNAiso Plus, PrimeScript RT reagent Kit with gDNA Eraser (Perfect
Real Time), primers and SYBR® Premix Ex TaqTM were purchased from
TAKARA (Dalian, Liaoning Province, China).
Corneal Infection
Mice
were anesthetized by chloral hydrate and magnified 40×
using the stereoscopic microscope, and diameter 2 mm scratch was made on
central corneal epithelium. The corneal surface was covered by A. fumigatus, and contact lens was
placed. Eyelids were sutured. Mice were examined at 1 and 3d post infection
(p.i.) to ensure corneas were infected.
Macrophages and Neutrophils Extraction Thioglycollate medium (3 g) or sodium
thioglycollate (3 g) was dissolved by 100 mL distilled water, and liquid was
stored at 4℃ after high pressure sterilization.
For macrophages extraction, 1 mL 3% thioglycollate medium was given by
intraperitoneal injection (i.p.) into C57BL/6 mice, and on 7d p.i. mice were
sacrificed by cervical dislocation. For neutrophils extraction, 1 mL 3% sodium
thioglycollate was given i.p. into C57BL/6 mice, and after 3h, mice were
sacrificed. Abdominal skin of sacrificial mice was wiped by 75% alcohol and
opened along the middle line. Total 5 mL medium with syringe was injected into
mice abdominal cavity. Macrophages and neutrophils were extracted by peritoneal
lavage. Cell suspensions were seeded in 12-well culture plates.
Cells Culture and Stimulation Macrophages and neutrophils were
extracted from susceptible C57BL/6 mice. Cells were seeded in growth medium
containing DMEM and 5% FBS. Cells suspensions (1×106/mL) were seeded
in 12-well culture plates, then cultured in a humidified incubator containing
5% CO2 at 37℃. Macrophages and neutrophils were
stimulated by A. fumigatus.
LOX-1 Neutralization Goat anti-mouse LOX-1 neutralizing
antibody (5 μg/5 μL) or control goat IgG (5 μg/5 μL) was given to the left eyes
of C57BL/6 mice by subconjunctival injection the day before infection. Then
corneas were infected with A. fumigatus
1d p.i. Macrophages and neutrophils were pretreated with LOX-1 neutralizing
antibody (10 μg/mL) or IgG (10 μg/mL) for 2h, then macrophages were incubated
with A. fumigatus for 12h stimulation
and neutrophils were incubated with A.
fumigatus for 8h stimulation.
RNA Isolation and Real-time
Reverse Transcription-polymerase Chain Reaction Assay Normal and infected
corneas were removed at 0, 1 and 3d p.i.. Macrophages were collected at 0, 4,
8, 12, 16, 20h stimulation, and neutrophils were collected at 0, 4, 8, 12, 16h
stimulation. After treatment with LOX-1 neutralizing antibody or IgG, corneas
were removed at 1d p.i., macrophages were collected at 12h stimulation, and
neutrophils were collected at 8h stimulation. RNA was separated from suspension
by RNAiso plus reagent, and then quantified using spectrophotometry rapidly.
RNA (1 μg) was used for first-strand cDNA synthesis according to the reverse
transcription system. Then cDNA (2 μL) was used for polymerase
chain reaction (PCR) in 20 μL reaction volume according to the
manufacturer’s instructions. cDNA was diluted with diethylpyrocarbonate
(DEPC)-treated water according to 1:25 proportion. LOX-1, TNF-α, IL-10, CXCL1,
IL-6 and MMP9 mRNA levels were tested by real-time reverse
transcription (RT)-PCR. Quantification of mRNA expression was analyzed by
threshold cycle (CT) method compared with normalized by β-actin. In Table 1,
primer pairs sequences used for PCR are shown.
Table 1 Nucleotide
sequences of mouse primers for real-time RT-PCR
|
Gene |
GenBank No. |
Primer sequence (5’-3’) |
|
β-actin |
NM_007393.3 |
F: GAT TAC TGC TCT GGC TCC TAG C |
|
R: GAC TCA TCG TAC TCC TGC TTG C |
||
|
LOX-1 |
NM_138648.2 |
F: AGG TCC TTG TCC ACA AGA CTG G |
|
R: ACG CCC CTG GTC TTA AAG AAT TG |
||
|
TNF-α |
NM_013693.2 |
F: ACC CTC ACA CTC AGA TCA TCT T |
|
R: GGT TGT CTT TGA GAT CCA TGC |
||
|
IL-6 |
NM_031168.1 |
F: CAC AAG TCC GGA GAG GAG AC |
|
R: CAG AAT TGC CAT TGC ACA AC |
||
|
CXCL1 |
NM_008176.3 |
F: TGC ACC CAA ACC GAA GTC |
|
R: GTC AGA AGC CAG CGT TCA CC |
||
|
MMP9 |
NM_0113599.2 |
F: CTC
TAC AGA GTC TTT GAG TCC GGC AG |
|
R: TCA GGA ACT TCC AGT ACC AAC CGT C |
||
|
IL-10 |
NM_010548.2 |
F: TGC TAA CCG ACT CCT TAA TGC AGG AC |
|
R: CCT TGA TTT CTG GGC CAT GCT TCT C |
Statistical Analysis The data was expressed as the
mean±standard error of the mean (SEM). Data analysis of cells stimulation at
multiple time points was performed by One-way ANOVA test using Graphpad Prism
3.0 software. t-test was used to
analyze the data of LOX-1 expression in corneas of C57BL/6 and BALB/c mice and
the data of corneas and cells treatment with LOX-1 neutralizing antibody or
control IgG. Values were considered to be significant at P<0.05.
RESULTS
LOX-1 Expression in Cornea of C57BL/6 and BALB/c
Mice To investigate the expression of
LOX-1 in corneas of C57BL/6 and BALB/c mice
after A. fumigatus corneal infection
for 0, 1, 3d p.i., mRNA levels of LOX-1 was tested in normal and infected
corneas of C57BL/6 and BALB/c mice by real-time RT-PCR. Results indicated that
the mRNA levels of LOX-1 (Figure 1, P<0.05)
was significantly higher in corneas of C57BL/6 mice than BALB/c mice at 0, 1,
3d p.i.

Figure 1 A.
fumigatus induced mRNA expression of LOX-1 in corneas of C57BL/6 and BALB/c
mice.
After infected with A. fumigatus for
0, 1, 3d, the mRNA levels of LOX-1 were significantly increased in corneas of
C57BL/6 mice than BALB/c mice at 0, 1, 3d p.i. aP<0.05.
C57BL/6 Mice Treatment with LOX-1 Neutralizing
Antibody Up-regulate LOX-1 Expression in Corneas of C57BL/6 than BALB/c To investigate whether LOX-1 can modulate
the corneal infection in corneas of C57BL/6 mice, C57BL/6 corneas were pretreated with LOX-1 neutralizing
antibody or IgG before infection, and then infected with A. fumigatus at 1d p.i. The mRNA levels of LOX-1 (Figure 2A, P<0.05); pro-inflammatory cytokines
TNF-α (Figure 2B, P<0.01), CXCL1
(Figure 2C, P<0.05) and IL-6
(Figure 2D, P<0.05); MMP9 (Figure
2E, P<0.01); Anti-inflammatory
cytokines IL-10 (Figure 2F, P<0.05)
were significantly decreased after LOX-1 neutralizing antibody treatment
compared with IgG treatment.
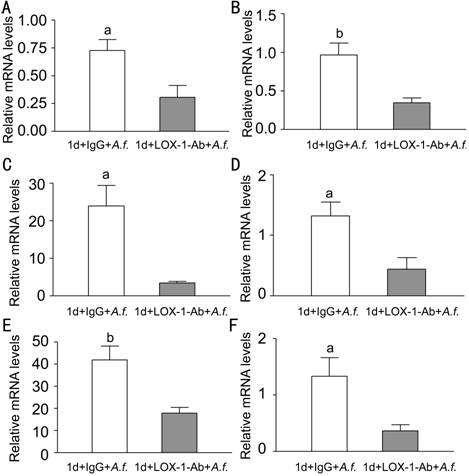
Figure 2
Effects of LOX-1 neutralizing antibody treatment on LOX-1, TNF-α, CXCL1,
IL-6, MMP9 and IL-10 in C57BL/6 mice corneas
Corneas of C57BL/6 mice treatment with LOX-1 neutralizing antibody
compared with IgG at 1d p.i. The mRNA expression of LOX-1 (A), TNF-α (B), CXCL1
(C), IL-6 (D), MMP9 (E), IL-10 (F)were significantly decreased. aP<0.05, bP<0.01.
Effects of LOX-1 on Macrophages of C57BL/6 Mice To investigate the expression of
LOX-1 and other cytokines after A.
fumigatus stimulation of macrophages, macrophages were stimulated with A. fumigatus for 0, 4, 8, 12, 16,
20h.Compared with untreated macrophages, LOX-1 mRNA levels (Figure 3A, P<0.001, P<0.05, P<0.01)
were significantly increased at 8,12, 20h after stimulation. TNF-α mRNA levels (Figure 3B, P<0.001, P<0.01) and CXCL1 mRNA levels (Figure 3C, P<0.001, P<0.01)
were significantly increased at 12, 16h, while levels of IL-6 (Figure 3D, P<0.001, P<0.01) were significantly increased at 8, 12h. MMP9 mRNA
levels (Figure 3E, P<0.05, P<0.001, P<0.001) were significantly increased at 12, 16, 20h, while
IL-10 mRNA levels (Figure 3F, P<0.01)
were significantly increased at 12h. To investigate the effect of LOX-1 on
macrophages, macrophages were pretreated with LOX-1 neutralizing antibody or
IgG for 2h, followed by stimulation of A.
fumigatus for 12h. Data indicated that mRNA levels of LOX-1 (Figure 4A, P<0.05) were significantly decreased
in antibody treatment compared with IgG treatment. Levels of pro-inflammatory
cytokines CXCL1 (Figure 4C, P<0.05)
and IL-6 (Figure 4D, P<0.05) mRNA
levels were significantly decreased, while TNF-α (Figure 4B) mRNA levels showed
no difference. MMP9 mRNA levels (Figure 4E, P<0.05)
were significantly increased, while anti-inflammatory cytokines IL-10 mRNA
levels (Figure 4F, P<0.05) were
significantly decreased.
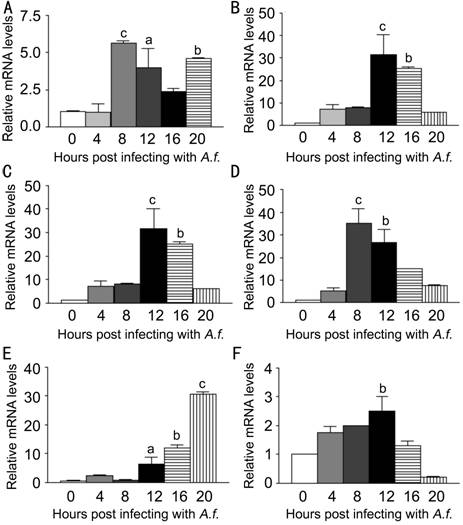
Figure 3
LOX-1, TNF-α, CXCL1, IL-6, MMP9 and IL-10 mRNA expression induced by A. fumigatus stimulation in macrophages extracted from C57BL/6 mice
Macrophages were infected with A. fumigatus and mRNA expressions were
evaluated at 0, 4, 8, 12, 16 and 20h. A: Compared with normal macrophages, the
mRNA expression of LOX-1 was significantly increased in the infected cells at
8, 12, 20h; B: The mRNA expression of TNF-α was significantly increased in the
infected cells at 12, 16h; C: The mRNA expression of CXCL1 was significantly
increased in the infected cells at 12, 16h; D: The mRNA expression of IL-6 was
significantly increased in the infected cells at 8, 12h; E: The mRNA expression
of MMP9 was significantly increased in the infected cells at 12, 16 and 20h; F:
The mRNA expression of IL-10 was significantly increased in the infected cells
at 12h. aP<0.05, bP<0.01, cP<0.001.
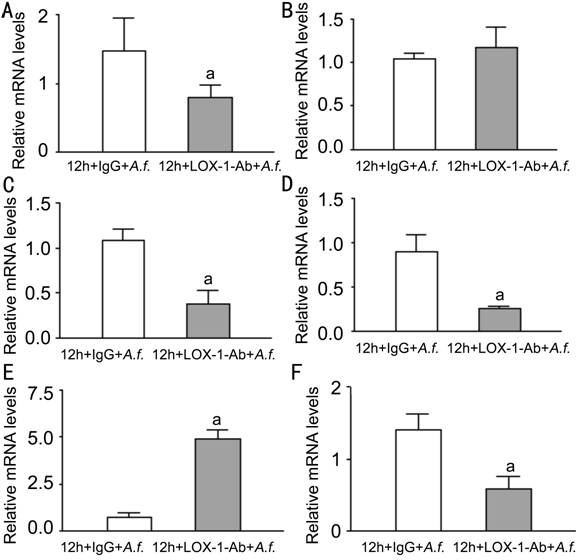
Figure 4
Effects of LOX-1 neutralizing antibody treatment on LOX-1, TNF-α, CXCL1,
IL-6, MMP9 and IL-10 in macrophages of C57BL/6 Macrophages treated with LOX-1
neutralizing antibody compared with IgG at 12h. A: The mRNA expression of LOX-1
was significantly decreased; B: The mRNA expression of TNF-α showed no
difference; C: The mRNA expression of CXCL1 was significantly decreased; D: The
mRNA expression of IL-6 was significantly decreased; E: The mRNA expression of
MMP9 was significantly increased; F: The mRNA expression of IL-10 was
significantly decreased. aP<0.05.
Effects of LOX-1 on Neutrophils of C57BL/6 Mice To investigate the expression of
LOX-1 and other cytokines after A.
fumigatus stimulation of neutrophils, neutrophils were stimulated with A. fumigatus for 0, 4, 8, 12, 16h. The
mRNA levels of LOX-1 and other cytokines were tested by PCR. Compared with
normal neutrophils, LOX-1 mRNA levels (Figure 5A, P<0.001, P<0.01)
were significantly increased at 8, 12h. TNF-α mRNA levels (Figure 5B, P<0.001, P<0.01, P<0.01)
were significantly increased at 4, 8, 12h, while CXCL1 mRNA levels (Figure 5C, P<0.05, P<0.01, P<0.01)
were significantly increased at 8, 12, 16h. Levels of IL-6 (Figure 5D, P<0.05) were significantly increased
at 8h. MMP9 mRNA levels (Figure 5E, P<0.001 P<0.05, P<0.01) were significantly increased at 8, 12, 16h, while IL-10
mRNA levels (Figure 5F, P<0.001)
were significantly increased at 8h. To investigate the effect of LOX-1 on
neutrophils, neutrophils were pretreated with LOX-1 neutralizing antibody or
IgG for 2h, followed by A. fumigatus
stimulation for 8h. The mRNA levels of LOX-1 (Figure 6A, P<0.05) were significantly decreased in LOX-1 neutralizing
antibody treatment compared with IgG treatment. The levels of pro-inflammatory
cytokines TNF-α (Figure 6B, P<0.01),
CXCL1 (Figure 6C, P<0.001) and
IL-6 (Figure 6D, P<0.05) were
significantly decreased. MMP9 mRNA levels (Figure 6E, P<0.01) were significantly decreased. Anti-inflammatory
cytokines IL-10 mRNA levels (Figure 6F, P<0.001)
were significantly decreased.
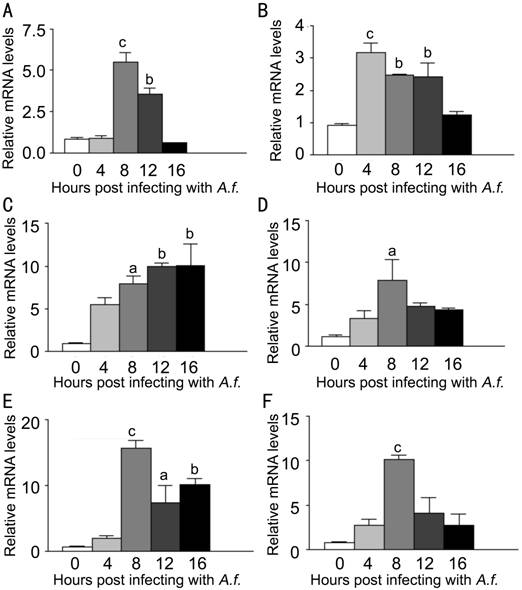
Figure 5
LOX-1, TNF-α, CXCL1, IL-6, MMP9 and IL-10 mRNA expression induced by A. fumigatus stimulation in neutrophils extracted from C57BL/6 mice
Neutrophils were infected with A. fumigatus, and mRNA expression was
evaluated at 0, 4, 8, 12 and 16h. A: Compared with normal neutrophils, the mRNA
expression of LOX-1 was significantly increased in the infected cells at 8,
12h; B: The mRNA expression of TNF-α was significantly increased in the
infected cells at 4, 8 and 12h; C: The mRNA expression of CXCL1 was significantly
increased in the infected cells at 8, 12 and 16h; D: The mRNA expression of
IL-6 was significantly increased in the infected cells at 8h; E: The mRNA
expression of MMP9 was significantly increased in the infected cells at 8, 12
and 16h; F: The mRNA expression of IL-10 was significantly increased in the
infected cells at 8h. aP<0.05,
bP<0.01, cP<0.001.
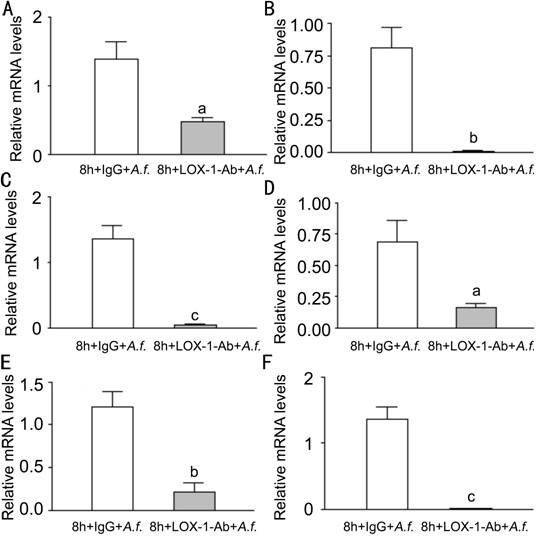
Figure 6
Effects of LOX-1 neutralizing antibody treatment on LOX-1 TNF-α, CXCL1,
IL-6, MMP9 and IL-10 in neutrophils of C57BL/6 LOX-1 neutralizing antibody treatment
compared with IgG at 8h. A: The mRNA expression of LOX-1 was significantly
decreased; B, C, D: The mRNA expression of TNF-α, CXCL1 and IL-6 were
significantly decreased; E: The mRNA expression of MMP9 was significantly
decreased; F: The mRNA expression of IL-10 was significantly decreased. aP<0.05, bP<0.01, cP<0.001.
DISCUSSION
FK, a serious
and infectious corneal ulcer disease, is common in many developing countries[9-11]. As the first line,
innate immune response plays an important role in defense against fungus. The
research of FK innate immune mechanism has a great significance to elucidate
the pathogenesis, and provides the direction and basis for clinical treatment.
LOX-1 belongs
to the C-type lectin family, and is expressed in endothelial cells, monocytes,
platelets, smooth muscle cells, and macrophages. There was study showed that in
LOX-1 knockout mice with
endotoxemia, the lung injury was significantly reduced, and NF-κB activation
was reduced in lungs[12].
LOX-1 expression can affect downstream inflammatory pathways[13-15]. After LOX-1 bound
by pathogen-associated molecular patterns (PAMPs), ROS is triggered which leads
to cell apoptosis by oxidative stress reaction and plays an important role in
inflammatory pathways, such as NF-κB. The inflammatory pathways NF-κB promotes
the expression of pro-inflammatory cytokines, such as TNF-α, CXCL1 and IL-6. In
addition to inflammatory pathways, LOX-1 activation induces pro-angiogenic
proteins, such as MMP-9[16-19].
In this study,
LOX-1 expression was significantly unregulated in mice corneas after A. fumigatus
infected and significantly higher in C57BL/6 than BALB/c, explaining that LOX-1
possibly participated in the inflammatory infection. Study of Zou et al[20] had
showed that the corneal transparency of C57BL/6 mice was lower than BALB/c mice
after corneas infection, and C57BL/6 corneas contained fewer pathogens than
BALB/c corneas. To explore the role of LOX-1 in inflammatory response of FK, LOX-1 neutralizing antibody was
used to treat C57BL/6 corneas. The data showed that the expression of LOX-1 was
inhibited and significantly decreased after treatment with neutralizing
antibody compared with control IgG. It has been known that LOX-1 activation can
induce the pro-inflammatory pathways NF-κB which promotes the expression of
pro-inflammatory cytokines. Data analysis showed that the expression of
pro-inflammatory cytokines TNF-α, CXCL1 and IL-6 were also significantly
decreased. The result is similar to Wu et
al[21] whose study
proved the inflammatory response and
lung injury caused by sepsis induction decreased in LOX-1 knockout mice.
It explained that LOX-1 inhibition can reduce pro-inflammatory cytokines
expression in inflammatory response.
The expression of inflammatory indicator MMP9 was also significantly decreased.
The study of Li et al[22] showed that stronger
LOX-1 signals increased the expression of MMP9 in atherogenic mice. IL-10 is an
anti-inflammatory cytokines which can down-regulated inflammation and
antagonize inflammatory mediators. IL-10 expression was significantly decreased
after treatment with LOX-1 neutralizing antibody compared with control IgG.
In vitro, macrophages and neutrophils were extracted from
C57BL/6 mice. To explore the effects of LOX-1 on macrophages and neutrophils, cells
were pretreated with LOX-1 neutralizing antibody or IgG for 2h. Then cells were
stimulated for 8h (macrophages) and 12h (neutrophils) which based on the result
of cells stimulation at multiple time points. The data showed that LOX-1,
CXCL1, IL-6, and IL-10 expressions were significantly decreased in macrophages;
LOX-1 and inflammatory cytokines expressions were significantly decreased in
neutrophils. These results were consistent with in vivo. The study of Lee et
al[23] showed that pro-inflammatory
cytokines expression IL-6 was significantly increased in human umbilical vein
endothelial cells treatment with LOX-1 key ligand ox-LDL compared with control
cells. And in research of González-Chavarría
et al[24] has
explained that ox-LDL increases the expression of MMP9 in prostate cancer
cells. It explained that inflammatory cytokines expressions reduced after LOX-1
inhibition.
In conclusion,
LOX-1 participates in the innate immune system against fungal infections in mice, and LOX-1 inhibition can reduce the inflammatory
response.
ACKNOWLEDGEMENTS
Foundations: Supported by the National Natural
Science Foundation of China (No.81470609; No.81170825); Key Project of Natural
Science Foundation of Shandong Province (No.ZR2012HZ001); Specialized
Research Fund for the Doctoral Program of Higher Education (No.20123706110003);
Youth Project of Natural Science Foundation of Shandong Province
(No.ZR2013HQ007).
Conflicts of Interest: He K, None; Yue LH, None;
Zhao GQ, None; Li C, None; Lin J, None;
Jiang N, None; Wang Q, None; Xu Q,
None; Peng XD, None; Hu LT, None; Zhang J, None.
REFERENCES
1 Nielsen E,
Heegaard S, Prause JU, Ivarsen A, Mortensen KL, Hjortdal J. Fungal
keratitis-improving diagnostics by confocal microscopy. Case Rep Ophthalmol 2013;4(3):303-310. [CrossRef] [PubMed] [PMC free article]
2 Xie L, Dong
X, Shi W. Treatment of fungal keratitis by penetrating keratoplasty. Br J Ophthalmol 2001;85(9):1070-1074. [CrossRef] [PubMed] [PMC free article]
3 Thomas PA,
Kaliamurthy J. Mycotic keratitis: epidemiology, diagnosis and management. Clin Microbiol Infect
2013;19(3):210-220. [CrossRef]
[PubMed]
4 Kvarnhammar
AM, Cardell LO. Pattern-recognition receptors in human eosinophils. Immunology 2012;136(1):11-20. [CrossRef] [PubMed] [PMC free article]
5 Sawamura T,
Kume N, Aoyama T, Moriwaki H, Hoshikawa H, Aiba Y, Tanaka T, Miwa S, Katsura Y,
Kita T, Masaki T. An endothelial receptor for oxidized low-density lipoprotein.
Nature 1997;386(6620):73-77. [CrossRef] [PubMed]
6 Lee IT, Shih
RH, Lin CC, Chen JT, Yang CM. Role of TLR4/NADPH oxidase/ROS-activated p38 MAPK
in VCAM-1 expression induced by lipopolysaccharide in human renal mesangial
cells. Cell Commun Signal
2012;10(1):33. [CrossRef]
[PubMed] [PMC free article]
7 Honjo M,
Nakamura K, Yamashiro K, Kiryu J, Tanihara H, McEvoy LM, Honda Y, Butcher EC,
Masaki T, Sawamura T. Lectin-like oxidized LDL receptor-1 is a cell-adhesion
molecule involved in endotoxin-induced inflammation. Proc Natl Acad Sci USA 2003;100(3):1274-1279. [CrossRef] [PubMed] [PMC free article]
8 Mehta JL,
Sanada N, Hu CP, Chen J, Dandapat A, Sugawara F, Satoh H, Inoue K, Kawase Y,
Jishage K, Suzuki H, Takeya M, Schnackenberg L, Beger R, Hermonat PL, Thomas M,
Sawamura T. Deletion of LOX-1 reduces atherogenesis in LDLR knockout mice fed
high cholesterol diet. Circ Res
2007;100(11):1634-1642. [CrossRef] [PubMed]
9 Kredics L,
Narendran V, Shobana CS, Vágvölgyi C, Manikandan P; Indo-Hungarian Fungal Keratitis
Working Group. Filamentous fungal infections of the cornea: a global overview
of epidemiology and drug sensitivity. Mycoses
2015;58(4):243-260. [CrossRef]
[PubMed]
10 Wang L, Sun
S, Jing Y, Han L, Zhang H, Yue J. Spectrum of fungal keratitis in central
China. Clin Experiment Ophthalmol
2009;37(8):763-771. [CrossRef] [PubMed]
11 Bharathi
MJ, Ramakrishnan R, Meenakshi R, Padmavathy S, Shivakumar C, Srinivasan M.
Microbial keratitis in South India: influence of risk factors, climate, and
geographical variation. Ophthalmic
Epidemiol 2007;14(2):61-69. [CrossRef] [PubMed]
12 Zhang P,
Liu MC, Cheng L, Liang M, Ji HL, Fu J. Blockade of LOX-1 prevents endotoxin-induced
acute lung inflammation and injury in mice. J
Innate Immun 2009;1(4):358-365.
[CrossRef] [PubMed]
13 Li D, Mehta
JL. Upregulation of endothelial receptor for oxidized LDL (LOX-1) by oxidized
LDL and implications in apoptosis of human coronary artery endothelial cells:
evidence from use of antisense LOX-1 mRNA and chemical inhibitors. Arterioscler Thromb Vasc Biol
2000;20(4):1116-1122. [CrossRef]
14 Kuge Y,
Kume N, Ishino S, Takai N, Ogawa Y, Mukai T, Minami M, Shiomi M, Saji H.
Prominent lectin-like oxidized low density lipoprotein (LDL) receptor-1 (LOX-1)
expression in atherosclerotic lesions is associated with tissue factor
expression and apoptosis in hypercholesterolemic rabbits. Biol Pharm Bull 2008;31(8):1475-1482. [CrossRef]
15 Ishino S,
Mukai T, Kuge Y, Kume N, Ogawa M, Takai N, Kamihashi J, Shiomi M, Minami M,
Kita T, Saji H. Targeting of lectinlike oxidized low-density lipoprotein
receptor 1 (LOX-1) with 99mTc-labeled anti-LOX-1 antibody: potential agent for
imaging of vulnerable plaque. J Nucl Med
2008;49(10):1677-1685. [CrossRef]
[PubMed]
16 Sugimoto K,
Ishibashi T, Sawamura T, Inoue N, Kamioka M, et al. LOX-1-MT1-MMP axis is crucial for RhoA and Rac1 activation
induced by oxidized low-density lipoprotein in endothelial cells. Cardiovasc Res 2009;84(1):127-136. [CrossRef] [PubMed]
17 Garbin U,
Fratta Pasini A, Stranieri C, Manfro S, Mozzini C, Boccioletti V, Pasini A,
Cominacini M, Evangelista S, Cominacini L. Effects of nebivolol on endothelial
gene expression during oxidative stress in human umbilical vein endothelial
cells. Mediators Inflamm
2008;2008:367590. [CrossRef]
[PubMed] [PMC free article]
18 Dandapat A,
Hu C, Sun L, Mehta JL. Small concentrations of oxLDL induce capillary tube
formation from endothelial cells via LOX-1-dependent redox-sensitive pathway. Arterioscler Thromb Vasc Biol
2007;27(11):2435-2442. [CrossRef]
[PubMed]
19 Hu C,
Dandapat A, Mehta JL. Angiotensin II induces capillary formation from
endothelial cells via the LOX-1 dependent redox-sensitive pathway. Hypertension 2007:50(5):952-957. [CrossRef] [PubMed]
20 Zou Y,
Zhang H, Li H, Chen H, Song W, Wang Y. Strain-dependent production of
interleukin-17/interferon-γ and matrix remodeling-associated genes in
experimental Candida albicans keratitis. Mol
Vis 2012;18:1215-1225. [PMC free article]
[PubMed]
21 Wu Z,
Sawamura T, Kurdowska AK, Ji HL, Idell S, Fu J. LOX-1 deletion improves
neutrophil responses, enhances bacterial clearance, and reduces lung injury in
a murine polymicrobial sepsis model. Infect
Immun 2011;79(7):2865-2870. [CrossRef] [PubMed] [PMC free article]
22 Li D, Patel
AR, Klibanov AL, Kramer CM, Ruiz M, Kang BY, Mehta JL, Beller GA, Glover DK,
Meyer CH. Molecular imaging of atherosclerotic plaques targeted to oxidized LDL
receptor LOX-1 by SPECT/CT and magnetic resonance. Circ Cardiovasc Imaging 2010;3(4):464-472. [CrossRef] [PubMed] [PMC free article]
23 Lee WJ, Ou
HC, Hsu WC, Chou MM, Tseng JJ, Hsu SL, Tsai KL, Sheu WH. Ellagic acid inhibits
oxidized LDL-mediated LOX-1 expression, ROS generation, and inflammation in
human endothelial cells. J Vasc Surg
2010;52(5):1290-1300. [CrossRef]
[PubMed]
24
González-Chavarría I, Cerro RP, Parra NP, Sandoval FA, Zuñiga FA, Omazábal VA,
Lamperti LI, Jiménez SP, Fernandez EA, Gutiérrez NA, Rodriguez FS, Onate SA,
Sánchez O, Vera JC, Toledo JR. Lectin-like oxidized LDL receptor-1 is an
enhancer of tumor angiogenesis in human prostate cancer cells. PLoS One 2014;9(8):e106219. [CrossRef] [PubMed] [PMC free article]
[Top]