
・Basic Research・Current Issue・ ・Achieve・ ・Search Articles・ ・Online Submission・ ・About IJO・ PMC
Inhibition effect of curcumin on
UVB-induced secretion of pro-inflammatory cytokines from corneal limbus
epithelial cells
Shih-Chun Chao1,2,3, Dan-Ning Hu4,5, Joan Roberts6,
Xilun Shen4, Chia-Yi Lee1, Chan-Wei Nien1,
Hung-Yu Lin1,7,8,9
1Department of
Ophthalmology, Show Chwan Memorial Hospital, Changhua 50093, Taiwan, China
2Department of Electrical
and Computer Engineering, National Chiao Tung University, Hsinchu 30010,
Taiwan, China
3Department of Optometry,
Central Taiwan University of Science and Technology, Taichung 40601, Taiwan,
China
4Tissue Culture Center, New
York Eye and Ear Infirmary of Mount Sinai, New York, NY 10003, USA
5Ichan School of Medicine
in Mount Sinai, New York, NY 10029, USA
6Fordham University, New
York, NY 10023, USA
7Institute of Medicine,
Chung Shan Medical University, Taichung 40201, Taiwan, China
8Department of Optometry,
Chung Shan Medical University, Taichung 40201, Taiwan, China
9Department of Optometry,
Yuanpei University of Medical Technology, Hsinchu 30015, Taiwan, China
Correspondence to: Hung-Yu Lin. Department of
Ophthalmology, Show Chwan Memorial Hospital, No.526, Sec. 1, Zhongshan Rd., Changhua
City, Changhua County 500, Taiwan, China. anthonyhungyulin@hotmail.com
Received:
2017-02-15
Accepted: 2017-03-23
Abstract
AIM: To study the
effects of curcumin on the secretion of interleukin (IL) -6 and IL-8 by corneal
limbus epithelial cells.
METHODS: Human corneal
limbus epithelial cells were isolated and cultured from donor eyes and
irradiated by UVB at different dosages with or without curcumin. MTT test was
used for studying the effects of UVB and curcumin on the cell viability. The role
of mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB) pathways on
the UVB-induced secretion of IL-6 and IL-8 were tested by addition of their
inhibitors to the culture with or without UVB-radiation. Levels of various
signal pathways, IL-6 and IL-8 in the cells and in the conditioned culture
medium were measured by ELISA analysis.
RESULTS: UVB at 20 mJ/cm2
or less and curcumin at 20 μmol/L or less
did not affect the cell viability of cultured limbus epithelial cells (P>0.05).
UVB irradiation at 10 and 20 mJ/cm2 induced a significant increase
of secretion of IL-6 and IL-8 and upregulated NF-κB and phosphorylated MAPK pathways
of cultured limbus epithelial cells (P<0.05). Various signal pathway
inhibitors, including SP600125 (JNK inhibitor), SB203580 (p38 MAPK inhibitor)
and BAY11-7082 (NF-κB inhibitor)
significantly decreased the UVB-induced secretion of IL-6 and IL-8 secretion (P<0.05).
Curcumin at 5-20 μmol/L
significantly inhibited UVB-induced secretion of IL-6 and IL-8 by limbus
epithelial cells in a dose-dependent manner; while curcumin alone did not
affect the secretion of IL-6 and IL-8. The upregulation of NF-κB and MAPK
pathways induced by UVB treatment was significantly inhibited by curcumin,
suggesting that NF-κB and MAPK pathways
are involved in the inhibitory effect of curcumin on UVB-induced production of
IL-6 and IL-8.
CONCLUSION: Curcumin may be
a promising agent to be explored for the prevention and treatment of pterygium.
KEYWORDS: curcumin; ultraviolet-B;
interleukin-6; interleukin-8; corneal limbus epithelial cells; pterygium
DOI:10.18240/ijo.2017.06.01
Citation: Chao SC,
Hu DN, Roberts J, Shen X, Lee CY, Nien CW, Lin HY. Inhibition effect of curcumin
on UVB-induced secretion of pro-inflammatory cytokines from corneal limbus
epithelial cells. Int J Ophthalmol 2017;10(6):827-833
Article
Outline
INTRODUCTION
Pterygium is a
common ocular surface disease. This is a wing-shaped fibrovascular lesion
originated from the limbus and covered by epithelial cells. Pterygium can
progress to the center of the cornea and causes loss of vision. Previous
studies suggested that pterygium is an inflammatory, invasive, and highly
vascularized growths, arise from activated limbus epithelial cells[1-4]. Epidemiological studies
demonstrated that chronic exposure to sunlight, especially ultraviolet (UV)
irradiation, is the main cause of pterygium. Chronic UV radiation causes the
development of pterygium and the recurrence of pterygium after its surgical
excision[4-6].
Experimental
animal pterygium models have not been established previously[4].
Therefore, an in vitro model has been developed for the investigation of
the pathogenesis and treatment of pterygium by using cultured human ocular
surface cells from normal tissues or excised pterygium specimens[7-10].
Pterygium
tissue contains a high level of pro-inflammatory cytokines. UVB irradiation on
human epithelial cells or fibroblasts isolated from normal ocular surface
tissues or surgical excised pterygium specimens stimulate the expression and
secretion of several pro-inflammatory cytokines and chemokines, such as tumor
necrosis factor (TNF)-α, interleukin (IL)-1, IL-6, and IL-8[1,7-10]. This in vitro model has
been repeatedly used for studying the pathogenesis of pterygium and for the
search of medications that might be used for the prevention and treatment of
pterygium[1,7-11].
Chronic
inflammatory reaction is involved in the pathogenesis of pterygium[1,4,7-9].
Up-regulation of various pro-inflammatory factors plays an important role in
the pathogenesis of pterygium[7-9].
IL-6 is up-regulated in pterygium tissues. This cytokine has a potent
pro-inflammatory effect and also stimulates angiogenesis[7-8,12]. IL-8 (CXCL8) attracts
neutrophil, T cell and monocytes into the tissues, leads to an inflammatory
reaction[13]. IL-8 also induces angiogenesis[13]. All of these effects of these two cytokines lead to
the development of inflammatory response and angiogenesis in the pterygium. The
expression of IL-6 and IL-8 could be induced by UVB irradiation in normal
corneal and pterygium tissues and their various cell components[7-9,14]. Pterygium
begins growing from limbus epithelial cells and UVB irradiation also induces
inflammatory reactions in these cells earlier than other cell types lined
ocular surface[2-3]. Therefore,
it is appropriate to use cultured limbus epithelial cells as an in vitro
model for the investigation of the effects of UVB and various medication on the
progress of pterygium
Curcumin
(diferuloylmethane), is a β-diketones, a yellow coloring agent extracted from
turmeric, has a wide array of pharmacological and biological activities
including chemopreventive, chemotherapeutic and anti-proliferative potentials[15]. In vitro study, experimental animal study and
clinical trials indicated that curcumin can inhibit inflammation via the
decrease of expression of various pro-inflammatory cytokines, chemokines,
transcription factors and relevant signal pathways[15-19]. Curcumin inhibits UVB-induced expression of IL-6,
IL-8 and TNF-α in keratinocytes through the down-regulation of
mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB)
signal pathways[15,20-21].
The effects of curcumin on UVB-induced inflammation in cells from pterygium or
normal ocular surface tissues have not been previously reported.
The purpose of
the present study was to investigate the effects of curcumin on UVB-induced
secretion of IL-6 and IL-8 from cultured human limbus epithelial cells and to
explore the possibility of using curcumin in the prevention and treatment of
pterygium.
MATERIALS AND METHODS
Curcumin Curcumin (99.5% purity) was obtained
from Sigma-Aldrich (St. Louis, MO, USA). Curcumin was dissolved in dimethyl
sulfoxide (DMSO) to make a 20 mmol/L stock solution and was added to the medium
at different concentrations. Cells were treated with 0.25% DMSO as the control
group.
Cell
Culture Limbus epithelial cells were isolated
by us (Hu DN) in the Tissue Culture Center, New York Eye and Ear Infirmary from
donor eyes supplied by the New York Eye Bank for Sight Restoration (New York,
NY, USA). The Eye Bank obtained the donor’s consent before the collection of
the eyes. The principles outlined in the Declaration of Helsinki (2008) have
been followed in the present study. The cornea with limbus and 2 mm wide of
sclera were excised from the eyeball, then, the cornea and sclera were excised
to leave approximately 1 mm on either side of the limbus. The limbus tissue was
washed with Hank's solution (GIBCO, Grand Island, NY, USA) three times and than
immersed in a 1.2 U/mL dispase II solution (Sigma) for 2h at 37℃. After
the enzymatic dissociation, the limbus epithelial cells were gently scraped by
using a iris spatula under the stereo-microscope to isolate the limbus
epithelial cells from the Bowman's membrane. Cells were collected and
centrifuged. Pellets were resuspended by Ham's F12 nutrient mixture with 10%
fetal bovine serum (all from GIBCO), seeded into the culture flask and
incubated in a CO2-regulated incubator in humidified 95% air/5% CO2
atmosphere. Seven days later, culture medium were replaced by the defined
Keratinocyte-serum free medium (K-SFM, GIBCO). Cells were observed under
phase-contrast microscope each day and the culture medium was changed three
time a week. After reaching confluence, the limbus epithelial cells were
detached using 0.25% trypsin solution (GIBCO), diluted 1:3 and subcultured.
Cell cultures in the second passage were used in this study.
Ultraviolet-B
Irradiation Limbus epithelial cells
were seeded into 12 well plates and grew to 75% confluence. Cultures were
washed and covered with a thin layer of PBS before irradiation to remove
potential phototoxic materials in culture medium. Cells were irradiated with
various dosages of UVB using 20037 312 UVB bulbs (Staratagene, La Jolia, CA,
USA), which emit UVB with a spectral peak at 312 nm. PMA 2100 Data Logging
Radiometer (Solar Light, Inc., Genside, PA, USA) was used for monitoring and
calibrating the intensity of UVB radiation. After UVB irradiation, cells were
washed with PBS and cultured with fresh pre-warmed K-SFM for various periods.
MTT Study Cell viability of limbus epithelial
cells cultured with or without UVB irradiation was tested by the MTT assay.
Cells at a density of 5×103 cells/well were seeded into each well in
black well 96-well plates (Sigma). After incubation for 24h, cells were
irradiated by UVB at 10, 20 and 50 mJ/cm2 as described above. After
24h, 50 µL tetrazolium bromide,
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, 1 mg/mL) (Sigma)
was added and cells were cultured at 37℃. Four hours later, the
medium was removed and DMSO (100 µL, Sigma) was added. The optical density as
the parameter of cell viability was measured at 540 nm with a microplate reader
(Multiskan EX, Thermo, Ventana, Finland). Viability of limbus epithelial cells
cultured with or without curcumin was also tested by the MTT assay. Briefly,
cells were seeded into 96-well plates. After 24h, cells were cultured with or
without curcumin at 10, 20 and 50 μmol/L concentrations for 24h and the cell
viability was analysis by MTT assay as described above.
Effects of
Ultraviolet-B and Curcumin on IL-6 and IL-8 Secretion Limbus epithelial cells were seeded into
12-well plates at the density of 2×105 cells per well. After
cultured for 24h, curcumin at the final concentrations of 5, 10, and 20 μmol/L
was added to the culture medium. One hour later, culture medium was withdrawn,
washed and cultured with PBS and irradiated with UVB at 20 mJ/cm2 as
described above. Immediately after irradiation, PBS was replaced by fresh
culture medium. Cells were cultured for 24h. Conditioned culture media were
collected and centrifuged to remove dead cells and debris. The supernatants
were stored at -70℃ until analysis. Tests were performed
in triplicate.
Study of
p38 Mitogen-activated Protein Kinase and c-Jun N-terminal Kinase Levels in
Cultured Limbus Epithelial Cells Limbus epithelial cells
(1×106) were seeded into 6-well plates. Curcumin was
added to the culture to obtain a final concentrations at 20 μmol/L 24h later.
Cultures were irradiated with UVB (20 mJ/cm2) 1h later. Cells were
harvested 24h later and protein was extracted. c-Jun N-terminal kinace (JNK)
and p38 MAPK ELISA kits (with sensitivity at 0.8 U/mL) (Biosource, Camarillo,
CA, USA) were used for the measurement of phosphorylated JNK and p38 MAPK
levels in protein extracted from collected cells according to the
manufacturer’s instructions, respectively. The results (in triplicate) were
expressed as percentages of the control, which were the cultures not treated by
curcumin and UVB.
Study of
NF-κB Levels in Nuclear Extracts from Cultured Limbus Epithelial Cells Limbus epithelial cells were seeded
and treated as described above. Cells were scraped 30min after the treatment of
UVB. Nuclear fraction was obtained by the treatment of collected cells with
hypotonic buffer (BioSource). Nuclear extracts were obtained by the treatment
of nuclear fraction with cell extraction buffer (BioSource). NF-κB ELISA kit
(Invitrogen) was used for the measurement of NF-κB levels according to the
manufacturer’s instructions. The results (in triplicate) were expressed as
percentages of the control, which were the cultures not treated by curcumin and
UVB.
Effects of
Mitogen-activated Protein Kinase and NF-κB Inhibitors on UVB-induced Secretion
of IL-6 and IL-8 Cells (0.2×106
cells/well) were seeded into multi-well plates (12 well). Signal pathway
inhibitors were added 24h later [JNK inhibitor: SP600125; p38 MAPK inhibitor:
SB203580; NF-κB inhibitor: BAY11-7082 (Calbiochem, the concentrations of the
MAPK inhibitors and NF-κB inhibitor were 10 µmol/L and 5 µmol/L, respectively)]
and cultured for 1h. Cells were then treated by UVB as described above. After
24h, conditioned medium was collected, centrifuged and stored at deep freezer.
Measurement
of IL-6 and IL-8 Protein Levels Enzyme-linked immunosorbent
assay (ELISA) was used for the measurement of IL-6 and IL-8 protein levels in
the supernatant of cultured limbus epithelial cells. Quantikine IL-6 ELISA kit
and IL-8 ELISA kit (R&D Systems, Minneapolis, MN, USA) were used to
determine the levels of IL-6 or IL-8 according to the manufacturer's
instruction. The optical density of the ELISA samples was measured at 450 nm
and corrected by 540 nm using a microplate reader (Multiskan EX, Thermo,
Ventana, Finland). The amounts of IL-6 and IL-8 (pg/mL) were calculated from a
standard curve. The sensitivity of the assay for IL-6 and IL-8 was 0.7 pg/mL
and 3.5 pg/mL, respectively.
Tests were performed in triplicate.
Statistical
Analysis Each
experiment was replicated 3 times and the data were presented as mean ± standard
deviation (SD). A Student's t-test was performed to assess the
significance of difference between the means of the tested group and the
controls. Values of P<0.05 were considered statistically significant.
All data analysis was performed using specific software (SPSS 19.0, SPSS Inc.,
Chicago, IL, USA).
RESULTS
MTT Study Cell viability of cultured limbus
epithelial cells irradiated by 10-20 mJ/cm2 UVB were not affected by
UVB irradiation (P>0.05 as compared with the controls). Cells irradiated
with UVB at 50 mJ/cm2 showed a significant decrease of cell
viability (P<0.05) (Figure 1A). Therefore, UVB at 20 mJ/cm2
or less were used in the following experiments. Curcumin at 10-20 μmol/L did
not affect the cell viability (P>0.05 as compared with the controls).
However, viability of cells treated with 50 μmol/L curcumin showed a slight but
significant decrease as compared with cells not treated with curcumin (P<0.05)
(Figure 1B). Therefore, curcumin at 20 μmol/L or less were used in the
following experiments.
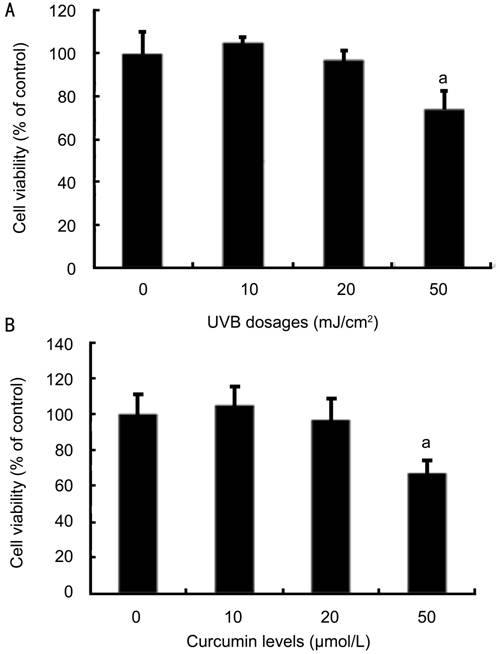
Figure 1 Effects of UVB and curcumin on viability of corneal
limbus epithelial cells A: UVB at 10-20
mJ/cm2 did not affect the viability of human limbus epithelial cells
as determined by MTT test which was significantly affected by UVB at 50 mJ/cm2;
B: Curcumin at 5-20 μmol/L did not affect the viability of human limbus
epithelial cells as determined by MTT test, whereas the cell viablity was
significantly affected by curcumin at 50 μmol/L. Error bars reveal the
means±standard deviation in triplicate tests. aP<0.05 as
compared with the cells not treated with UVB and curcumin.
Effects of
UVB on IL-6 and IL-8 Secretion Cultured
human limbus epithelial cells showed relatively low levels of constitutive
secretion of IL-6 and IL-8 (Figure 2). UVB irradiation at 5, 10 and 20 mJ/cm2
induced a dose-dependent increase
of IL-6 and IL-8 secretion by cultured limbus epithelial cells (Figure 2). IL-6
and IL-8 secretion levels in cells irradiated with 10 and 20 mJ/cm2 UVB
were significantly greater than those in cells not treated with UVB (P<0.05).
UVB at 20 mJ/cm2 caused an increase of IL-6 and IL-8 secretion to
5.8- and 4.7-fold of cells not irradiated, respectively (Figure 2).
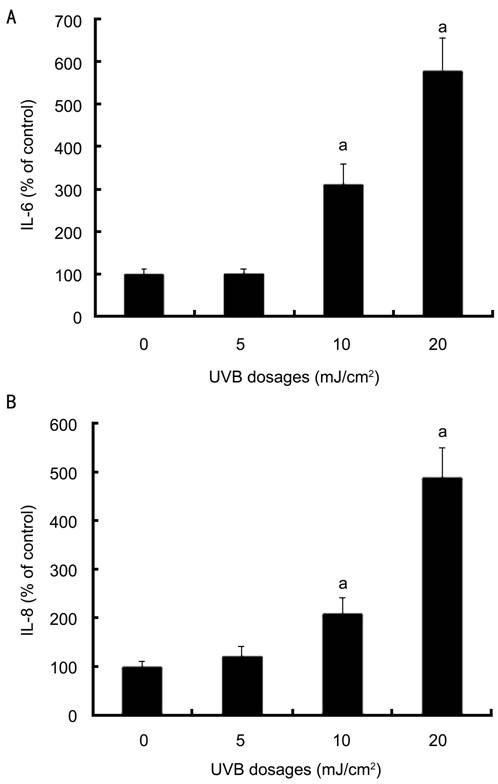
Figure 2
Effects of UVB on secretion of IL-6 and IL-8 by cultured corneal limbus
epithelial cells A: UVB irradiation at 10
and 20 mJ/cm2 induced a significant increase of IL-6 secretion and
IL-8 secretion by cultured limbus epithelial cells; B: The increase of IL-8
secretion. was measured after the same management in (A). Error bars reveal the
means±standard deviation in triplicate tests. aP<0.05 as
determined by ELISA analysis.
Effects of
Curcumin on UVB-induced IL-6 and IL-8 Secretion Curcumin did not affect the
constitutive secretion of IL-6 and IL-8 in cultured human limbus epithelial
cells (Figure 3). Curcumin at 5, 10 and 20 μmol/L dose-dependently inhibited
UVB-induced IL-6 and IL-8 secretion. Both IL-6 and IL-8 secretion in cells
treated with UVB and curcumin at 5-20 μmol/L was significantly decreased as
compared with the positive controls (cells treated with UVB alone, P<0.05)
(Figure 3). IL-6 and IL-8 secretion levels in cells treated with UVB and 20
μmol/L curcumin showed no significant difference from those in the negative
controls (cells not treated with UVB, P>0.05), indicating that
curcumin at this dosage can completely block UVB-induced secretion of these two
pro-inflammatory cytokines in cultured limbus epithelial cells.
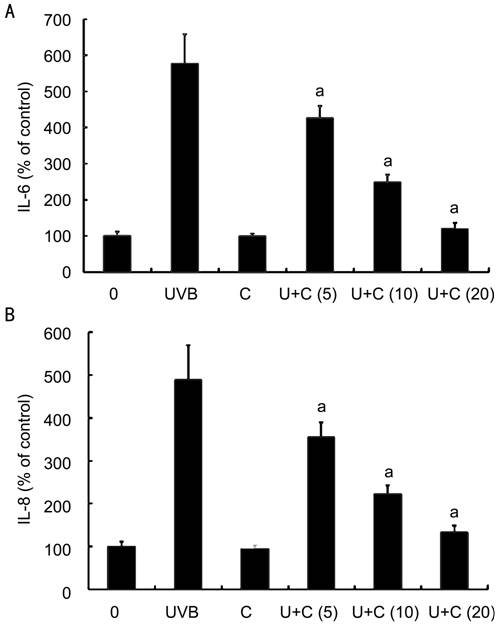
Figure 3
Effects of curcumin on UVB-induced secretion of IL-6 and IL-8 in corneal limbus
epithelia cells A: Curcumin alone (20
μmol/L) did not affect the secretion of IL-6 and IL-8 while curcumin at 5
μmol/L (U+C5), 10 μmol/L (U+C10) and 20 μmol/L (U+C20) significantly inhibited
the UVB-induced secretion of IL-6 and IL-8 by corneal limbus epithelial cells
in a dose-dependent manner. Error bars reveal the means±standard deviation in
triplicate tests. aP<0.05 as compared with cells
irradiated with UVB alone.
Effects of
Ultraviolet-B on NF-κB and Phosphorylated Mitogen-activated Protein Kinase Levels UVB treatment induced significant
increase of phosphorylated JNK and p38 MAPK levels in limbus epithelial cells (P<0.05)
(Figure 4). UVB treatment also induced significant increase of NF-κB levels in
nuclear extracts of the limbus epithelial cells (P<0.05) (Figure 4).
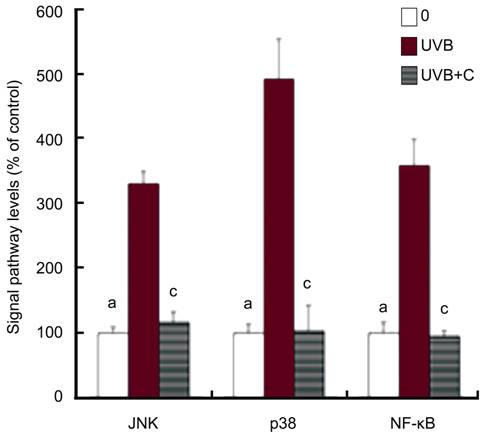
Figure 4
Effects of curcumin on UVB-induced expression of NF-κB and phosphorylated JNK
and p38 MAPK levels in corneal limbus epithelia cells UVB irradiation (20 mJ/cm2)
induced a significant increase of NF-κB and phosphorylated JNK and p38 MAPK
levels of cultured limbus epithelial cells (aP<0.05), as
compared with cells without curcumin and UV irradiation. Curcumin at 20 μmol/L
(UVB+C) significantly decreased UVB-induced expression of NF-κB and
phosphorylated JNK and p38 MAPK levels in corneal limbus epithelial cells (cP<0.05).
Error bars reveal the means±standard deviation in triplicate tests.
Effects of
Curcumin on UVB-induced Expression of NF-κB and Phosphorylated Mitogen-activated
Protein Kinase UVB-induced
increase of phosphorylated JNK and p38 MAPK levels and NF-κB levels in limbus
epithelial cells was significantly reduced by the treatment of curcumin (P<0.05)
(Figure 4).
Effects of
Mitogen-activated Protein Kinase and NF-κB Inhibitors on UVB-induced IL-6 and
IL-8 Secretion JNK inhibitor, p38 MAPK
inhibitor and NF-κB inhibitor significantly decreased the UVB-induced secretion
of IL-6 secretion by cultured human limbus epithelial cells (P<0.05,
as compared with cells irradiated with 20 mJ/cm2 UVB) (Figure 5A).
Secretion of IL-6 in cells treated with UVB and JNK or p38 MAPK inhibitor still
slightly higher than that in cells not treated with UVB (P<0.05);
while secretion of IL-6 in cells treated with UVB and NF-κB inhibitor did not
differ from cells not treated with UVB (P>0.05) (Figure 5A). These
results suggested that NF-κB inhibitor can completely block the UVB-induced
secretion of IL-6 in limbus epithelial cells, while JNK or p38 MAPK inhibitor only
had a partially blocking effect.
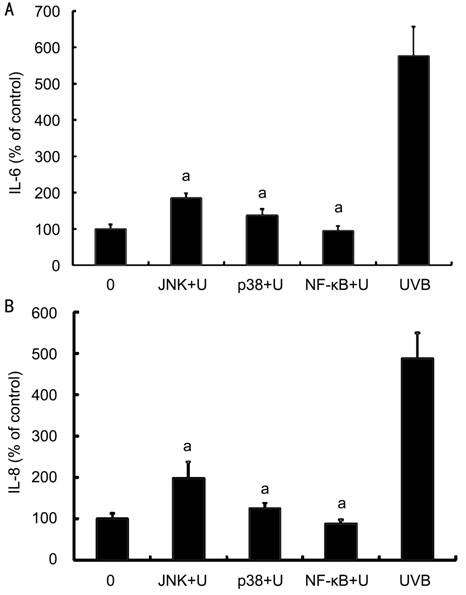
Figure 5
Effects of MAPK and NF-κB inhibitors on UVB-induced secretion of IL-6 and IL-8
by corneal limbus epithelial cells A: Various signal pathway
inhibitors, including SP600125, JNK inhibitor (JNK); SB203580, p38 MAPK
inhibitor (p38) and BAY11-7082, NF-κB inhibitor (NF-κB) significantly decreased
the UVB-induced secretion of IL-6 by cultured limbus epithelial cells; B: The
secretion of IL-8 was measured after the same management in (A). Error bars
reveal the means±standard deviation in triplicate tests. aP<0.05
as compared with cells irradiated with UVB alone.
DISCUSSION
The effects of
UVB on the production of IL-6 and IL-8 of limbus epithelial cells have not been
reported previously. In the present study, UVB induced significantly increase
of secretion of IL-6 and IL-8 by cultured limbus epithelial cells. This is
consistent with previously reports, which described the UVB-induced IL-6 and IL-8
by other types of ocular surface epithelial cells, such as normal corneal
epithelial cells or pterygium epithelial cells[7-9].
The signal
pathways involved in UVB-induced production of IL-6 and IL-8 by corneal
epithelial cells, pterygium epithelial cells and keratinocytes include MAPK and
NF-κB pathways[7-9,20-21]. UVB may directly activate the NF-κB in the
cytoplasm. Activated NF-κB translocates to the nuclei and work as a
transcription factor for stimulating the expression of various pro-inflammatory
cytokines, including IL-6 and IL-8[21]. Alternatively,
UVB may first activate MAPK signal pathway. The activated MAPKs can translocate
into the nucleus and phosphorylates the target transcription factors, such as
NF-κB. Our study suggested that curcumin inhibited the UVB-induced expression
of IL-6 and IL-8 via MAPK and NF-κB signal pathways. This result is
consistent with the previous reports that UVB-induced production of IL-6 and
IL-8 is relevant to the activation of MAPK and NF-κB signal pathways in
epithelial cells from normal cornea or pterygium specimens and in keratinocytes[7-9,20-21].
The present
study revealed that curcumin at low and non-lethal dosages (5-20 μmol/L) does not
affect the constitutive secretion of IL-6 and IL-8 by limbus epithelial cells,
but curcumin at the same dosages can significantly inhibits UVB-induced
secretion of IL-6 and IL-8 in limbus epithelial cells. This results are
consistent with the results found in previous studies that curcumin can inhibit
UVB-induced production and secretion of IL-6 and IL-8 in keratinocytes[20-21].
Curcumin is a
natural anti-inflammatory compound with a long history of use, has been used as
a remedy for the treatment and prevention of inflammatory diseases[16-19]. Aggarwal et al[16] reported that more than 60 clinical trials had been
conducted for studying the efficacy and safety of curcumin. In addition to
these studies, another 35 clinical trials have evaluated the efficacy of
curcumin. Curcumin was found to be effective in the treatment of various
chronic inflammatory diseases including psoriasis, inflammatory bowel diseases
and different types of arthritis, such as rheumatoid arthritis and
osteoarthritis[16,18]. It has
even been demonstrated to be effective in the treatment of other severe diseases
such as Alzheimer's[22], cystic fibrosis[23], and AIDS[24]. Clinical
trials also demonstrated that curcumin is a safe, nontoxic remedy, can be given
orally and is quite safe and affordable[15-19].
The main
limitation of curcumin is its insolubility in aqueous systems. Various attempts
to enhance the bioavailability and efficacy of curcumin have been reported by
the encapsulation of curcumin into liposomes, phospholipid complexes or
nanoparticle. The primary aim of these studies is to achieve increased
solubilization of curcumin and, at the same time, to protect curcumin against
inactivation by hydrolysis[13,25].
Therefore, in the future, curcumin can be used not only as an oral
nutrition supplement, but also can be used locally, such as the eye drops for
the treatment of various ocular inflammatory diseases.
The present
study found that curcumin at low and safe dosages significantly inhibits the
UVB-induced secretion of IL-6 and IL-8 by cultured limbus epithelial cells; but
does not affect the constitutive secretion of IL-6 and IL-8. These results
suggest that curcumin may be a promising agent to be explored for the
prevention of progress of pterygium, and also may be used for the prevention of
recurrence of pterygium after its surgical excision.
ACKNOWLEDGEMENTS
Conflicts
of Interest: Chao SC, None; Hu DN, None; Roberts J, None; Shen X,
None; Lee CY, None; Nien CW, None; Lin HY, None.
REFERENCES
1 Solomon A, Li DQ, Lee SB, Tseng SC. Regulation of collagenase,
stromelysin, and urokinase-type plasminogen activator in primary pterygium body
fibroblasts by inflammatory cytokines. <ii>Invest Ophthalmol Vis
Sci</ii> 2000;41(8):2154-2163. [PubMed]
2 Di Girolamo N, Coroneo MT, Wakefield D. UVB-elicited induction
of MMP-1 expression in human ocular surface epithelial cells is mediated
through the ERK1/2 MAPK-dependent pathway. <ii>Invest Ophthalmol Vis Sci
</ii> 2003;44(11):4705-4714. [PubMed]
3 Dushku N, John MK, Schultz GS, Reid TW. Pterygia pathogenesis:
corneal invasion by matrix metalloproteinase expressing altered limbal
epithelial basal cells. <ii>Arch Ophthalmol </ii> 2001;119(5):
695-706. [PubMed]
4 Di Girolamo N, Chui J, Coroneo MT, Wakefield D. Pathogenesis of
pterygia: role of cytokines, growth factors, and matrix metalloproteinases.
<ii>Prog Retin Eye Res </ii> 2004;23(2):195-228. [PubMed]
5 Taylor HR, West SK, Rosenthal FS, Munoz B, Newland HS, Emmett
EA. Corneal changes associated with chronic UV irradiation. Arch Ophthalmol 1989;107(10):1481-1484. [CrossRef]
6 Sekelj S, Dekaris I, Kondza-Krstonijević E, Gabrić N, Predović J, Mitrović S.
Ultraviolet light and pterygium. <ii>Coll Antropol </ii>
2007;31(Suppl 1):45-47. [PubMed]
7 Di Girolamo N, Kumar RK, Coroneo MT, Wakefield D. UVB-mediated
induction of interleukin-6 and -8 in pterygia and cultured human pterygium
epithelial cells.<ii> Invest Ophthalmol Vis Sci </ii>
2002;43(11):3430-3437. [PubMed]
8 Di Girolamo N, Wakefield D, Coroneo MT. UVB-mediated induction
of cytokines and growth factors in pterygium epithelial cells involves cell
surface receptors and intracellular signaling. <ii>Invest Ophthalmol Vis
Sci</ii> 2006;47(6):2430-2437. [PubMed]
9 Nolan TM, Di Girolamo N, Sachdev NH, Hampartzoumian T, Coroneo
MT, Wakefield D. The role of ultraviolet irradiation and heparin-binding
epidermal growth factor-like growth factor in the pathogenesis of pterygium. Am J Pathol 2003;162(2):567-574. [CrossRef]
10 Notara M, Refaian N, Braun G, Steven P, Bock F, Cursiefen C. Short-term
UVB-irradiation leads to putative limbal stem cell damage and niche
cell-mediated upregulation of macrophage recruiting cytokines. <ii>Stem
Cell Res </ii> 2015;15(3):643-654. [PubMed]
11 Di Girolamo N, Coroneo M, Wakefield D. Epidermal growth factor
receptor signaling is partially responsible for the increased matrix
metalloproteinase-1 expression in ocular epithelial cells after UVB radiation. Am J Pathol 2005;167(2):489-503. [CrossRef]
12 Hu DN, Chen M, Zhang DY, Ye F, McCormick SA, Chan CC. Interleukin-1β
increases baseline expression and secretion of interleukin-6 by human uveal
melanocytes in vitro via p38 MAPK/NF-κB pathway. <ii>Invest Ophthalmol
Vis Sci </ii> 2011;52(6):3767-3774. [PubMed]
13 Hu DN, Bi M, Zhang DY, Ye F, McCormick SA, Chan CC.
Constitutive and LPS-induced expression of MCP-1 and IL-8 by human uveal
melanocytes in vitro and relevant signal pathways. <ii>Invest Ophthalmol
Vis Sci</ii> 2014;55(9):5760-5769. [PMC free article] [PubMed]
14 Kennedy M, Kim KH, Harten B, Brown J, Planck S, Meshul C, Edelhauser
H, Rosenbaum JT, Armstrong CA, Ansel JC. Ultraviolet irradiation induces the
production of multiple cytokines by human corneal cells. <ii>Invest
Ophthalmol Vis Sci</ii> 1997;38(12):2483-2491. [PubMed]
15 Aggarwal BB, Sung B. Pharmacological basis for the role of
curcumin in chronic diseases: an age-old spice with modern targets.<ii>
Trends Pharmacol Sci </ii>2009;30(2):85-94. [PubMed]
16 Aggarwal BB, Gupta SC, Sung B. Curcumin: an orally bioavailable
blocker of TNF and other pro-inflammatory biomarkers.<ii> Br J
Pharmacol</ii> 2013;169(8):1672-1692. [PMC free article] [PubMed]
17 Schaffer M, Schaffer PM, Bar-Sela G. An update on Curcuma as a
functional food in the control of cancer and inflammation. <ii>Curr Opin
Clin Nutr Metab Care</ii> 2015;18(6):605-611. [PubMed]
18 Shehzad A, Rehman G, Lee YS. Curcumin in inflammatory diseases. Bio Factors 2013;39(1):69-77. [CrossRef]
19 Chin KY. The spice for joint inflammation: anti-inflammatory role of
curcumin in treating osteoarthritis. <ii>Drug Des Devel Ther </ii>
2016;10:3029-3042. [PMC free article] [PubMed]
20 Cho JW, Park K, Kweon GR, Jang BC, Baek WK, Suh MH, Kim CW, Lee
KS, Suh SI. Curcumin inhibits the expression of COX-2 in UVB-irradiated human
keratinocytes (HaCaT) by inhibiting activation of AP-1: p38 MAP kinase and JNK
as potential upstream targets. <ii>Exp Mol Med</ii>
2005;37(3):186-192. [PubMed]
22 Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS,
Chen PP, Kayed R, Glabe CG, Frautschy SA, Cole GM. Curcumin inhibits formation
of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in
vivo. <ii>J Biol Chem </ii> 2005;280(7):5892-5901. [PubMed]
23 Egan ME, Pearson M, Weiner SA, Rajendran V, Rubin D,
Glöckner-Pagel J, Canny S, Du K, Lukacs GL, Caplan MJ. Curcumin, a major
constituent of turmeric, corrects cystic fibrosis defects.
<ii>Science</ii> 2004; 304(5670):600-602. [PubMed]
24 Sui Z, Salto R, Li J, Craik C, Ortiz de Montellano PR.
Inhibition of the HIV-1 and HIV-2 proteases by curcumin and curcumin boron
complexes. <ii>Bioorg Med Chem </ii> 1993;1(6):415-422. [CrossRef]
25 Nardo L, Maspero A, Selva M, Bondani M, Palmisano G, Ferrari E,
Saladini M. Excited state dynamics of bis-dehydroxycurcumin carboxylic acid, a
water-soluble derivative of the photosensitizer curcumin.<ii> J Phys Chem
A</ii> 2012;116(37):9321-9330. [PubMed]