
・Clinical Research・ Current
Issue IF in JCR CiteScore ・Submission・ In Press Recent Accepted PMC RSS
Citation: Brennan N, Dehabadi MH,
Nair S, Quartilho A, Bunce C, Reekie I, Obikpo
R. Efficacy and safety of bimatoprost in glaucoma and ocular hypertension
in non-responder patients. Int J Ophthalmol 2017;10(8):1251-1254
Efficacy and safety of bimatoprost in glaucoma and
ocular hypertension in non-responder patients
Nicholas Brennan 1, Mohammad H Dehabadi
2, Sandhya Nair 3, Ana Quartilho 4, Catey
Bunce 4, Ian Reekie 3, Raal Obikpo 3
1Moorfields Eye Hospital NHS Foundation Trust, London EC1V 2PD, UK
2London North West Healthcare NHS Trust, London HA1 3UJ, UK
3North Middlesex University Hospital NHS Trust, London N18 1QX, UK
4NIHR Biomedical Research Centre, Moorfields Eye Hospital NHS Foundation
Trust and UCL Institute of Ophthalmology, London EC1V 2PD, UK
Correspondence to: Ian Reekie. North
Middlesex University Hospital, Sterling Way, London N18 1QX, UK.
ian.reekie@cantab.net
Received:
2016-10-27
Accepted: 2017-02-13
Abstract
AIM: To
establish the efficacy and safety of bimatoprost 0.03% monotherapy in glaucoma
and ocular hypertension (OHT) patients with inadequate intraocular pressure
(IOP)on current therapy.
METHODS: Pre-
and post-switch IOPs were analyzed for 59 consecutive patients who were
switched from current therapy to bimatoprost monotherapy between 2011-2015.
Demographic information, diagnosis, and any adverse events were recorded.
Change in IOP post-pre switch was analyzed using a 2-sided Student's paired t-test
at the 5% significance level.
RESULTS: There
was a statistically significant mean reduction in IOP at the first follow up
visit, which was maintained at subsequent follow up visits for patients
regardless of diagnosis, or pre-switch treatment (P<0.001). Subgroup
analysis also demonstrated a statistically significant mean reduction in IOP
when looking at OHT patients only, as well as patients with any diagnosis
switched from latanoprost monotherapy to bimatoprost monotherapy (P<0.001).
CONCLUSION: This
is the largest independent data set which supports switching glaucoma patients
with poor response to current treatment onto bimatoprost monotherapy before
considering other adjuvant medical or more invasive therapy.
KEYWORDS: glaucoma;
ocular hypertension; bimatoprost; latanoprost
DOI:10.18240/ijo.2017.08.11
Citation: Brennan N, Dehabadi MH, Nair S,
Quartilho A, Bunce C, Reekie I, Obikpo R.
Efficacy and safety of bimatoprost in glaucoma and ocular hypertension in
non-responder patients. Int J Ophthalmol 2017;10(8):1251-1254
INTRODUCTION
Glaucoma
is a complex neurodegenerative condition estimated to effect 64.3 million
people worldwide in 2013, a number that is predicted to rise to 111.8 million
by 2040[1]. The only modifiable risk factor in the
treatment of glaucoma to date is the reduction of intraocular pressure (IOP)[2]. Prostaglandin analogues (PGAs; bimatoprost,
latanoprost, Travaprost) are commonly the first line agents used to lower IOP
in primary open angle glaucoma (POAG), and ocular hypertension (OHT)[3]. Although the precise mechanism of action of these
drugs is not known, it is widely accepted that they act to increase aqueous
outflow via two pathways; uveoscleral outflow increase by extracellular
matrix remodeling[4], and trabecular outflow
increase[5].
Meta-analyses
have shown bimatoprost 0.03% to be equivalent in its IOP lowering efficacy when
compared to Travaprost, or Latanoprost[6]. In
recent years however, there has been a small, but growing body of Allergan
sponsored literature[7-14], as
well as independent studies[15-18]
supporting the use of Bimatoprost in patients with OHT, normal tension glaucoma
(NTG), and POAG who are deemed ‘non-responders’ on their current treatment. The
largest independent study currently in the literature consisted of 46 patients
with POAG or OHT and found no significant benefit from a switch to bimatoprost
monotherapy[17].
We
present an independent observational study of glaucoma and OHT patients with
inadequate IOP control on current therapy that were switched to bimatoprost
0.03% monotherapy.
SUBJECTS AND METHODS
Consecutive
‘non-responder’ patients with IOPs above their target IOP on current treatment
were prospectively identified by the lead glaucoma consultant between July 2011
and Jan 2015. These patients were switched from their current IOP lowering
agent to bimatoprost 0.03% monotherapy only.
Data
collection was carried out retrospectively, through case note review and
clinical electronic data base searches. Information on patient demographics,
diagnosis, pre-switch treatment, pre-switch IOP, IOP at all available
post-switch clinics, as well as any adverse events were recorded.
To
be included patients required a minimum of one follow up visit; documentation
of pre- and post-switch IOP recordings, pre-switch therapy and documentation of
any adverse events. Any participants with active ocular disease except glaucoma
or receiving ocular treatment which may affect the IOP were excluded. The
structure of the study is shown in Figure 1.
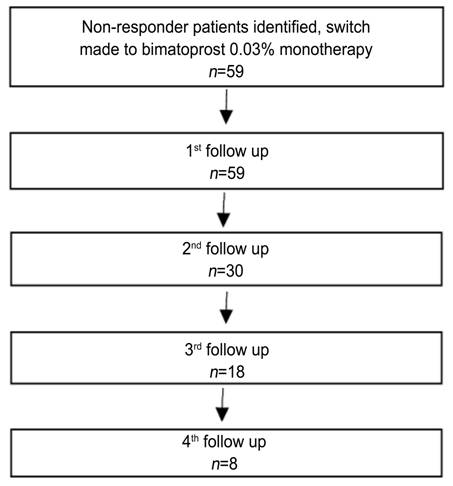
Figure
1 Study structure.
This
study was approved by the institutional review board and followed the
regulations of the Personal Information Protection and Electronic Documents Act
and the Good Clinical Practice Guidelines of the Declaration of Helsinki.
Change
in IOP post-pre switch was analyzed using a 2-sided Student's paired t-test
at the 5% significance level for left eye and then for right eye (to assess
consistency).
RESULTS
Adequate
follow up data for analysis was obtained for a total of 59 consecutive patients
following the clinical records search. Patient demographics are detailed in
Table 1. The majority of patients had OHT as their working diagnosis, with over
half on monotherapy with latanoprost before being switched to bimatoprost. The
mean pre-switch IOP for all study patients regardless of diagnosis was 23.4 mm
Hg.
Table
1 Baseline characteristics of study patients
n (%)
|
Characteristics |
Values |
|
Age
(a) Mean (range) |
64 (42-88) |
|
Sex M F |
31 (53) 28 (47) |
|
Race Caucasian Black Asian |
23 (39) 24 (41) 12 (20) |
|
Diagnosis OHT POAG NTG PAC-OHT PACG |
43 (73) 6 (10) 4 (7) 5 (8) 1 (2) |
|
IOP-lowering
Rx Latanoprost Travoprost Travoprost/Timolol
combination drop Latanoprost+Dorzolamide/Timolol
combination drop Dorzolamide+Travoprost/Timolol
combination drop Brinzolamide+Bimatoprost/Timolol
combination drop Latanoprost/Timolol
combination drop |
39 (66) 11 (19) 3 (5) 2 (3) 2 (3) 1 (2) 1 (2) |
|
Mean
Pre-switch IOP (mm Hg) Right eye Left eye |
23.2±4.4 23.3±3.7 |
Intraocular
Pressure Effects of Bimatoprost Monotherapy at First Follow up The average
time between switch to bimatoprost and the first follow up appointment was
104±44d. The mean reduction in IOP from pre-switch IOP at this time point for
right eyes was: -4.24 mm Hg; 95%CI (-5.49 to -2.1); P<0.001 (n=58),
and the mean reduction in IOP at this time point for left eyes was: -4.42 mm
Hg; 95%CI (-5.4 to -2.45); P<0.001 (n=59).
At
the first follow up visit, 16 (27%) of patients were deemed to have
unsatisfactory IOP, and were either switched to other therapy, or listed for
selective laser trabeculoplasty. The remaining 33 (55.9%) patients remained on
bimatoprost monotherapy, as their IOP was deemed satisfactory.
Subgroup
Analysis
Ocular
hypertension patients switched to bimatoprost The mean
reduction in IOP for OHT patients at first follow up appointment for right eyes
(n=47) was -4.11 mm Hg; 95%CI (-5.62 to -2.59); P<0.001. The
mean reduction in IOP for OHT patients at first follow up appointment for left
eyes (n=48) was -4.52 mm Hg; 95%CI (-5.67 to -3.37); P<0.001.
Patients
switched from latanoprost monotherapy to bimatoprost monotherapy For patients
with any diagnosis, switched from latanoprost monotherapy to bimatoprost
monotherapy (n=37), the mean reduction in IOP at first follow up
appointment was -5.27 mm Hg; 95%CI (-6.87 to -3.67); P<0.001 for
right eyes, and -5.27 mm Hg; 95%CI (-6.56 to -3.98); P<0.001 for left
eyes.
Intraocular
Pressure Effects of Bimatoprost Monotherapy at Second Follow up Second
follow up appointment data were available for 30 patients. The average time
from switch to bimatoprost to second follow up appointment was 320±109d. The
mean reduction in IOP from pre-switch IOP at this time point for right eyes
was: -6.31 mm Hg; 95%CI (-8.58 to -4.04); P<0.001 (n=30), and
the mean reduction in IOP at this time point for left eyes was: -7.95 mm Hg; 95%CI
(-8.75 to -5.25); P<0.001 (n=30).
The
summary of mean IOP at each follow up visit can be found in Table 2.
Table
2 Mean IOP at each follow-up visit
|
IOP |
Pre-switch
(n=59) |
1st follow
up (104±44d; n=59) |
2nd follow
up (320±109d; n=30) |
3rd follow
up (490±145d; n=18) |
4th follow
up (708±160d; n=8) |
|
Mean
IOP (mm Hg) |
23.2 |
18.9 |
16.6 |
15.5 |
16.8 |
|
Range |
14-34 |
10-27 |
10-37 |
8-20 |
13-24 |
Adverse
Events Of all
patients in the study, four adverse events were recorded; three patients
reported increased conjunctival hyperaemia post-switch from Travaprost, and
treatment was discontinued. Another patient reported frequent headaches
associated with the switch to bimatoprost, but in this case, these side effects
were deemed minor, and the patient continued on bimatoprost monotherapy.
DISCUSSION
We
present the largest independent data set published to date following the
progress of patients switched to bimatoprost 0.03% monotherapy due to inadequate
response to previous therapy. Our findings suggest that for some patients with
glaucoma who fail to respond adequately to mono, dual, and triple medical
therapy, bimatoprost 0.03% appears to offer statistically, and clinically
significant additional IOP reduction. For over 55.9% of non-responder patients
a switch to bimatoprost monotherapy provided adequate IOP response at first
follow up. As demonstrated by our long-term follow up data, the initial IOP
reductions seen appear to be sustained, or even modestly improved by 10mo.
Our
subgroups analyses show that there is a statistically significant reduction in
IOP in OHT patients who are switched from any current treatment to bimatoprost
monotherapy. This is important, as the aim with OHT patients should always be
to achieve adequate control using medical monotherapy, and bimatoprost appears
to allow this to occur in patients not responding to other therapy.
The
second subgroup analysis compared latanoprost monotherapy to bimatoprost
monotherapy in patients with a mixture of diagnoses; again, a statistically
significant reduction in IOP was seen upon switch, suggesting that
non-responders to latanoprost monotherapy should always have a trial switch to
bimatoprost monotherapy before moving onto dual medical therapy, or selective
laser trabeculoplasty.
To
date, latanoprost remains the most commonly prescribed first line PGA in
patients with OHT and POAG, and this is confirmed by our baseline patient
demographics. Incidence of latanoprost nonresponse has been reported to be as
high as 28.1% in the Japanese population[19]. A
mixture of industry sponsored[9-10,13], and independent[15-16] short- and long-term studies have demonstrated an
additional IOP lowering effect of bimatoprost when compared to latanoprost.
A
number of reasons have been put forward for bimatoprost’s additional IOP
lowering efficacy when compared with other PGAs; PGAs such as latanoprost are
pro-drugs that require de-esterification to yield an active drug. It has been
speculated that poor de-esterification of latanoprost could explain the cohort
of latanoprost non-responders[15]. PGAs act
primarily via prostaglandin F2α prostanoid receptors[20], whereas there is in vitro, and ex vivo evidence
based on ahuman anterior segment model that bimatoprost acts on a distinct
prostamide receptor in the trabecular meshwork, increasing outflow by
approximately 40%[21].
Results
from our small observational study support switching glaucoma patients with
poor response to current treatment onto bimatoprost monotherapy before
considering other adjuvant medical or more invasive therapy. The benefits of
this approach include sustained IOP reduction on monotherapy, avoidance of
increased cost and side effects of poly-pharmacy, and improved patient
compliance due to simplicity of regime.
While
the exact mechanisms by which Bimatoprost produces its additional IOP lowering
effects on non-responders remains to be elucidated, there is a growing body of
evidence that this prostamide appears to exhibit additional IOP lowering
efficacy when compared to other PGAs.
Weaknesses
of this study include a relatively small patient cohort of 59, and
non-blindness of examiners to the patient’s treatment. It is also noted that
patient compliance to a single medication regimen may be better than to a
multi-medication regimen.
ACKNOWLEDGEMENTS
Conflicts
of Interest: Brennan N, None; Dehabadi
MH, None; Nair S, None; Quartilho A,
None; Bunce C, None; Reekie I, None; Obikpo R,
None.
REFERENCES
1 Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY.
Global prevalence of glaucoma and projections of glaucoma burden through 2040:
a systematic review and meta-analysis. Ophthalmology
2014; 121(11):2081-2090. [CrossRef] [PubMed]
2 VanVeldhuisen PC, Ederer F, Gaasterland DE, Sullivan
EK, Beck A, Prum BE, Cyrlin MN, Weiss H. The Advanced Glaucoma Intervention
Study (AGIS): 7. The relationship between control of intraocular pressure and
visual field deterioration. Am J
Ophthalmol 2000;130(4):429-440. [CrossRef]
3 Li X, He F, Gabelt BT, Wang Y, Cai S, Cao, Fan N,
Kaufman PL, Liu X. Effects of latanoprost and bimatoprost on the expression of
molecules relevant to ocular inflow and outflow pathways. PLoS One 2016;11(3):e0151644. [CrossRef] [PMC free article] [PubMed]
4 Richter M, Krauss AH, Woodward DF, Lütjen-Drecoll E.
Morphological changes in the anterior eye segment after long-term treatment
with different receptor selective prostaglandin agonists and a prostamide. Invest Ophthalmol Vis Sci
2003;44(10):4419-4426. [CrossRef] [PubMed]
5 Lim KS, Nau CB, O’Byrne MM, Hodge DO, Toris CB,
McLaren JW, Johnson DH. Mechanism of action of bimatoprost, latanoprost, and
travoprost in healthy subjects. A crossover study. Ophthalmology 2008;115(5):790-795. [CrossRef] [PMC free article] [PubMed]
6 Kymes SM, Burk C, Feinman T, Williams JM, Hollander
DA. Demonstration of an online tool to assist managed care formulary
evidence-based decision making: meta-analysis of topical prostaglandin analog
efficacy. Ther Clin Risk Manag
2011;7:283-290. [CrossRef] [PMC free article] [PubMed]
8 Casson RJ, Liu L, Graham SL, Morgan WH, Grigg JR,
Galanopoulos A, Crawford A, House PH. Efficacy and safety of bimatoprost as
replacement for latanoprost in patients with glaucoma or ocular hypertension: a
uniocular switch study. J Glaucoma
2009;18(8):582-588. [CrossRef] [PubMed]
9 Williams RD. Efficacy of bimatoprost in glaucoma and
ocular hypertension unresponsive to latanoprost. Adv Ther 2002;19(6):275-281. [CrossRef] [PubMed]
10 Law SK, Song BJ, Fang E, Caprioli J. Feasibility and
efficacy of a mass switch from latanoprost to bimatoprost in glaucoma patients
in a prepaid health maintenance organization. Ophthalmology 2005;112(12): 2123-2130. [CrossRef] [PubMed]
11 Bournias TE, Lee D, Gross R, Mattox C. Ocular
hypotensive efficacy of bimatoprost when used as a replacement for latanoprost
in the treatment of glaucoma and ocular hypertension. J Ocul Pharmacol Ther 2003;19(3):193-203. [CrossRef] [PubMed]
12 Pfennigsdorf S, Ramez O, vonKistowski G, Mäder B,
Eschstruth P, Froböse M, Thelen U, Spraul C, Schnober D, Cooper H, Laube T.
Multicenter, prospective, open-label, observational study of bimatoprost 0.01%
in patients with primary open-angle glaucoma or ocular hypertension. Clin Ophthalmol 2012;6:739-746. [CrossRef] [PMC free article] [PubMed]
13 Myers JS, Vold S, Zaman F, Williams JM, Hollander
DA. Bimatoprost 0.01% or 0.03% in patients with glaucoma or ocular hypertension
previously treated with latanoprost: two randomized 12-week trials. Clin Ophthalmol 2014;27(8):643-652. [CrossRef] [PMC free article] [PubMed]
14 Konstas AG, Holló G, Irkec M, Tsironi S, Durukan I,
Goldenfeld M, Melamed S. Diurnal IOP control with bimatoprost versus
latanoprost in exfoliative glaucoma: a crossover, observer-masked, three-centre
study. Br J Ophthalmol
2007;91(6):757-760. [CrossRef] [PMC free article] [PubMed]
15 Gandolfi SA, Cimino L. Effect of bimatoprost on
patients with primary open-angle glaucoma or ocular hypertension who are
nonresponders to latanoprost. Ophthalmology
2003;110(3):609-614. [CrossRef]
16 Sonty S, Donthamsetti V, Vangipuram G, Ahmad A.
Long-term IOP lowering with bimatoprost in open-angle glaucoma patients poorly
responsive to latanoprost. J Ocul
Pharmacol Ther 2008;24(5): 517-520. [CrossRef] [PubMed]
17 Brittain CJ, Saxena R, Waldock A. Prospective
comparative switch study from timolol 0.5% and latanoprost 0.005% to
bimatoprost 0.03%. Adv Ther
2006;23(1):68-73. [CrossRef] [PubMed]
18 Sato S, Hirooka K, Baba T, Mizote M, Fujimura T,
Tenkumo K, Ueda H, Shiraga F. Efficacy and safety of switching from topical
latanoprost to bimatoprost in patients with normal-tension glaucoma. J Ocul Pharmacol Ther
2011;27(5):499-502. [CrossRef] [PubMed]
19 Ikeda Y, Mori K, Ishibashi T, Naruse S, Nakajima N,
Kinoshita S. Latanoprost nonresponders with open-angle glaucoma in the Japanese
population. Jpn J Ophthalmol
2006;50(2):153-157. [CrossRef] [PubMed]
20 Vielhauer GA, Fujino H, Regan JW. Cloning and
localization of hFPS: a six-transmembrane mRNA splice variant of the human FP
prostanoid receptor. Arch Biochem Biophys
2004;421(2):175-185. [CrossRef]
21 Wan Z, Woodward DF, Cornell CL, Fliri HG, Martos JL,
Pettit SN, Wang JW, Kharlamb AB, Wheeler LA, Garst ME, Landsverk KJ, Struble
CS, Stamer WD. Bimatoprost, prostamide activity, and conventional drainage. Invest Ophthalmol Vis Sci 2007;48(9):4107-4115. [CrossRef] [PMC free article] [PubMed]