
・Review・ Current
Issue IF in JCR CiteScore ・Submission・ In Press Recent Accepted PMC RSS
Citation: Tang LJ, Gu CL, Zhang P. Intraocular lymphoma. Int J Ophthalmol
2017;10(8):1301-1307
Intraocular lymphoma
Li-Juan Tang1, Chang-Lin Gu2,
Ping Zhang1
1Department of Ocular Pathology, State Key Laboratory of
Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou
510060, Guangdong Province, China
2Department of Traditional Chinese Medicine, the Second Health
Service Center of Tangxia, Tianhe District, Guangzhou 510665, Guangdong
Province, China
Correspondence
to: Ping Zhang. Department of Ocular Pathology, State Key Laboratory
of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University,
Guangzhou 510060, Guangdong Province, China. pingss@126.com
Received:
2017-01-26
Accepted: 2017-04-04
Abstract
Intraocular
lymphoma (IOL) is a rare lymphocytic malignancy which contains two main distinct
forms. Primary intraocular lymphoma (PIOL) is mainly a sub-type of primary
central nervous system lymphoma (PCNSL). Alternatively, IOL can originate from
outside the central nervous system (CNS) by metastasizing to the eye. These
tumors are known as secondary intraocular lymphoma (SIOL). The IOL can arise in
the retina, uvea, vitreous, Bruch’s membrane and optic nerve. There are
predominantly of B-cell origin; however there are also rare T-cell variants.
Diagnosis remains challenging for ophthalmologists and pathologists, due to its
ability to masquerade as noninfectious or infectious uveitis, white dot
syndromes, or occasionally as other metastatic cancers. Laboratory tests
include flow cytometry, immunocytochemistry, interleukin detection (IL-10: IL-6,
ratio >1), and polymerase chain reaction (PCR) amplification.
Methotrexate-based systemic chemotherapy with external beam radiotherapy and
intravitreal chemotherapy with methotrexate are useful for controlling the
disease, but the prognosis remains poor. Therefore, it is important to make an
early diagnose and treatment. This review is focused on the clinical
manifestations, diagnosis, treatment and prognosis of the IOL.
KEYWORDS: intraocular
lymphoma; central nervous system; diagnosis; treatment; prognosis
DOI:10.18240/ijo.2017.08.19
Citation: Tang LJ, Gu CL, Zhang P. Intraocular lymphoma. Int J Ophthalmol
2017;10(8):1301-1307
BACKGROUND
The
designation of intraocular lymphoma (IOL) includes primary intraocular lymphoma
(PIOL), mainly arising from the central nervous system (CNS) and secondary
intraocular lymphoma (SIOL, from outside the CNS as metastasis from a
non-ocular neoplasm)[1-2]. IOL
incidence is very low. Most cases are of B-cell origin and associated with
primary CNS non-Hodgkin’s lymphoma. Fewer cases are of T-cell origin.
Intraocular T-cell lymphomas are uncommon, some are secondary to metastatic
systemic T-cell lymphomas including primary cutaneous peripheral T-cell
lymphoma (PCPTCL), the NK-T cell lymphoma, and rarely adult T-cell leukemia/lymphoma
(ATL)[3-7], and the disease is
usually confined to the iris and ciliary body and peripheral choroid. The most
common PIOL by far is primary vitreoretinal lymphoma (PVRL). SIOL has different
clinical features and prognosis[8], and the most
common subtype is systemic diffuse large B cell lymphoma (DLBCL)[9]. Although still rare, the incidence of IOL has
increased in the recent years, and prognosis remains poor. Here, we summarize
mainly the current literature on IOL.
EPIDEMIOLOGY
The
incidence of IOL has been increasing in recent years, due to the increase in
the patients of immunodeficiency and immunosuppression, the increase in life
expectancy, and the improvements in diagnostic tools[8,10]. The overall incidence of IOL has been estimated to
represent 1.86% of ocular malignant tumors[11].
The median age of this disease is 50-60y[12-13], with a range between 15-85 years of age[14]. These are estimated to represent 4%-6% of primary
brain tumors and 1%-2% of extranodal lymphomas[15-16]. Among IOL patients, the percentage of cases that
involve the CNS is 60%-80%[17]. While 15%-25% of
primary central nervous system lymphoma (PCNSL) patients develop ophthalmic
manifestations of lymphoma, 56%-90% of PIOL patients have or will develop CNS
manifestations of lymphoma[18]. In terms of
gender, some reported that women were more commonly affected than men by 2:1[19-21]. But some reported that even
greater cases occurred in men[22]. There appears
to be no racial predilection for the disease[22-23].
ETIOLOGY
The
etiology of IOL remains unclear. Multiple hypotheses of lymphomagenesis are
involved. Immunocompromise, Epstein-Barr virus, and Toxoplasma gondii infection
may be the related factors[24-26].
Moreover, an infectious antigen driven B-cell expansion may be the primary
trigger, which then becomes cloned[8]. Thus,
genetic, immunologic, and microenvironmental factors are probably necessary in
order to induce malignant B-cell phenotype[27].
Proofs of causation are still lacking, and the lymphomagenesis requires further
investigation.
CLINICAL
FEATURES
PIOL
is a masquerade syndrome that mimics uveitis, even responds to steroid therapy,
which makes the diagnosis difficult. Ocular disease is bilateral in 64%-83% of
cases[28]. Blurred vision, reduced vision, and
floaters are the common initial subjective symptoms[17].
More than 50% of patients have significant vitreous haze and cells that can be
seen insheets or clumps with vision impairment[29].
Posterior vitreous detachment and hemorrhage may occur occasionally[30]. Posterior uveitis is the most common presenting
symptoms, and anterior segment inflammatory findings are frequently absent[18]. Another characteristic from optical coherence
tomography (OCT) is the development of creamy lesions with orange-yellow
infiltrates to the retina or retinal pigment epithelium (RPE)[1,31-32]. They can give rise to a
characteristic “leopard skin” pigmentation overlying the mass which may be seen
in fluorescein angiography (FA)[32-35].
There may beisolated subretinal lesionsor associated exudative retinal
detachment[23,33]. A single
vitreous lesion is rare, sometimes simple vitreous inflammatory response or
optic nerve infiltration may occur[36]. At
presentation of PIOL, 56%-90% patients have or will develop CNS manifestations
of lymphoma[14]. Sometimes IOL may masquerade as
bilateral granulomatous panuveitis[37]. When
there is infiltration to the brain, behavioral changes and alteration in
cognitive function may occur[38].
Intraocular
T-cell lymphomas are uncommon, some of them are secondary to metastatic
systemic T-cell lymphomas. SIOL should be considered when there is a bilateral
sudden and severe inflammatory reaction of the anterior segment that does not
respond to treatment or recurs. Anterior reaction and keratic precipitates may
be presented especially in SIOL[39]. The most
common ocular manifestation of this disease is non-granulomatous anterior
uveitis and vitritis. Other rare ocular symptoms include inflammatory glaucoma,
neurotrophic keratopathy, fully dilated pupil, and choroidal detachment[40]. Previous systemic primary site reported indicated
that the skin was the most common site. Concurrent CNS involvement was reported
in 31.0% cases[41].
DIAGNOSTIC
TESTS
The
delay between a positive diagnosis and the onset of ocular or neurological
symptoms usually ranges from 4-40mo[23,28,42], although more rapid progression may occur[43].The diagnosis of IOL requires a multidisciplinary
approach, involving morphological assessment in conjunction with traditional
immunocytochemistry and molecular analysis [such as flow cytometry and
polymerase chain reaction (PCR) analysis]. Histologic identification remains
one of the essential procedures in diagnosing IOL[44-45]. Morphologically and immunohistochemically, the
typical lymphoma cells are usually with scanty cytoplasm, an elevated nucleus:
cytoplasm ratio, round, oval, bean, or irregular shaped nuclei with a coarse
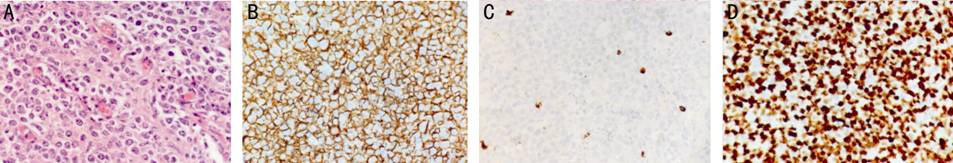
chromatin and prominent or multiple nucleoli[46-47] (Figure 1A). In B cell lymphoma the predominance of
lymphoma cells were identified as CD20, CD79α positive and CD3 negative (Figure
1B, 1C). And in T cell lymphoma, medium to large sized lymphoid cells with
atypical nuclei are visualized with HE staining (Figure 2A). While the tumor cells
are identified as CD3 positive and CD20 negative (Figure 2B, 2C). Both the two
types of IOL are with high Ki-67 positive rate (average >80%) indicates
extensive proliferation (Figures 1D, 2D). Specimens can be obtained by fine
needle vitreous aspiration or pars plana vitrectomy. And multiple biopsies may
be required to reach a definite pathological diagnosis. Removed ocular fluids (via
aqueous tap, vitreous tap, or diagnostic vitrectomy) need to be delivered
quickly for laboratory analysis, to prevent cell degeneration that can make
diagnosis difficult[47]. Furthermore, a negative
vitrectomysample is common, sparse number of cells is also the main reason for
misdiagnosis. In addition, vitreous specimens contain many reactive
T-lymphocytes, necrotic cells, debris, and fibrin that can also confound the
identification of malignant cells[46]. Then,
retinal or chorioretinal biopsies may be required. Microscopically, the typical
lymphoma cells are large B-cell lymphoid cells with scanty cytoplasm, an
elevated (nucleus:cytoplasm) ratio, heteromorphic deeply stained nuclei with a
coarse chromatin pattern and prominent nucleoli can be seen (Figure 3). Pars
plana vitrectomy has several advantages, including improved vision by clearance
of vitreous debris and maximizing the sample size[44,48-49], although the lymphoma may
extend to the epibulbar space through the sclerotomy port following vitrectomy[50].
Figure
1 Histopathological and immunohistological of IOL origining from B cell The atypical
cells have pleomorphic nuclei with conspicuous nucleoli and scanty cytoplasm
(A, HE staining, 200×). It is positive for CD20 (B, 200×) and negative for CD3
(C, 200×); the high Ki-67 positive rate (average >80%) indicates extensive
proliferation (D, 200×).
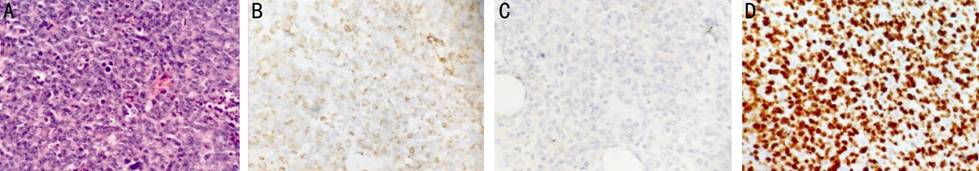
Figure
2 Histopathological and immunohistological of IOL origining from T cell Medium-to-large-sized lymphoid cells with
atypical nuclei are visualized with HE staining (A, 200×) and identified as CD3
positive (B, 200×), CD20 negative (C, 200×); the high Ki-67 positive rate
(average >80%) indicates extensive proliferation (D, 200×).
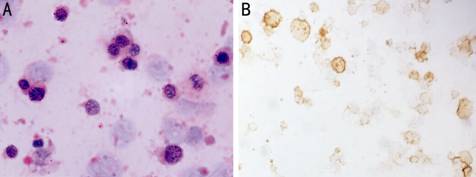
Figure
3 Cytology of the vitreous specimen reveals some atypical, large lymphoid
cells, with large, deeply stained, irregular nuclei and coarse chromatin (A,
400×), which is identified as CD20 positive (B, 400×).
Molecular
analysis detecting immunoglobulin gene rearrangements in the lymphoma cells and
ocular cytokine analysis of vitreous fluid show elevated interleukin (IL-10)
with an IL-10:IL-6 ratio >1.0 are helpful for the diagnosis, while
inflammatory conditions typically show elevated IL-6[17,51-53]. In addition, if the eyes have
no function or conservative treatment is impossible, a diagnostic enucleation
may become necessary[54]. Flow cytometry can
examine cell surface markers and demonstrate monoclonal B-cell populations. IOL
is typically comprised of amonoclonal B-cell population with restricted κ or λ
chains. A κ:λ ratio of 3 or 0.6 is a highly sensitive marker for lymphoma[55]. PCR has been used to amplify the immunoglobulin
heavy chain DNA. In B-cell lymphomas, molecular analysis can detect IgH gene
rearrangements, while in T-cell lymphomas, T-cell receptor gene rearrangements
can be detected[56]. Detection of the bcl-2 t
(14;18) translocation is also an effective method to diagnose IOL. Wallace et
al[57] reported that 40 of 72 (55%) PIOL
patients expressed the bcl-2 t (14;18) translocation at the major breakpoint
region. A PCR analysis of EB virus with aqueous humor might be useful for
supporting the diagnosis of intraocular NK-cell lymphoma[58].
Microdissection with a minimum of 15 atypical lymphoid cells has been shown to
have a diagnostic efficiency of 99.5% by using PCR[59].
Besides,
ophthalmological examinations frequently demonstrate the presence of vitritis,
usually in association with infiltrates of the retina and the retinal pigment
epithelium. Hyperfluorescense on fundus autofluorescence imaging can
demonstrate active sub-retinal pigment epithelium deposits, while
hypofluorescent spots can correspond to areas where tumor cells were suspected
to have once been[60]. Fluorescein angiography
(FA) may show mottling, granularity, and late staining patterns with a
characteristic “leopard spotappearance”[23,32,34-35,61].
OCT demonstrates hyper reflective lesions at the level of the RPE in PVRL. The
valuable diagnostic tools include fundoscopy, FA, OCT, fundus autofluorescence,
and fluorescein and indocyanine green angiography. There has been reported that
ophthalmological examinations finding had a positive predictive value of 88.9%
and a negative predictive value of 85%[32]. In
addition, once cerebral lymphoma is suspected, contrast-enhanced cranial
magnetic resonance imaging (MRI) is the best imaging modality. Lesions are
often isointense to hypointense on T2-weighted MRI, with variable surrounding
edema and a homogeneous and strong pattern of enhancement[62-63].
TREATMENT
Due
to the rarity of IOL, standard and optimal therapy is not defined. Treatment
modalities for IOL include intravitreal chemotherapy, systemic chemotherapy,
and radiotherapy, which is used alone or in an appropriate combination. The
therapies vary according to the disease degree, the presence or absence of CNS
involvement, and performance status of the patients[64].
The current recommendation for the treatment of IOL without CNS or systemic
involvement should be limited to local treatment, including intraocular
methotrexate and/or ocular radiation in order to minimize systemic toxicities[38]. Ocular irradiation with prophylactic CNS treatment
is used to control IOL, maintain vision, and prevent CNS involvement[24]. The average external beam radiation dose is close to
40 Gy, but can range from 30 to 50 Gy[29]. The
complications of radiotherapy include radiation retinopathy, vitreous
hemorrhage, dry eye syndrome, conjunctivitis, neovascular glaucoma, optic
atrophy, punctate epithelial erosions or cataract[29].
While the treatment of the patients with CNS involvement includes a combination
of radiotherapy and chemotherapy[63,65].
As with systemic chemotherapy, the mainstay of intravitreal chemotherapy is
methotrexate[66]. Rituximab is an anti-CD20
monoclonal antibody. And intravitreal rituximab is often used to decrease the
frequency of methotrexate injections or for methotrexate-resistant IOL[67-69]. Initial response was good with
clearance of PIOL, but subsequent relapse required intravitreal methotrexateand
radiation[67]. Methotrexate can also use alone or
in combination with other medications, such as thiotepa and dexamethasone[34,70-72]. High
dose methotrexate is the most active drug, producing a response rate of up to
72% when used alone and up to 94%-100% in combinations[73-74]. Combined intravitreal methotrexate and systemic
high-dosemethotrexate treatment is effective in patients with PIOL[75]. However, polychemotherapy is also associated with
higher drug toxicity[29]. In addition,
intravitreal chemotherapy with 0.4 mg methotrexate in 0.1 mL achieved local
tumor control in relapsed IOL[76-77].
Also the intravitreal chemotherapy is a primary treatment in combination with
systemic chemotherapy[70]. Drug resistance may
occur with repeated injections[78]. For relapsed
or refractory PIOL with PCNSL has been treated with intrathecal methotrexate
and cytarabine[79]. These treatment decisions are
often complex and require personalized treatment for different patients.
PROGNOSIS
IOL
is a rare lymphocytic malignancy, the reported mortality rate range between 9%
and 81% in follow-up periods, and the survival time is 12-35mo[19,80-82]. However,
the reported mortality rate of IOL is very inconsistent because of the rare
patient populations, variation in treatment modalities, and the delayed
diagnosis. Tumor recurrence is common, and sometimes the existing treatment
cannot effectively prevent the local recurrence and the CNS involvement. The
prognosis depends on the following aspects: 1) whether the CNS is involved. A
trend toward better survival was seen among patients with isolated ocular
presentation[83-84]. Moreover,
the IOL patients with CNS involvement are almost died in the short term.
Neuroimaging is important for the patients after treatment since the IOL
patients carry a risk for recurrence or CNS involvement[85];
2) histopathologic type is another important factors. Generally T cell type has
poorer prognosis than B cell type; 3) treatment opportunity, early treatment
after onset of symptoms may improve the prognosis for a better visual outcome[86]. In PCNSL, median survival of patients treated with
radiotherapy alone or chemotherapy plus radiotherapy ranges from 10 to 16mo[38]; 4) the vision and survival rates are both poor in
recurrent patients. Due to the rarity of the disease, there is often
misdiagnosis, delay in diagnosis, and mismanagement of IOL.
CONCLUSION
In
conclusion, IOL often masquerades as intraocular inflammation resulting in
misdiagnosis or delayed diagnosis, with subsequent inappropriate management and
high mortality rates. Patients with suspected IOL should undergo cytopathologic
examination of vitreal fluid or vitrectomy before therapy. On the ocular
cytokine levels, mainly IL-10:IL-6 ratio and molecular analysis can provide
useful supplementary data for the diagnosis. Once IOL is diagnosed, all
patients are required to be examined the subtle neurological symptoms and signs
with the oncologist. Meanwhile, neuroimaging is performed to detect any
evidence of CNS involvement. Optimal therapy for IOL is considered a great
challenge to the clinical. Intravitreal chemotherapy with more than one agent
may be proved to be useful in controlling the ocular disease. When the CNS
involved, methotrexate-based systemic chemotherapy with external beam
radiotherapy should be undertaken. Because of the rarity of this disease,
multicenter studies are needed to obtain optimized treatment methods in order
to get better vision and prognosis.
METHOD OF LITERATURE SEARCH
PubMed
(1970 to end of 2016) database was searched using the search terms IOL, PIOL
and SIOL. Removing duplicate articles, excluding articles that clearly related
to extraocular lymphoma, and removing foreign language papers provided a total
of 86 unique articles in English.
ACKNOWLEDGEMENTS
Foundation:
Supported by the National Natural Science Foundation of China
(No.30371515).
Conflicts
of Interest: Tang LJ, None; Gu CL, None; Zhang P, None.
REFERENCES
1 Buggage RR, Chan CC, Nussenblatt RB. Ocular
manifestations of central nervous system lymphoma. Curr Opin Oncol 2001;13(3):137-142. [CrossRef]
2 Fredrick DR, Char DH, Ljung BM, Brinton DA. Solitary
intraocular lymphoma as an initial presentation of widespread disease. Arch Ophthalmol 1989;107(3):395-397. [CrossRef]
3 Levy-Clarke GA, Buggage RR, Shen D, Vaughn LO, Chan
CC, Davis JL. Human T-cell lymphotropic virus type-1 associated T-cell
leukemia/lymphoma masquerading as necrotizing retinal vasculitis. Ophthalmology 2002;109(9):1717-1722. [CrossRef]
4 Goldey SH, Stern GA, Oblon DJ, Mendenhall NP, Smith
LJ, Duque RE. Immunophenotypic characterization of an unusual T-cell lymphoma
presenting as anterior uveitis. A clinicopathologic case report. Arch Ophthalmol 1989;107(9):1349-1353. [CrossRef] [PubMed]
5 Saga T, Ohno S, Matsuda H, Ogasawara M, Kikuchi K.
Ocular involvement by a peripheral T-cell lymphoma. Arch Ophthalmol 1984; 102(3):399-402. [CrossRef]
6 Hunyor AP, Harper CA, O'Day J, McKelvie PA.
Ocular-central nervous system lymphoma mimicking posterior scleritis with
exudative retinal detachment.
Ophthalmology 2000;107(10):1955-1959. [CrossRef]
7 Chaput F, Amer R, Baglivo E, Touitou V, Kozyreff A,
Bron D, Bodaghi B, LeHoang P, Bergstrom C, Grossniklaus HE, Chan CC, Pe’er J,
Caspers LE. Intraocular T-cell lymphoma: clinical presentation, diagnosis,
treatment, and outcome. Ocul Immunol
Inflamm 2016;22:1-10. [CrossRef]
[PubMed]
8 Chan CC. Molecular pathology of primary intraocular
lymphoma. Trans Am Ophthalmol Soc
2003;101:275-292. [PMC free article] [PubMed]
9 Coupland SE, Damato B. Understanding intraocular
lymphomas. Clin Exp Ophthalmol 2008;36(6):564-578.
[CrossRef] [PubMed]
10 Mulay K, Narula R, Honavar SG. Primary
vitreoretinal lymphoma. Indian J
Ophthalmol 2015;63(3):180-186. [CrossRef]
[PMC free article] [PubMed]
11 Reddy EK, Bhatia P, Evans RG. Primary orbital
lymphomas. Int J Radiat Oncol Biol Phys
1988;15(5):1239-1241. [CrossRef]
12 Cho BJ, Yu HG. Risk factors for intraocular
involvement in patients with primary central nervous system lymphoma. J Neurooncol 2014;120(3):523-529. [CrossRef]
[PubMed]
13 DeAngelis LM, Yahalom J, Thaler HT, Kher U.
Combined modality therapy for primary CNS lymphoma. J Clin Oncol 1992;10(4):635-643. [CrossRef]
[PubMed]
14 Chan CC, Rubenstein JL, Coupland SE, Davis JL,
Harbour JW, Johnston PB, Cassoux N, Touitou V, Smith JR, Batchelor TT, Pulido
JS. Primary vitreoretinal lymphoma: a report from an International Primary
Central Nervous System Lymphoma Collaborative Group symposium. Oncologist 2011;16(11):1589-1599. [CrossRef]
[PMC free article] [PubMed]
15 Freeman LN, Schachat AP, Knox DL, Michels RG, Green
WR. Clinical features, laboratory investigations, and survival in ocular
reticulum cell sarcoma. Ophthalmology 1987;94(12):1631-1639. [CrossRef]
16 Hochberg FH, Miller DC. Primary central nervous
system lymphoma. J Neurosurg
1988;68(6):835-853. [CrossRef]
[PubMed]
17 Kimura K, Usui Y, Goto H. Clinical features and
diagnostic significance of the intraocular fluid of 217 patients with
intraocular lymphoma. Jpn J Ophthalmol
2012;56(4):383-389. [CrossRef]
[PubMed]
18 Chan CC, Sen HN. Current concepts in diagnosing and
managing primary vitreoretinal (intraocular) lymphoma. Discov Med 2013;15(81): 93-100. [PMC free article] [PubMed]
19 Berenbom A, Davila RM, Lin HS, Harbour JW.
Treatment outcomes for primary intraocular lymphoma: implications for external
beam radiotherapy. Eye (Lond)
2007;21(9):1198-1201. [CrossRef]
[PubMed]
20 Buettner H, Bolling JP. Intravitreal large-cell
lymphoma. Mayo Clin Proc 1993;68(10):1011-1015. [CrossRef]
21 Peterson K, Gordon KB, Heinemann MH, DeAngelis LM.
The clinical spectrum of ocular lymphoma. Cancer
1993;72(3):843-849. [CrossRef]
22 Qualman SJ, Mendelsohn G, Mann RB, Green WR.
Intraocular lymphomas. Natural history based on a clinicopathologic study of
eight cases and review of the literature. Cancer
1983;52(5):878-886. [CrossRef]
23 Cassoux N, Merle-Beral H, Leblond V, Bodaghi B,
Miléa D, Gerber S, Fardeau C, Reux I, Xuan KH, Chan CC, LeHoang P. Ocular and
central nervous system lymphoma: clinical features and diagnosis. Ocul Immunol Inflamm 2000;8(4):243-250.
[CrossRef]
24 Margolis L, Fraser R, Lichter A, Char DH. The role
of radiation therapy in the management of ocular reticulum cell sarcoma. Cancer 1980;45(4):688-692. [CrossRef]
25 Harris NL, Jaffe ES, Diebold J, Flandrin G,
Muller-Hermelink HK, Vardiman J, Lister TA, Bloomfield CD. World Health
Organization classification of neoplastic diseases of the hematopoietic and
lymphoid tissues: report of the Clinical Advisory Committee meeting-Airlie
House, Virginia, November 1997. J Clin
Oncol 1999;17(12):3835-3849. [CrossRef]
[PubMed]
26 Shen DF, Herbort CP, Tuaillon N, Buggage RR,
Egwuagu CE, Chan CC. Detection of Toxoplasma gondii DNA in primary intraocular
B-cell lymphoma. Mod Pathol
2001;14(10):995-999. [CrossRef]
[PubMed]
27 Chan CC. Primary intraocular lymphoma: clinical
features, diagnosis, and treatment. Clin
Lymphoma 2003;4(1):30-31. [CrossRef]
28 Hoffman PM, McKelvie P, Hall AJ, Stawell RJ,
Santamaria JD. Intraocular lymphoma: a series of 14 patients with
clinicopathological features and treatment outcomes. Eye (Lond) 2003;17(4):513-521. [CrossRef]
[PubMed]
29 Hwang CS, Yeh S, Bergstrom CS. Diagnostic
vitrectomy for primary intraocular lymphoma: when, why, how? Int Ophthalmol Clin 2014;54(2):155-171.
[CrossRef] [PMC free article] [PubMed]
30 Gill MK, Jampol LM. Variations in the presentation
of primary intraocular lymphoma: case reports and a review. Surv Ophthalmol 2001; 45(6):463-471. [CrossRef]
31 Hormigo A, DeAngelis LM. Primary ocular lymphoma:
clinical features, diagnosis, and treatment. Clin Lymphoma 2003;4(1):22-29. [CrossRef]
[PubMed]
32 Fardeau C, Lee CP, Merle-Beral H, Cassoux N,
Bodaghi B, Davi F, Lehoang P. Retinal fluorescein, indocyanine green
angiography, and optic coherence tomography in non-Hodgkin primary intraocular
lymphoma. Am J Ophthalmol 2009;147(5):886-894.
[CrossRef] [PubMed]
33 Levy-Clarke GA, Byrnes GA, Buggage RR, Shen DF,
Filie AC, Caruso RC, Nussenblatt RB, Chan CC. Primary intraocular lymphoma
diagnosed by fine needle aspiration biopsy of a subretinal lesion. Retina 2001;21(3):281-284. [CrossRef]
34 Velez G, Chan CC, Csaky KG. Fluorescein
angiographic findings in primary intraocular lymphoma. Retina 2002;22(1):37-43. [CrossRef]
35 Meleth AD, Sen HN. Use of fundus autofluorescence
in the diagnosis and management of uveitis. Int
Ophthalmol Clin 2012;52(4):45-54. [CrossRef]
[PMC free article] [PubMed]
36 Hedayatfar A, Phaik Chee S. Presumptive primary
intraocular lymphoma presented as an intraocular mass involving the optic nerve
head. J Ophthalmic Inflamm Infect
2012;2(1):49-51. [CrossRef] [PMC free article] [PubMed]
37 Kanavi MR, Soheilian M, Bijanzadeh B, Peyman GA.
Diagnostic vitrectomy (25-gauge)in a case with intraocular lymphoma masquerading
as bilateral granulomatous panuveitis. Eur
J Ophthalmol 2010;20(4): 795-798. [PubMed]
38 Grimm SA, McCannel CA, Omuro AM, Ferreri AJ, Blay
JY, Neuwelt EA, Siegal T, Batchelor T, Jahnke K, Shenkier TN, Hall AJ, Graus F,
Herrlinger U, Schiff D, Raizer J, Rubenstein J, Laperriere N, Thiel E,
Doolittle N, Iwamoto FM, Abrey LE. Primary CNS lymphoma with intraocular
involvement: International PCNSL Collaborative Group Report. Neurology 2008;71(17):1355-1360. [CrossRef] [PMC free article] [PubMed]
39 Abusamra K, Oray M, Ebrahimiadib N, Lee S, Anesi S,
Foster CS. Intraocular lymphoma: descriptive data of 26 patients including
clinico-pathologic features, vitreous findings, and treatment outcomes. Ocul Immunol Inflamm 2016;20:1-6. [CrossRef]
[PubMed]
40 Lin TC, Lin PY, Wang LC, Chen SJ, Chang YM, Lee SM.
Intraocular involvement of T-cell lymphoma presenting as inflammatory glaucoma,
neurotrophic keratopathy, and choroidal detachment. J Chin Med Assoc 2014;77(7):385-388. [CrossRef]
[PubMed]
41 Levy-Clarke GA, Greenman D, Sieving PC, Byrnes G,
Shen D, Nussenblatt R, Chan CC. Ophthalmic manifestations, cytology,
immunohistochemistry, and molecular analysis of intraocular metastatic T-cell
lymphoma: report of a case and review of the literature. Surv Ophthalmol 2008;53(3):285-295. [CrossRef] [PMC free article] [PubMed]
42 Grimm SA, Pulido JS, Jahnke K, Schiff D, Hall AJ,
Shenkier TN, Siegal T, Doolittle ND, Batchelor T, Herrlinger U, Neuwelt EA,
Laperriere N, Chamberlain MC, Blay JY, Ferreri AJ, Omuro AM, Thiel E, Abrey LE.
Primary intraocular lymphoma: an International Primary Central Nervous System
Lymphoma Collaborative Group Report. Ann
Oncol 2007;18(11):1851-1855. [CrossRef]
[PubMed]
43 Vogel MH, Font RL, Zimmerman LE, Levine RA.
Reticulum cell sarcoma of the retina and uvea. Report of six cases and review
of the literature. Am J Ophthalmol
1968;66(2):205-215. [CrossRef]
44 Char DH, Kemlitz AE, Miller T. Intraocular biopsy. Ophthalmol Clin North Am 2005;18(1):177-185. [CrossRef]
[PubMed]
45 Mastropasqua R, Thaung C, Pavesio C, Lightman S,
Westcott M, Okhravi N, Aylward W, Charteris D, da Cruz L. The role of
chorioretinal biopsy in the diagnosis of intraocular lymphoma. Am J Ophthalmol 2015; 160(6):1127-1132.
[CrossRef] [PubMed]
46 Coupland SE, Bechrakis NE, Anastassiou G, Foerster
AM, Heiligenhaus A, Pleyer U, Hummel M, Stein H. Evaluation of vitrectomy
specimens and chorioretinal biopsies in the diagnosis of primary intraocular
lymphoma in patients with Masquerade syndrome. Graefes Arch Clin Exp Ophthalmol 2003;241(10):860-870. [CrossRef]
[PubMed]
47 Karma A, von Willebrand EO, Tommila PV, Paetau AE,
Oskala PS, Immonen IJ. Primary intraocular lymphoma: improving the diagnostic
procedure. Ophthalmology
2007;114(7):1372-1377. [CrossRef]
[PubMed]
48 Gonzales JA, Chan CC. Biopsy techniques and yields
in diagnosing primary intraocular lymphoma. Int
Ophthalmol 2007;27(4):241-250. [CrossRef]
[PMC free article] [PubMed]
49 Margolis R. Diagnostic vitrectomy for the diagnosis
and management of posterior uveitis of unknown etiology. Curr Opin Ophthalmol 2008; 19(3):218-224. [CrossRef]
[PubMed]
50 Cursiefen C, Holbach LM, Lafaut B, Heimann K,
Kirchner T, Naumann GO. Oculocerebral non-Hodgkin's lymphoma with uveal
involvement: development of an epibulbar tumor after vitrectomy. Arch Ophthalmol 2000;118(10):1437-1440.
[CrossRef]
51 Davis JL. Intraocular lymphoma: a clinical
perspective. Eye (Lond) 2013;27(2):153-162.
[CrossRef] [PMC free article] [PubMed]
52 Rajagopal R, Harbour JW. Diagnostic testing and
treatment choices in primary vitreoretinal lymphoma. Retina 2011;31(3):435-440. [CrossRef]
[PubMed]
53 Sugita S, Takase H, Sugamoto Y, Arai A, Miura O,
Mochizuki M. Diagnosis of intraocular lymphoma by polymerase chain reaction
analysis and cytokine profiling of the vitreous fluid. Jpn J Ophthalmol 2009;53(3):209-214. [CrossRef]
[PubMed]
54 Trudeau M, Shepherd FA, Blackstein ME,
Gospodarowicz M, Fitzpatrick P, Moffatt KP. Intraocular lymphoma: report of
three cases and review of the literature. Am
J Clin Oncol 1988;11(2):126-130. [CrossRef]
55 Davis JL, Miller DM, Ruiz P. Diagnostic testing of
vitrectomy speciments. Am J Ophthalmol 2005;140(5):822-829.
[CrossRef] [PubMed]
56 Shen DF, Zhuang Z, LeHoang P, Böni R, Zheng S,
Nussenblatt RB, Chan CC. Utility of microdissection and polymerase chain
reaction for the detection of immunoglobulin gene rearrangement and
translocation in primary intraocular lymphoma. Ophthalmology 1998;105(9):1664-1669. [CrossRef]
57 Wallace DJ, Shen D, Reed GF, Miyanaga M, Mochizuki
M, Sen HN, Dahr SS, Buggage RR, Nussenblatt RB, Chan CC. Detection of the bcl-2
t(14;18) translocation and proto-oncogene expression in primary intraocular
lymphoma. Invest Ophthalmol Vis Sci
2006;47(7):2750-2756. [CrossRef] [PMC free article] [PubMed]
58 Tagawa Y, Namba K, Ogasawara R, Kanno H, Ishida S.
A case of mature natural killer-cell neoplasm manifesting multiple choroidal
lesions: primary intraocular natural killer-cell lymphoma. Case Rep Ophthalmol 2015;6(3):380-384. [CrossRef]
[PMC free article] [PubMed]
59 Wang Y, Shen D, Wang VM, Sen HN, Chan CC. Molecular
biomarkers for the diagnosis of primary vitreoretinal lymphoma. Int J Mol Sci 2011;12(9):5684-5697. [CrossRef]
[PMC free article] [PubMed]
60 Ishida T, Ohno-Matsui K, Kaneko Y, Tobita H,
Shimada N, Takase H, Mochizuki M. Fundus autofluorescence patterns in eyes with
primary intraocular lymphoma. Retina
2010;30(1):23-32. [CrossRef]
[PMC free article] [PubMed]
61 Garweg JG, Wanner D, Sarra GM, Altwegg M, Loosli H,
Kodjikian L, Halberstadt M. The diagnostic yield of vitrectomy specimen
analysis in chronic idiopathic endogenous uveitis. Eur J Ophthalmol 2006;16(4):588-594. [PubMed]
62 Kuker W, Nagele T, Korfel A, Heckl S, Thiel E,
Bamberg M, Weller M, Herrlinger U. Primary central nervous system lymphomas
(PCNSL): MRI features at presentation in 100 patients. J Neurooncol 2005;72:169-177.
[CrossRef] [PubMed]
63 Hoang-Xuan K, Bessell E, Bromberg J, et al. Diagnosis and treatment of
primary CNS lymphoma in immunocompetent patients: guidelines from the European
Association for Neuro-Oncology. Lancet
Oncol 2015;16(7):e322-332. [CrossRef]
64 Sagoo MS, Mehta H, Swampillai AJ, Cohen VM, Amin
SZ, Plowman PN, Lightman S. Primary intraocular lymphoma. Surv Ophthalmol 2014;59(5):503-516. [CrossRef] [PubMed]
65 Stubiger N, Kakkassery V, Gundlach E, Winterhalter
S, Pleyer U. Diagnostics and treatment of primary vitreoretinal lymphoma. Ophthalmologe 2015;112(3):223-230. [CrossRef]
[PubMed]
66 Huang YC, Jou JR. Intravitreal injections of
methotrexate in treatment of primary central nervous system lymphoma with
intraocular involvement. Kaohsiung J Med
Sci 2016;32(12):638-639. [CrossRef]
[PubMed]
67 Itty S, Olson JH, O’Connell DJ, Pulido JS.
Treatment of primary intraocular lymphoma (PIOL) has involved systemic,
intravitreal or intrathecal chemotherapy and/or radiotherapy. Retina 2009;29(3):415-416. [CrossRef]
[PubMed]
68 Kim H, Csaky KG, Chan CC, Bungay PM, Lutz RJ,
Dedrick RL, Yuan P, Rosenberg J, Grillo-Lopez AJ, Wilson WH, Robinson MR. The
pharmacokinetics of rituximab following an intravitreal injection. Exp Eye Res 2006;82(5):760-766. [CrossRef]
[PubMed]
69 Kitzmann AS, Pulido JS, Mohney BG, Baratz KH, Grube
T, Marler RJ, Donaldson MJ, O'Neill BP, Johnston PB, Johnson KM, Dixon LE,
Salomao DR, Cameron JD. Intraocular use of rituximab. Eye (Lond) 2007;21(12):1524-1527. [CrossRef]
[PubMed]
70 Fishburne BC, Wilson DJ, Rosenbaum JT, Neuwelt EA.
Diagnosis and treatment of primary CNS lymphoma in immunocompetent patients:
guidelines from the European Association for Neuro-Oncology. Intravitreal
methotrexate as an adjunctive treatment of intraocular lymphoma. Arch Ophthalmol 1997;115(9):1152-1156. [CrossRef]
71 de Smet MD, Vancs VS, Kohler D, Solomon D, Chan CC.
Diagnosis and treatment of primary CNS lymphoma in immunocompetent patients:
guidelines from the European Association for Neuro-Oncology. Intravitreal
chemotherapy for the treatment of recurrent intraocular lymphoma. Br J Ophthalmol 1999;83(4):448-451. [CrossRef]
[PMC free article] [PubMed]
73 Tempescul A, Pradier O, Marianowski-Cochard C,
Ianotto JC, Berthou C. Combined therapy associating systemic platinum-based
chemotherapy and local radiotherapy into the treatment of primary intraocular
lymphoma. Ann Hematol
2011;90(9):1117-1118. [CrossRef]
[PubMed]
74 Korfel A, Schlegel U. Diagnosis and treatment of
primary CNS lymphoma. Nat Rev Neurol
2013;9(6):317-327. [CrossRef]
[PubMed]
75 Ma WL, Hou HA, Hsu YJ, Chen YK, Tang JL, Tsay W,
Yeh PT, Yang CM, Lin CP, Tien HF. Clinical outcomes of primary intraocular
lymphoma patients treated with front-line systemic high-dose methotrexate and
intravitreal methotrexate injection. Ann
Hematol 2016;95(4):593-601. [CrossRef]
[PubMed]
76 de Smet MD, Stark-Vancs V, Kohler DR, Smith J,
Wittes R, Nussenblatt RB. Intraocular levels of methotrexate after intravenous
administration. Am J Ophthalmol
1996;121(4):442-444. [CrossRef]
77 Wang JK, Yang CM, Lin CP, Shan YD, Lo AY, Tien HF.
An Asian patient with intraocular lymphoma treated by intravitreal
methotrexate. Jpn J Ophthalmol
2006;50(5):474-478. [CrossRef]
[PubMed]
78 Sen HN, Chan CC, Byrnes G, Fariss RN, Nussenblatt
RB, Buggage RR. Intravitreal methotrexate resistance in a patient with primary
intraocular lymphoma. Ocul Immunol
Inflamm 2008;16(1):29-33. [CrossRef]
[PMC free article] [PubMed]
79 Mason JO, Fischer DH. Intrathecal chemotherapy for
recurrent central nervous system intraocular lymphoma. Ophthalmology 2003;110(6): 1241-1244. [CrossRef]
80 Akpek EK, Ahmed I, Hochberg FH, Soheilian M, Dryja
TP, Jakobiec FA, Foster CS. Intraocular-central nervous system lymphoma:
clinical features, diagnosis, and outcomes. Ophthalmology
1999;106(9):1805-1810. [CrossRef]
81 Dunleavy K, Wilson WH. Primary intraocular lymphoma:
current and future perspectives. Leuk
Lymphoma 2006;47(9):1726-1727. [CrossRef]
[PubMed]
82 Isobe K, Ejima Y, Tokumaru S, Shikama N, Suzuki G,
Takemoto M, Tsuchida E, Nomura M, Shibamoto Y, Hayabuchi N. Treatment of
primary intraocular lymphoma with radiation therapy: a multi-institutional
survey in Japan. Leuk Lymphoma 2006;47(9):1800-1805.
[CrossRef] [PubMed]
83 Kim MM, Dabaja BS, Medeiros J, Kim S, Allen P,
Chevez-Barrios P, Gombos DS, Fowler N. Survival outcomes of primary intraocular
lymphoma: a single-institution experience. Am
J Clin Oncol 2016;39(2): 109-113. [CrossRef]
[PubMed]
84 Lee S, Kim MJ, Kim JS, Oh SY, Kim SJ, Kwon YH,
Chung IY, Kang JH, Yang DH, Kang HJ, Yoon DH, Kim WS, Kim HJ, Suh C.
Intraocular lymphoma in Korea: the consortium for improving survival of
lymphoma (CISL) study. Blood Res 2015;50(4):242-247.
[CrossRef] [PMC free article] [PubMed]
85 Jahnke K, Thiel E, Bechrakis NE, Willerding G,
Kraemer DF, Fischer L, Korfel A. Ifosfamide or trofosfamide in patients with
intraocular lymphoma. J Neurooncol
2009;93(2):213-217. [CrossRef]
[PubMed]
86 Frenkel S, Hendler K, Siegal T, Shalom E, Pe’er J.
Intravitreal methotrexate for treating vitreoretinal lymphoma: 10 years of
experience. Br J Ophthalmol
2008;92(3):383-388. [CrossRef]
[PubMed]