
・Basic Research・Current Issue・ ・Achieve・ ・Search Articles・ ・Online Submission・ ・About IJO・ PMC
Scutellaria barbata attenuates diabetic retinopathy by
preventing retinal inflammation and the decreased expression of tight junction
protein
Xi-Yu Mei, Ling-Yu Zhou, Tian-Yu Zhang, Bin Lu, Li-Li Ji
The MOE Key
Laboratory for Standardization of Chinese Medicines, Shanghai Key Laboratory of
Complex Prescription, Institute of Chinese Materia Medica, Shanghai University
of Traditional Chinese Medicine, Shanghai 201203, China
Correspondence
to: Li-Li
Ji. The MOE Key Laboratory for Standardization of Chinese Medicines and
Shanghai Key Laboratory of Complex Prescription, Institute of Chinese Materia
Medica, Shanghai University of Traditional Chinese Medicine, 1200 Cailun Road,
Shanghai 201203, China. lichenyue1307@126.com
Received:
2016-11-16 Accepted:
2017-03-13
Abstract
AIM: To observe the
attenuation of ethanol extract of Herba Scutellaria barbata (SE) against
diabetic retinopathy (DR) and its engaged mechanism.
METHODS: C57BL/6J mice
were intraperitoneally injected with streptozotocin (STZ, 55 mg/kg) for 5
consecutive days to induce diabetes. The diabetic mice were orally given with
SE (100, 200 mg/kg) for 1mo at 1mo after STZ injection. Blood-retinal barrier
(BRB) breakdown was detected by using Evans blue permeation assay. Real-time
polymerase chain reaction (RT-PCR),
Western blot and immunofluorescence staining were used to detect mRNA and
protein expression. Enzyme-linked immunosorbent assay (ELISA) was used to
detect serum contents of tumor necrosis factor-α (TNF-α) and
interleukin (IL)-1β.
RESULTS: SE (100, 200
mg/kg) reversed the breakdown of BRB in STZ-induced diabetic mice. The
decreased expression of retinal claudin-1 and claudin-19, which are both tight
junction (TJ) proteins, was reversed by SE. SE decreased the increased serum
contents and retinal mRNA expression of TNF-α and IL-1β. SE also
decreased the increased retinal expression of intercellular cell adhesion
molecule-1 (ICAM-1). SE reduced the increased phosphorylation of nuclear factor
kappa B (NFκB) p65 and its
subsequent nuclear translocation in retinas from STZ-induced diabetic mice.
Results of Western blot and retinal immunofluorescence staining of ionized
calcium-binding adapter molecule 1 (Iba1) demonstrated that SE abrogated the
activation of microglia cells in STZ-induced diabetic mice.
CONCLUSION: SE attenuates
the development of DR by inhibiting retinal inflammation and restoring the
decreased expression of TJ proteins including claudin-1 and claudin-19.
KEYWORDS: scutellaria barbata; diabetic retinopathy;
tight junction; inflammation; nuclear factor kappa B; microglia
DOI:10.18240/ijo.2017.06.07
Citation: Mei XY, Zhou LY, Zhang TY, Lu B, Ji LL. Scutellaria
barbata attenuates diabetic retinopathy by preventing retinal inflammation
and the decreased expression of tight junction protein. Int J Ophthalmol 2017;10(6):870-877
Article Outline
INTRODUCTION
With the
elevating of living standards, the incidence of diabetes is increasing in both
developed and developing countries. Diabetic retinopathy (DR) is one of the
most common and serious microvascular complications of diabetes mellitus (DM),
and vison loss due to DR has become a major cause of blindness in adult[1-2]. The pathogenesis of DR is
generally divided into two stages including non-proliferative diabetic
retinopathy (NPDR) and proliferative diabetic retinopathy (PDR) according to
the international clinical DR disease severity scale[3].
In the early NPDR stage, an increase in vasopermeability due to blood-retinal
barrier (BRB) breakdown is a key factor for diabetic macular edema (DEM), which
will lead to considerable vision loss in diabetic patients[4-5]. Moreover, NPDR will further progress into PDR, and
retinal angiogenesis occurs in this later stage, which will cause vitreous
hemorrhage, traction retinal detachment, and finally vision loss[2].
It has been
reported that inflammation plays an important role in regulating the
development of DR, and DR is generally considered as an inflammatory disease[6]. A variety of reports demonstrated the increased adhesion
of leukocytes to retinal vessels in diabetic animals, and the levels of various
pro-inflammatory cytokines were increased in the retina and vitreous in
diabetes attributed to the activation of pro-inflammatory transcriptional
factors including nuclear factor kappa B (NFκB)[7-11]. Such increased leukostasis and the elevated
pro-inflammatory cytokines and other growth factors will contribute to the
breakdown of BRB, which is the early key event in the development of DR[7-9]. By using pharmacologic inhibitors
or anti-inflammatory agents to inhibit the production of inflammatory
mediators, some therapeutic approaches have been identified that obviously
attenuated the development of DR, especially the early stage of DR[11-12]. Recently, some traditional
Chinese medicines or formulas have been reported to attenuate DR by inhibiting
inflammation, such as Dang-Gui-Bu-Xue-Tang (an aqueous extract of Radix
astragali, Angelica sinensis and Panax notoginseng)[13], Rhodiola sachalinensis (Gaoshan Hongjingtian)[14], and Dendroboum chrysotoxum Lindl[15].
Herba
Scutellaria barbata (SE), named Ban-Zhi-Lian in Chinese, is the dried whole plant
of Scutellaria barbata D. Don. (Labiatae). It has been used as a drug
for clearing away heat and toxic materials, promoting blood circulation and
removing blood stasis, and reducing swelling and alleviating pain for thousands
of years in China. Modern pharmacological studies demonstrated that S.
barbata had a variety of activities including anti-cancer,
anti-angiogenesis, anti-inflammation, anti-complement, and antioxidant[16-20]. In this study, we aimed to
observe whether the ethanol extract of SE can attenuate NPDR in streptozotocin
(STZ)-induced diabetic mice by inhibiting retinal inflammation and restoring
the decreased expression of tight junction (TJ) proteins.
MATERIALS AND METHODS
Materials
Reagents Antibodies for 536Ser
phosphorylated NFκBp65, NFκBp65, Lamin B1 and β-actin were all purchased from
Cell Signaling Technology (Danvers, MA, USA). Antibody for Iba1 was bought from
GeneTax Inc. (Alton Parkway Irvine, CA, USA). Antibodies for claudin-1 and
claudin-19 were purchased from Santa Cruz (Santa Cruz, CA, USA). Antibody for
intercellular cell adhesion molecule-1 (ICAM-1) was purchased from Biobasic Inc
(Shanghai, China). Peroxidase-conjugated goat anti-rabbit immunoglobulin G
(IgG) (H+L) and anti-mouse IgG (H+L) were purchased from Jackson ImmunoResearch
(West Grove, PA, USA). Alexa Fluor 488 goat anti-Rabbit IgG were purchased from
BD Biosciences (Franklin Lakes, NJ, USA). NE-PER nuclear and cytoplasmic
extraction reagents, and Pierce BCA Protein Assay Kits were purchased from
ThermoFisher Scientific (Waltham, MA, USA). Enzyme-linked immunosorbent assay
(ELISA) kits were obtained from RapidBio (West Hills, CA, USA). Trizol reagent
and 4’,6-Diamidino-2-phenylindole (DAPI) were purchased from Life Technology
(Carlsbad, CA, USA). PrimeScriptRT Master Mix and SYBR Premix Ex TaqTM
were purchased from Takara (Shiga, Japan). Other reagents unless noted were
purchased from Sigma Chemical Co. (St. Louis, MO, USA).
Preparation
of Herba Scutellaria barbata
The
powder of SE was soaked in 80% ethanol for 2h at room temperature. The
macerated plant material was extracted under reflux for 2h three times. The
combined extraction was concentrated and dried under vacuum using a rotary
evaporator under reduced pressure.
Experimental
animals Specific pathogen free male
C57BL/6J mice (weight: 18-22 g) were purchased from Shanghai Laboratory Animal
Center of Chinese Academy of Science (Shanghai, China). The mice were fed with
a standard laboratory diet and given free access to tap water, living in a
controlled room temperature (22℃±1℃), humidity (65%±5%) with a 12:12-h
light/dark cycle. All animals have received humane care in compliance with the
institutional animal care guidelines approved by the Experimental Animal
Ethical Committee of Shanghai University of Traditional Chinese Medicine.
Methods
Treatment
of animals Fifty mice were
intraperitoneally injected (i.p.) with STZ (55 mg/kg) for 5 consecutive days,
while the other sixteen mice were i.p. with physiological saline and served as
control animals. The concentration of serum glucose was measured 7d after the
last injection, and the mice with high glucose concentration (>16.5 mmol/L)
were considered as diabetic mice. In this experiment, the glucose concentration
in 48 mice was >16.5 mmol/L, and those mice were randomly divided into three
groups: DM group (n=16), DM+SE (100 mg/kg; n=16), DM+SE (200
mg/kg; n=16), respectively. At 1mo after the injection of STZ, the mice
were orally given with SE (100, 200 mg/kg, intragastric administration) for 1
consecutive month. At 2mo after the injection of STZ, the mice were
anesthetized by sodium pentobarbital (30 mg/kg, i.p.), the blood samples were
taken from the abdominal aorta, and the eyes were removed immediately. The body
weight was monitored and the concentration of blood glucose was determined by
Glucometer (Accu-Check Performa Nano, Roche Diagnostics, Germany) during the
whole experimental process.
Evans blue
permeation assay BRB breakdown was
evaluated as described in our previously published papers[15,21]. In brief, mice were injected with 2% Evans blue (10
μL/g, i.p.) in phosphate buffered saline (PBS), and blood was extracted through
the left ventricle at 2h after injection. The mice were further perfused with
PBS to completely remove the Evans blue dye in vessels. Retinas were carefully
dissected and the weight was determined after thoroughly drying. Next, the
retinas were incubated in 120 μL formamide for 18h at 70℃ to extract Evans blue
dye. The extract was centrifuged twice at 10 000× g for 1h at 4℃, and the
absorbance was determined at 620 nm. The concentration of Evans blue dye in the
extracts was calculated using a standard curve of Evans blue dye in formamide
and then normalized to the dried retinal weight.
Real-time
polymerase chain reaction analysis Total RNA in retinas was
isolated by using Trizol reagent, and the RNA content was determined by
measuring the optical density at 260 nm. cDNA was synthesized according to the
instruction described in the kits. Real-time polymerase chain reaction (RT-PCR)
was performed by using kits, and the relative expression of target genes was
normalized to actin, analyzed by the 2-ΔΔCt method and given as
ratio compared with the control. The primer sequences used in this study are
shown in Table 1.
Table 1 The
list of primers for RT-PCR
|
Target |
Primer |
Sequence |
|
Tnf |
FP |
5’-AGGCACTCCCCCAAAAGAT-3’ |
|
RP |
5’-CAGTAGACAGAAGAGCGTGGTG-3’ |
|
|
Il1β |
FP |
5'-AGTTGACGGACCCCAAAAG-3' |
|
RP |
5'-CTTCTCCACAGCCACAATGA-3' |
|
|
Icam1 |
FP |
5'-CCGCTGTGCTTTGAGAACT-3' |
|
RP |
5'-GGTCCTTGCCTACTTGCTG-3' |
|
|
Actin |
FP |
5'-TACAGCTTCACCACCACAGC-3' |
|
RP |
5'-TCTCCAGGGAGGAAGAGGAT-3' |
|
|
Cldn1 |
FP |
5'-CAGAAGATGTGGATGGCTGTC-3' |
|
RP |
5'-GGGGTCAAGGGGTCATAGAA-3' |
|
|
Cldn5 |
FP |
5'-TTGGAAGGGGCTGTGGAT-3' |
|
RP |
5'-CGGTCAAGGTAACAAAGAGTGC-3' |
|
|
Cldn19 |
FP |
5'-GCAAACTCTACGATTCACTCCTG-3' |
|
RP |
5'-CCACGACACTGAGCACCAT-3' |
|
|
Ocln |
FP |
5'-TTCCTCTGACCTTGAGTGTGG-3' |
|
RP |
5'-CTCTTGCCCTTTCCTGCTTT-3' |
|
|
Tjp1 |
FP |
5’-CTCCAGGTGCTTCTCTTGCT-3’ |
|
RP |
5’-TATCTTCGGGTGGCTTCACT-3’ |
FP: Forward
primer; RP: Reverse primer.
Western-blot
analysis Cytosolic and nuclear
proteins in retinas were isolated as described in NE-PER nuclear and
cytoplasmic extraction kits. After centrifugation, protein concentration of the
resulting supernatants was determined. The protein concentration in each sample
was normalized to the equal protein concentration. The protein samples were
subjected to SDS-PAGE and then electrophoretically transferred onto an
immobilon-P PVDF membrane (Millipore). The membranes were incubated with
primary and secondary antibodies. Immunoblots were visualized using a chemiluminescent
reagent. The grey densities of the protein bands were normalized by using
β-actin or Lamin B1 density as an internal control, respectively.
Enzyme-linked
immunosorbent assay analysis The whole blood was
centrifuged at 3000 rpm, 4℃ for 15min, and serum was collected for ELISA
analysis as described in kits.
Immunofluorescence
staining Paraffin-embedded sections
of retinas (5 mm) were de-paraffinized in xylene, and re-hydrated in an ethanol
gradient with distilled water. Retinas were incubated with 5% bovine serum
albumin to minimize non-specific binding after endogenous peroxidase activity
was quenched. After rinsing three times, retinas were incubated with Iba1
antibody at 4℃ overnight, and further incubated with Alexa fluor 488 goat
anti-rabbit IgG (H+L) antibody at room temperature for 1h. After rinsing three
times again, retinas were incubated with DAPI for 10min, and images were
captured under an inverted microscope (IX81, Olympus, Japan).
High
performance liquid chromatography analysis
Analysis
was performed on a prominence high performance liquid chromatography instrument
(HPLC; Agilent) equipped with auto-sampler, quaternary pump, column heater
compartment and DAD with an on-line degasser. The sample was separated on a
Sepax HP-C18 column (4.6×250 mm, 5 μm). The mobile phase consisted of methanol,
water and ethylic acid (v/v/v =35:61:4). The flow rate was 1.0 mL/min, and
column temperature was set at 25℃. The DAD detector monitored signals between
190 nm and 400 nm, and the on-line UV spectra were recorded at 335 nm for
scutellarin.
Statistical
Analysis Data were expressed as
means±standard error of the mean (SEM). The significance of differences between
groups was evaluated by one-way ANOVA with LSD post hoc test, and P<0.05
was considered as statistically significant differences.
RESULTS
Measurement
of Body Weight and Blood Glucose Concentration As shown in Figure 1A, the body
weight of diabetic mice was lower than that of normal control mice (P<0.001).
However, there was no obvious alteration in the body weight of mice after SE
treatment. Next, Figure 1B showed that blood glucose concentration in diabetic
mice was higher than that in normal control mice (P<0.001). Also, SE
had no much effect on blood glucose concentration in diabetic mice.
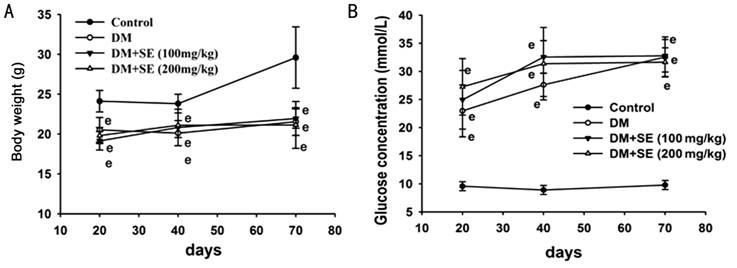
Figure 1
Analysis of serum glucose level and body weight A: Body weight; B: Serum glucose
level. n=16, eP<0.001 compared to control.
Herba
Scutellaria Barbata Attenuated Blood-retinal Barrier Breakdown in Diabetic
Mice As shown in Figure 2A, the increased
leakage of Evans blue dye was observed in retinas from STZ-induced diabetic
mice (P<0.001), indicating the increased retinal vessel leakage.
After diabetic mice were treated with SE (100, 200 mg/kg), such increased
retinal vessel leakage was almost totally abrogated (P<0.001).
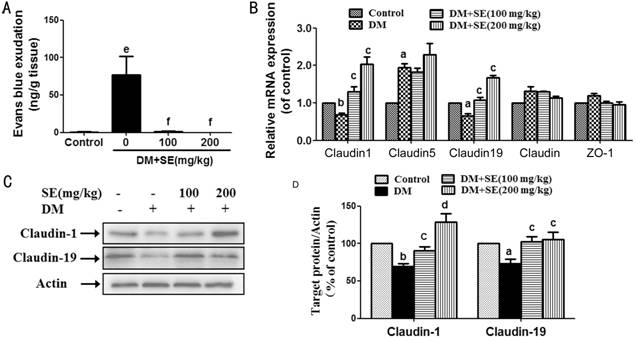
Figure 2 SE
attenuated BRB breakdown and restored the decreased expression of claudin-1 and
claudin-19 in retinas from STZ-induced diabetic mice A: BRB breakdown is detected by Evans
blue dye leakage assay (n=6); B: Retinal mRNA expression of occludin,
ZO-1, claudin-1, claudin-5 and claudin-19 (n=3-5); C: Retinal protein
expression of claudin-1 and claudin-19, representative blots for claudin-1,
claudin-19 and actin, and the results represent at least three independent
experiments; D: The quantitative densitometric analysis of claudin-1 and
claudin-19, and the results are presented as percentage of control (n=4-5).
aP<0.05, bP<0.01, eP<0.001
compared to control; cP<0.05, dP<0.01,
fP<0.001 compared to DM.
Herba
Scutellaria Barbata Reversed the Decreased Expression of Tight Junction
Proteins As shown in Figure 2B,
retinal mRNA expression of claudin-1 and claudin-19 was decreased in
STZ-induced diabetic mice (P<0.05, P<0.01), whereas SE
(100, 200 mg/kg) reversed such decrease (P<0.05). Retinal mRNA
expression of claudin-5 was weakly increased in STZ-induced diabetic mice (P<0.05),
but there was no alteration after SE (100, 200 mg/kg) treatment. Retinal mRNA
expression of zonula occludens-1 (ZO-1) and occludin was not changed in
STZ-induced diabetic mice with or without SE treatment.
Next, the
results of Western-blot showed that the protein expression of claudin-1 and
claudin-19 was decreased in retinas from STZ-induced diabetic mice (P<0.05,
P<0.01), whereas SE (100, 200 mg/kg) reversed the decreased retinal
protein expression of claudin-1 and claudin-19 (P<0.05, P<0.01)
(Figure 2C, 2D).
Herba
Scutellaria Barbata Reduced the Increased Expression of Intercellular Cell
Adhesion Molecule-1, Tumor Necrosis Factor-α and Interleukin-1β As shown in Figure 3A, retinal mRNA
expression of ICAM-1, tumor necrosis factor-α (TNF-α) and interleukin (IL)-1β
was increased in STZ-induced diabetic mice (P<0.05, P<0.01).
However, SE (100, 200 mg/kg) reduced the increased retinal mRNA expression of
IL-1β and ICAM-1, and SE (200 mg/kg) reduced the increased retinal mRNA
expression of TNF-α in diabetic mice (P<0.05) (Figure 3A). In
addition, SE (100, 200 mg/kg) reduced the elevated serum content of IL-1β, and SE
(100 mg/kg) reduced the elevated serum content of TNF-α in STZ-induced diabetic
mice (P<0.01, P<0.001) (Figure 3B). SE (100, 200 mg/kg)
also reduced the increased retinal ICAM-1 protein expression in STZ-induced
diabetic mice (P<0.05) (Figure 3C, 3D).
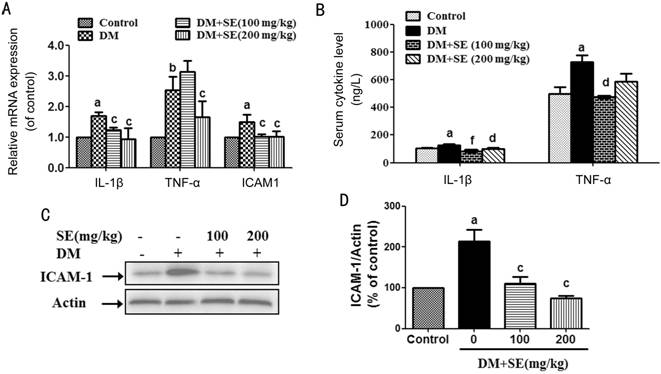
Figure 3 SE
reduced the increased ICAM-1, TNF-α and IL-1β expression in STZ-induced
diabetic mice A: Retinal mRNA expression
of TNF-α, IL-1β, and ICAM-1 (n=3-4); B: Serum contents of TNF-α and
IL-1β (n=6); C: Retinal protein expression of ICAM-1, representative
blots for ICAM-1 and actin, and the results represent four independent
experiments; D: The quantitative densitometric analysis of ICAM-1, and the
results are presented as percentage of control (n=4). aP<0.05,
bP<0.01 compared to control; cP<0.05,
dP<0.01, fP<0.001 compared to DM.
Herba
Scutellaria Barbata Abrogated Retinal Nuclear Factor Kappa B Activation in
Diabetic Mice As shown in Figure 4A, 4B,
retinal expression of phosphorylated NFκBp65 was increased in STZ-induced
diabetic mice, whereas SE (200 mg/kg) reduced such increase (P<0.001).
The expression of nuclear NFκBp65 was increased in retinas from STZ-induced
diabetic mice (P<0.001), but there was no significant change in the
expression of cytosolic NFκBp65 (Figure 4A, 4C). Further, SE (100, 200 mg/kg)
reduced the increased nuclear expression of NFκBp65 in STZ-induced diabetic
mice (P<0.05, P<0.001) (Figure 4A, 4C).
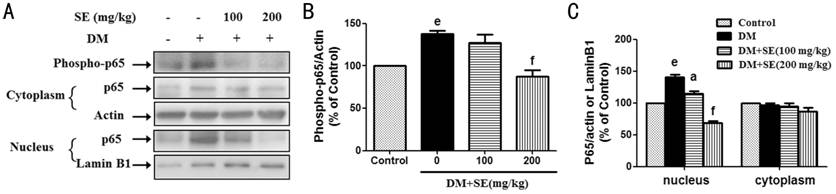
Figure 4 SE
abrogated the activation of NFκB signaling pathway in retinas from STZ-induced
diabetic mice A: SE reduced the
increased expression of phosphorylated NFκBp65 and its subsequent nuclear
translocation, representative blots for phosphorylated NFκBp65, nuclear and
cytosolic NFκBp65, actin and Lamin B1, and the results represent at least three
independent experiments; B: The quantitative densitometric analysis of
phosphorylated NFκBp65, and the results are presented as percentage of control
(n=3); C: The quantitative densitometric analysis of nuclear and
cytosolic NFκBp65, and the results are presented as percentage of control (n=3-4).
eP<0.001 compared to control; aP<0.05,
fP<0.001 compared to DM.
Herba
Scutellaria Barbata Inhibited the Activation of Microglia Cells in Diabetic
Mice Ionized calcium-binding adapter
molecule 1 (Iba1) is an often used biomarker for microglia[22].
As shown in Figure 5A, 5B, Iba1 expression was increased in retinas from
STZ-induced diabetic mice, whereas SE (200 mg/kg) reduced such increase (P<0.001).
Next, we observed the expression of Iba1 in retinas from STZ-induced diabetic
mice by using Iba1 immunofluorescence staining assay. As shown in Figure 5C,
the number of Iba1-positive microglia cells was increased in ganglion cell
layer (GCL) and inner plexiform layer (IPL) in retinas from STZ-induced
diabetic mice than from normal control mice. Moreover, such increase was
reduced in SE (100, 200 mg/kg)-treated mice.

Figure 5 SE
abrogated the activation of retinal microglia cells in STZ-induced diabetic
mice A: SE reduced the elevated protein
expression of Iba1, representative blots for Iba1 and actin, and the results
represent three independent experiments; B: The quantitative densitometric
analysis of Iba1, and the results are presented as percentage of control (n=3);
C: Retinal expression of Iba1 in STZ-induced diabetic mice, the representative
pictures of retinal immunofluorescence staining of Iba1 and DAPI, and also the
merge of Iba1- and DAPI-stained images are shown at the left (scale bars: 20
mm), the enlarged representative pictures of retinal Iba1-stained images are
shown at the right (scale bars: 5 mm), red arrows indicate microglia cells.
eP<0.001 compared to control; fP<0.001
compared to DM.
High
Performance Liquid Chromatography Analysis of Herba Scutellaria Barbata Scutellarin is the main compound in
SE, and it is also the chemical marker used by the Chinese pharmacopoeia for
evaluating the quality of SE[23]. The chemical
structure of scutellarin was shown in Figure 6A. The HPLC chromatograms
and calibration curve of scutellarin were shown in Figure 6B-6D, and the HPLC
results demonstrated that the amount of scutellarin in SE was 4.09%.
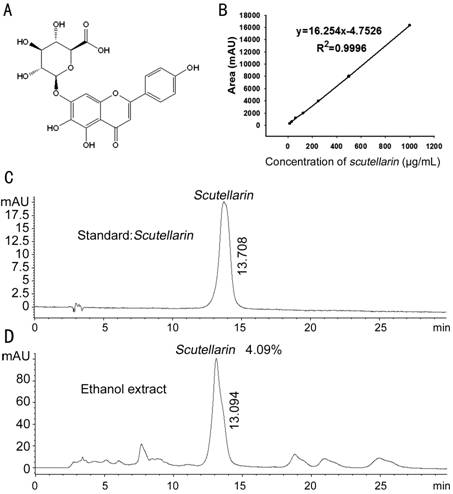
Figure 6
HPLC analysis of the content of scutellarin in SE A: The chemical structure of scutellarin;
B: The calibration curve of scutellarin; C: HPLC chromatogram of scutellarin;
D: HPLC chromatogram of scutellarin in SE.
DISCUSSION
The increased
retinal blood vessel permeability due to the breakdown of BRB is the hallmark
for NPDR, which is the early stage of DR[4-5].
In this study, we found that SE reduced the increased BRB leakage in
STZ-induced diabetic mice. This is the first study demonstrated that SE had
protection against NPDR, which indicates the potential application of SE for
the treatment of DR.
The BRB is div
ided into an inner and an outer BRB. The inner BRB is composed by TJ between
retinal capillary endothelial cells and the outer BRB is formed by TJ between
retinal pigment epithelial cells[4,24].
Both inner and outer BRB TJ proteins are mainly composed of occludins,
claudins, ZO proteins, and junctional adhesion molecules (JAMs)[25]. ZO-1, occludin, claudin-1, claudin-5 and claudin-19
are the main reported BRB TJ proteins, and they are all reported to be critical
for maintaining the integrity of BRB[26]. In this
study, we found that the mRNA and protein expression of claudin-1 and
claudin-19 was both reduced in retinas from STZ-induced diabetic mice, and such
decrease was reversed by SE. The results imply that SE restored BRB dysfunction
by up-regulating the expression of claudin-1 and claudin-19. However, there was
no alternation in the mRNA expression of occludin and ZO-1, and retinal mRNA
expression of claudin-5 was weakly increased in STZ-induced diabetic mice. A
variety of reports demonstrated the reduced expression of occludin and
claudin-5 in retinas from STZ-induced diabetic rats[27-29]. As for ZO-1, some reports showed the decreased
expression of retinal ZO-1[27-28],
whereas others demonstrated that there was no alteration in retinal ZO-1
expression in STZ-induced diabetic rats[29].
However, we got the different results in STZ-induced diabetic mice in this
study. We think that different species applied in this experiment and the
duration of maintaining diabetes may be the reason for acquiring such different
results.
The increased
interaction between leukocytes and retinal endothelial cells plays an important
role in the early stage of DR, which initiates retinal inflammation and leads
to the increased leukostasis and permeability of BRB[30].
ICAM-1 plays a critical role in regulating the adhesion of leukocytes to
endothelial cells, and the increased ICAM-1 expression has been reported to be
related with the development of DR[31]. TNF-α and
IL-1β, two well-known pro-inflammatory cytokines, have also been found to be
implicated in the pathogenesis of DR[32-33].
In this study, we found that SE decreased retinal expression of ICAM-1, TNF-α
and IL-1β, and also reduced the increased serum TNF-α and IL-1β levels in
STZ-induced diabetic mice. All those effects will contribute to SE-induced the
reduction in retinal leukostasis and inflammation during the development of
DR.
Transcription
factor NFκB is a key factor involved in inflammation by regulating the
expression of pro-inflammatory cytokines such as TNF-α and IL-1β, and ICAM-1[27,34]. Next, we observed the effects
of SE on NFκB activation in retinas from STZ-induced diabetic mice. The results
showed that SE reduced the increased phosphorylation of NFκBp65 and its
subsequent nuclear translocation, indicating that SE attenuated retinal
inflammation in STZ-induced diabetic mice by inhibiting the activation of NFκB
signaling pathway.
Previous
studies have already demonstrated the activation of retinal microglia cells and
its important roles in regulating retinal inflammation during the development
of DR[35]. Iba1 is an often used biomarker for
microglia[22]. In this study, the results of Western-blot and Iba1
immunofluorescence staining showed that SE reduced the increased activation of
microglia cells in STZ-induced diabetic mice, which may contribute to its
attenuation on retinal inflammation.
In this study,
we can see that SE (200 mg/kg) had better effect than SE (100 mg/kg) in all the
experiments except in the result of serum TNF-α content. SE (100 mg/kg)
restored the decreased expression of TJ proteins and weakly inhibited retinal
inflammation, but did not reduce the increased retinal Iba1 expression. All
these results evidenced that SE ameliorated STZ-induced DR in mice.
In conclusion,
this study demonstrated that SE prevented BRB breakdown by inhibiting retinal
inflammation through abrogating NFκB signaling pathway, restoring the decreased
expression of TJ proteins including claudin-1 and claudin-19, and reducing the
activation of retinal microglia cells. This study indicates the huge potential
application of SE in the treatment of DR.
ACKNOWLEDGEMENTS
Foundations:
Supported
by the National Natural Science Foundation of China (No.81173517; No.81322053).
Conflicts
of Interest: Mei XY, None; Zhou LY, None; Zhang TY, None; Lu
B, None; Ji LL, None.
REFERENCES
1 Fantes RJ, Durairaj VD, Oliver SC. Diabetic
retinopathy: an update on treatment. <ii>Am J Med
</ii>2010;123(3):213-216. [PubMed]
2 Cheung N, Mitchell P, Wong TY. Diabetic retinopathy.
<ii>Lancet</ii> 2010; 376(9735):124-136.[CrossRef]
3 Wilkinson CP, Ferris III FL, Klein RE, Lee PP,
Agardh CD, Davis M, Dills D, Kampik A, Pararajasegaram R, Verdaguer JT; Global
Diabetic retinopathy Project Group. Proposed international clinical diabetic
retinopathy and diabetic macular edema disease severity scales. <ii>Ophthalmology
</ii> 2003;110(9):1677-1682.[CrossRef]
4 Cunha-Vaz J, Faria de Abreu JR, Campos AJ. Early
breakdown of the blood-retinal barrier in diabetes. <ii>Br J Ophthalmol
</ii>1975;59(11):649-656.[CrossRef]
5 Do carmo A, Ramos P, Reis A, Proenca R, Cunha-vaz
JG. Breakdown of the inner and outer blood retinal barrier in
streptozotocin-induced diabetes. <ii>Exp Eye Res
</ii>1998;67(5):569-575. [PubMed]
6 Semeraro F, Cancarini A, dell’Omo R, Rezzola S,
Romano MR, Costagliola C. Diabetic retinopathy: vascular and inflammatory
disease.<ii> J Diabetes Res </ii> 2015;2015:582060. [PMC free article] [PubMed]
7 Leal EC, Manivannan A, Hosoya K, Terasaki T,
Cunha-Vaz J, Ambrosio AF, Forrester JV. Inducible nitric oxide synthase isoform
is a key mediator of leukostasis and blood-retinal barrier breakdown in
diabetic retinopathy. <ii>Invest Ophthalmol Vis Sci
</ii>2007;48(11):5257-5265. [PubMed]
8 Barber AJ, Antonetti DA, Gardner TW. Altered
expression of retinal occludin and glial fibrillary acidic protein in
experimental diabetes. The Penn State Retina Research Group. <ii>Invest
Ophthalmol Vis Sci </ii>2000;41(11):3561-3568. [PubMed]
9 Joussen AM, Murata T, Tsujikawa A, Kirchhof B,
Bursell SE, Adamis AP. Leukocyte-mediated endothelial cell injury and death in
the diabetic retina. <ii>Am J Pathol </ii>2001;158(1):147-152.[CrossRef]
10 Joussen AM, Poulaki V, Le ML, Koizumi K, Esser C,
Janicki H. A central role for inflammation in the pathogenesis of diabetic
retinopathy. <ii>FASEB J </ii>2004;18(12):1450-1452.[CrossRef]
11 Kern TS. Contributions of inflammatory processes to
the development of the early stages of diabetic retinopathy. <ii>Exp
Diabetes Res</ii> 2007;2007: 95103.[CrossRef]
12 Gologorsky D, Thanos A, Vavvas D. Therapeutic
interventions against inflammatory and angiogenic mediators in proliferative
diabetic retinopathy. <ii>Mediators Inflamm </ii>2012;2012:629452.
[PMC free article] [PubMed]
<no>13 Gao D, Guo Y, Li X, Li X, Li Z, Xue M, Ou
Z, Liu M, Yang M, Liu S, Yang S. An aqueous extract of Radix Astragali,
Angelica sinensis, and Panax notoginseng is effective in preventing diabetic
retinopathy. <ii>Evid Based Complement Alternat Med</ii>
2013;2013:578165.</no>
<no>14 Zhao HS, Shi XY, Wei WB, Wang NL. Effect
of the regimen of Gaoshan Hongjingtian on the mechanism of poly (ADP-ribose)
polymerase regulation of nuclear factor kappa B in the experimental diabetic
retinopathy. <ii>Chin Med J (Engl)
</ii>2013;126(9):1693-1699.</no>
<no>15 Yu ZY, Gong CY, Lu B, Yang L, Sheng YC,
Ji LL, Wang ZT. Dendrobium chrysotoxum Lindl. alleviates diabetic retinopathy
by preventing retinal inflammation and tight junction protein decrease.
<ii>J Diabetes Res</ii> 2015;2015:518317.</no>
16 Jiang Q, Li Q, Chen H, Shen A, Cai Q, Lin J, Peng
J. Scutellaria barbata D. Don inhibits growth and induces apoptosis by
suppressing IL-6 inducible STAT3 pathway activation in human colorectal cancer
cells. <ii>Exp Ther Med </ii>2015;10(4):1602-1608.[CrossRef]
17 Tao GY, Balunas MJ. Current therapeutic role and
medicinal potential of Scutellaria barbata in traditional chinese medicine and
western research. <ii>J Ethnopharmacol</ii> 2016;182:170-180. [PubMed]
18 Dai ZJ, Lu WF, Gao J, Kang HF, Ma YG, Zhang SQ,
Diao Y, Lin S, Wang XJ, Wu WY. Anti-angiogenic effect of the total flavonoids
in Scutellaria barbata D. Don. <ii>BMC Complement Altern Med
</ii>2013;13:150. [PMC free article] [PubMed]
19 Ye CL, Huang Q. Extraction of polysaccharides from
herbal Scutellaria barbata D. Don. (Ban-Zhi-Lian) and their antioxidant
activity. <ii>Carbohydr Polym </ii> 2012;89(4):1131-1137. [PubMed]
20 Wu YF, Chen DF. Anti-complementary effect of
polysaccharide B3-PS1 in Herba Scutellariae Barbatae (Scutellaria barbata).
<ii>Immunopharmacol Immunotoxicol </ii>2009;31(4):696-701. [PubMed]
21 Yu ZY, Lu B, Sheng YC, Zhou LY, Ji LL, Wang ZT.
Andrographolide ameliorates diabetic retinopathy by inhibiting retinal
angiogenesis and inflammation. <ii>Biochim Biophys Acta
</ii>2015;1850(4):824-831. [PubMed]
22 Ahmed Z, Shaw G, Sharma VP, Yang C, McGowan E,
Dickson DW. Actin-binding proteins coronin-1a and IBA-1 are effective
microglial markers for immunohistochemistry. <ii>J Histochem Cytochem
</ii>2007;55(7):687-700. [PubMed]
<no>23 Chinese Pharmacopoeia Commission.
Pharmacopeia of the people's republic of China (2015 version). Beijing: The
medicine science and technology press of China 2015;pp118-119.</no>
24 Cunha-Vaz JG. The blood-retinal barriers.
<ii>Doc Ophthalmol </ii> 1976; 41(2):287-327.[CrossRef]
25 Cunha-Vaz J, Bernardes R, Lobo C. Blood-retinal
barrier. <ii>Eur J Opthalmol </ii> 2011;21(Suppl 6):S3-S9. [PubMed]
26 Erickson KK, Sundstrom JM, Antonetti DA. Vascular
permeability in ocular disease and the role of tight junctions.
<ii>Angiogenesis</ii> 2007;10(2): 103-117. [PubMed]
27 Leal EC, Martins J, Voabil P, Liberal J, Chiavaroli
C, Bauer J, Cunha-Vaz J, Ambrosio AF. Calcium dobesilate inhibits the
alternations in tight junction proteins and leukocyte adhesion to retinal
endothelial cells induced by diabetes. <ii>Diabetes</ii>
2010;59(10):2637-2645. [PMC free article] [PubMed]
28 Fan LL, Yan H. FTY720 attenuates retinal
inflammation and protects blood-retinal barrier in diabetic rats.
<ii>Invest Ophthalmol Vis Sci</ii> 2016;57(3):1254-1263. [PubMed]
29 Tzeng TF, Hong TY, Tzeng YC, Liou SS, Liu IM.
Consumption of polyphenol-rich Zingiber Zerumbet Rhizome extracts protects
against the breakdown of the Blood-retinal barrier and retinal inflammation
induced by diabetes. <ii>Nutrients</ii> 2015;7(9):7821-7841. [PMC free article] [PubMed]
30 Patel N. Targeting leukostasis for the treatment of
early diabetic retinopathy. <ii>Cardiovasc Hematol Disord Drug
Targets</ii> 2009;9(3):
222-229.[CrossRef]
31 Ugurlu N, Gerceker S, Yulek F, Ugurlu B, Sari C,
Baran P, Cagil N. The levels of the circulating cellular adhesion molecules
ICAM-1, VCAM-1 and endothelin-1 and the flow-mediated vasodilation values in
patients with type 1 diabetes mellitus with early-stage diabetic retinopathy.
<ii>Intern Med </ii> 2013;52(19):2173-2178.[CrossRef]
32 Capitao M, Soares R. Angiogenesis and inflammation
crosstalk in diabetic retinopathy. <ii>J Cell Biochem</ii>
2016;117(11):2443-2453. [PubMed]
33 Kowluru RA, Odenbach S. Role of interleukin-1beta
in the pathogenesis of diabetic retinopathy. <ii>Br J Ophthalmol
</ii>2004;88(10): 1343-1347. [PMC free article] [PubMed]
34 Baeuerle PA, Henkel T. Function and activation of
NF-kappa B in the immune system. <ii>Ann Rev Immunol
</ii>1994;12:141-179. [PubMed]
35 Grigsby JG, Cardona SM, Pouw CE, Muniz A, Mendiola
AS, Tsin AT, Allen DM, Cardona AE. The role of microglia in diabetic
retinopathy. <ii>J Ophthalmol</ii> 2014;2014:705783. [PMC free article] [PubMed]