��Clinical
Research��
Postoperative
adjunctive bevacizumab versus placebo in primary trabeculectomy surgery for
glaucoma
1Department of Ophthalmology,
2Department of Ophthalmology and Vision Sciences,
3Department of Ophthalmology,
Correspondence to:
Received:
Abstract
AIM: To compare the effectiveness of postoperative
adjunctive use of subconjunctival bevacizumab in altering the outcome of
primary trabeculectomy in terms of sustained lowering of intraocular pressure
(IOP) and reduction of postoperative bleb vascularization and fibrosis.
METHODS: A prospective, one center, randomized,
placebo-control study. Fifty-nine patients (59 eyes) with uncontrolled IOP
under maximal tolerated medical treatment (MTMT) were recruited. A primary
trabeculectomy with mitomycin C (MMC) was done and the patients were randomized
to either postoperative subconjunctival injection of bevacizumab (1.25 mg/0.05
mL) or balanced salt solution (BSS). Forty-seven patients (47 eyes) completed
at least one year of follow up and were included in the study. The main outcome
measure was the IOP, and secondary outcome measures include bleb morphology,
vascularization, and fibrosis, as well as the need for glaucoma medications and
5-fluorouracil (5-FU) needling.
RESULTS: At 1-year follow up, there was no significant
difference between groups for IOP (P=0.65), bleb morphology (P=0.65),
and the need for glaucoma medications (P=0.65) or 5-FU needling
requirements (P=0.11). However, the bevacizumab group had a higher rate
of success results, lower use of glaucoma medications after surgery, and
optimal bleb aspect in more patients, but more 5-FU needling procedures required.
CONCLUSION: A bigger sample size is needed in order to
determine whether the differences found in the bevacizumab group are
statistically significant.
KEYWORDS: glaucoma; trabeculectomy; bevacizumab; bleb
DOI:10.18240/ijo.2019.10.08
Citation:
Muhsen S, Compan J, Lai T, Kranemann C, Birt C.
Postoperative adjunctive bevacizumab versus placebo in primary trabeculectomy
surgery for glaucoma. Int J Ophthalmol 2019;12(10):1567-1574
INTRODUCTION
Glaucoma,
remains a significant public health problem as a leading cause of irreversible
visual loss[1]. In a recent systematic review and
Meta-analysis it has been estimated that the overall global prevalence of
glaucoma was 3.54%, with the highest prevalence in Africa, and that the number
of people with glaucoma worldwide (aged 40-80y) will increase from 64.3 million
in the year 2013 to 111.8 million in 2040, disproportionally affecting people
residing in both Asia and Africa[2]. The single
most effective therapeutic option currently availableaims at lowering the
intraocular pressure (IOP) by medical, laser, or surgical techniques[3]. Glaucoma surgery is sought when further IOP lowering
is required despite the use of maximal tolerable medical and indicated laser
therapy. Trabeculectomy with intra-operative use of mitomycin C (MMC) is the
current standard form of surgery performed. Trabeculectomy aims to create a
functioning aqueous drainage channel into a filtering bleb from which aqueous
subsequently flows transconjunctivally or is absorbed into the episcleral and
subconjunctival capillaries. The success of bleb formation is depends on
preventing wound scarring. The events of wound healing after trabeculectomy are
mediated by both fibroblast activity and angiogenesis[4],
which leads to increased vascular permeability, vascularization and fibrosis.
The result of injury-mediated factors is conjunctival and episcleral fibrosis
allowing the closure of the drainage space leading to premature surgery failure[5]. The ability to modulate and reduce these factors in
the immediate postoperative period could lead to better long-term bleb
sustainability. Currently, adjunctive medications which modulate wound healing
such as anti-metabolites like 5-fluorouracil (5-FU) and anti-proliferative
agents such as MMC target fibroblast activity and are effective in curbing the
fibrosis process[6-9]. However,
excessive inhibition of wound healing noted in antimetabolite-augmented
trabeculectomy is frequently linked to complications such as postoperative
infections, hypotony, corneal toxicity, and thin-walled avascular blebs, which
are susceptible to leakage[10-11].
As a result, methods to tone down the wound-healing response with safer
medications are under investigation.
Recent
advances in ophthalmic wound healing research, including vascular endothelial
growth factor (VEGF) inhibitors, antibodies, RNAi, gene therapy, nanoparticles,
liposomes, dendrimers, proteoglycans and small molecule inhibitors are under
development[12]. VEGF plays a central
role in early phases of wound healing, including angiogenesis and vascular
maintenance. Increased vascular permeability along with increased VEGF
concentration has been shown to occur during the early phases of wound repair,
allowing deposition of the fibrin matrix necessary for cellular migration. VEGF
is produced or released locally in surgical wounds and its neutralization
decreases angiogenesis and endothelial cell chemotaxis[13-14].
In light of this understanding of the wound healing
process, the adjunctive use of VEGF inhibitors has been recently attempted in
trabeculectomy. It has been postulated that the use of these selective wound
modulators may enhance surgical efficacy and, at the same time, offer a more
favorable outcome in regards of success and safety[15-17]. Bevacizumab (Avastin; Genetech, Inc,.
Our hypothesis is that adjunctive subconjunctival
single use of bevacizumab in the early postoperative period following
trabeculectomy with MMC can modulate the wound healing and promote healthy
filtering bleb formation. At the same time we postulate that adjunctive use of
bevacizumab could potentially decrease long-term risk of bleb failure and hence
result in sustained lower IOP.
The present study compares the efficacy of a single,
postoperative subconjunctival bevacizumab injection versus placebo, in patients
with different types of glaucoma that underwent a primary trabeculectomy with
MMC.
SUBJECTS
AND METHODS
Ethical Approval
This study was
registered at the Clinical Trials Registry in July, 2010 (NCT01166594). This is
a one center, prospective, randomized, placebo-controlled study, which was
conducted in compliance with the tenets of Declaration of Helsinki and
after approval of the Research Ethics Board of Sunnybrook Health Sciences
Centre. Signed informed consent was obtained from all patients.
Between June 2010 and September 2013, 59 patients
diagnosed with glaucoma and uncontrolled IOP, receiving maximal tolerated
medical therapy (MTMT), and requiring a primary trabeculectomy, were recruited
in the Glaucoma Clinic, Sunnybrook Health Sciences Centre,
The age, sex, race, type of glaucoma, preoperative
best corrected visual acuity (BCVA), IOP measured by calibrated Goldman
applanation tonometer, central corneal thickness (CCT), number of glaucoma medications,
number of previous laser trabeculoplasty treatments, previous cataract surgery,
visual field mean deviation, pattern standard deviation and mean ocular
coherence tomography (OCT) retinal nerve fiber layer (RNFL) thickness were
recorded before surgery.
A standard fornix based trabeculectomy was performed
by a single surgeon (Birt C) at Sunnybrook Health Sciences Centre between July
2010 and January 2013. The MMC exposure time varied between 60 and 150s
depending on surgeon��s judgment of the individual patient��s risk factors (these
primarily included age, race, preoperative and target IOP, use of topical
medications and previous anterior segment surgery). A square 4��
All patients received prednisolone acetate 1% drops,
10 times per day during the first week and approximately 6 times per day during
the second week with slow taper for the next two months postoperatively. A
fourth generation fluoroquinolone was also used 4 times per day for the first
two weeks after surgery. Laser suture lysis was performed at the discretion of
the treating physician.
On postoperative days 1, 7, 14, 30, 60, 90, 180 and
365, the IOP, BCVA, number of glaucoma medications and complications were
recorded. The bleb appearance was formally graded according to the Indiana
Bleb-Grading Scale (IBGS) at each visit[22]. This
scale assesses bleb height, bleb extension, grade of vascularity and leak
presence or absence. The use of 5-FU needling for additional wound modulation
was also recorded. A window of ��7d
was allowed for the 30, 60 and 90d visits and a range of ��14d was allowed for
the 180 and 365d visits.
Success,
Qualified Success, and Failure Criteria Success was defined as postoperative
IOP<
Statistical
Analysis
Past studies measuring IOP post trabeculectomy result in average
standard deviation of
RESULTS
Between June 2010 and September 2013, 59 patients
were recruited. Randomization assigned 30 patients to the bevacizumab group and
29 patients to the placebo group. Five patients withdrew their consent after
surgery, four patients were found to have had a violation of the inclusion
criteria and three patients were lost to follow up. Forty-seven patients
completed at least one year of follow up, 23 patients in group A (Avastin) and
24 patients in group B (BSS). The demographic characteristics of the study
patients are summarized in Table 1.
Table 1 Demographics characteristics of the study
patients n
(%)
|
Parameters |
Group A (n=23) |
Group B (n=24) |
P |
|
Mean age (y) |
63.7��10.8 |
61.3��10.4 |
0.86 |
|
Range |
36 to 85 |
29 to 76 |
|
|
Sex |
|
|
|
|
Male |
9 (19.1) |
13 (27.7) |
0.36 |
|
Female |
14 (29.8) |
11 (23.4) |
|
|
Race |
|
|
|
|
White |
15 (31.9) |
17 (36.1) |
0.22 |
|
Black |
6 (12.7) |
3 (6.3) |
|
|
Asian |
0 (0.0) |
3 (6.3) |
|
|
South Asian |
2 (4.2) |
1 (2.1) |
|
|
Type of glaucoma |
|
|
|
|
POAG |
19 (40.4) |
11 (23.4) |
0.03 |
|
PXFG |
2 (4.2) |
7 (14.9) |
|
|
Other |
2 (4.2) |
6 (12.8) |
|
The groups were similar and comparable. No
statistically significant differences were found at baseline other than a
higher number of patients diagnosed with primary open angle glaucoma (POAG)
present in the bevacizumab group.
Baseline clinical characteristics are shown in Table
2. No statistically significant differences were found between groups regarding
age, visual acuity, IOP, CCT, number of glaucoma medications, 24-2 visual field
parameters, OCT RNFL thickness, number of previous laser trabeculoplasty
treatments, and the number of patients with previous cataract surgery.
Table 2 Baseline group comparison
mean��SD
|
Parameters |
Group A (n=23) |
Group B (n=24) |
P |
|
Mean age (y) |
63.7��10.8 |
61.3��10.4 |
0.86 |
|
VA (logMAR) |
0.2��0.3 |
0.4��0.7 |
0.45 |
|
IOP (mm Hg, range) |
23.2��7.2 (15-40) |
25.7��9.2 (14-44) |
0.26 |
|
CCT |
540��38 |
552��39 |
0.30 |
|
Meds (range) |
3.3��0.9 (1-5) |
3.5��1.0 (2-6) |
0.65 |
|
MD (dB, range) |
-10.3 (-0.9 to -24.5) |
-14.9 (-2.1 to -30.5) |
0.34 |
|
PSD (dB, range) |
7.53 (1.96-13.51) |
8.78 (1.18-13.44) |
0.25 |
|
OCT RNFL thickness |
64.2��17.9 |
64.9��20.6 |
0.54 |
|
Previous trabeculoplasty |
|
|
|
|
No. of patients
(%) |
6 (26) |
4 (16.6) |
0.67 |
|
Previous cataract surgery |
|
|
|
|
No. of patients
(%) |
1 (4.3) |
2 (8.3) |
1.00 |
VA: Visual acuity; CCT: Central corneal thickness;
Meds: No. of medications; MD: Mean deviation; PSD: Pattern standard deviation;
OCT RNFL: Ocular coherence tomography retinal nerve fiber layer overall
thickness.
The preoperative IOP was 23.2��
Both groups had a similar drop in number of glaucoma
medications required (3.3 to

Figure 1 5-FU Needling group comparison at 1-year
follow-up.
Postoperative complications included bleb leaks,
hypotony, choroidal detachments and others. During the first 2wk after surgery
8 patients (17%) presented with a bleb leak in group A and 7 patients (14.8%)
in group B. It was resolved using one or more of the following techniques: 48h
eye patch, bandage contact lens, additional suture to the wound, or full bleb
revision. Only one patient (group B) presented a late bleb leak (1-year visit)
that required a bleb revision. No statistically significant differences were
found between groups comparing bleb leak rates (P=0.92). The 14 patients
(29.8%) developed hypotony during the 1-year postoperative follow up in group A
and 11 patients (23.4%) in group B (P=0.46). Six patients (12.8%)
presented with choroidal detachments in group A and 4 patients (8.5%) in group
B (P=0.67). Six patients (12.8%) developed other postoperative
complications including 3 cases of anterior chamber shallowing that required
reformation, 2 cases of hyphema and 1 case of iris blocking sclerostomy in
group A. In group B, 7 patients (14.8%) presented with other complications
including 2 cases of suprachoroidal hemorrhage, 1 case of branch retinal vein
occlusion, 1 case of corneal abrasion, 1 case of anterior chamber shallowing
that required reformation and 2 cases of hyphema (P=1.00; Table 3).
Table 3 Postoperative complications at 1-year follow
up n (%)
|
Complications |
Group A (n=23) |
Group B (n=24) |
P |
|
Bleb leak |
8 (17) |
7 (14.8) |
0.92 |
|
Hypotony |
14 (29.8) |
11 (23.4) |
0.46 |
|
Choroidal detachment |
6 (12.8) |
4 (8.5) |
0.67 |
|
Other |
6 (12.8) |
7 (14.8) |
1.00 |
The preoperative BCVA was 0.2��
The preoperative mean deviation in 24-2 Humphrey
visual field was -10.3��
The mean OCT RNFL thickness was 64.2��
Table 4 Group comparison at baseline and 1-year
follow up mean��SD
|
Baseline and 1-year group comparison |
Group A (baseline) |
Group B (baseline) |
Group A (1-year) |
Group B (1-year) |
P
(baseline to 1-year) |
|
VA (logMAR) |
0.2��0.3 |
0.4��0.7 |
0.66��1.8 |
0.47��0.66 |
0.65 |
|
IOP (mm Hg, range) |
23.2��7.2 (15-40) |
25.7��9.2(14-44) |
13.8��4.5 (8-24) |
13.2��5.1 (4-24) |
0.34 |
|
Meds (range) |
3.3��0.9 (1-5) |
3.5��1.0 (2-6) |
0.4��1.1 (0-4) |
0.5��1.1 (0-5) |
0.65 |
|
MD (dB, range) |
-10.3 (-0.9 to -24.5) |
-14.9 (-2.1 to -30.5) |
-10.4 (-2.1 to -23.5) |
-13.5 (-1.6 to -30.2) |
0.32 |
|
PSD (dB, range) |
7.53 (1.96 to 13.51) |
8.78 (1.18 to 13.44) |
8.74 (1.27 to 19.01) |
7.89 (2.4 to 13.12) |
0.56 |
|
OCT RNFL thickness |
64.2��17.9 |
64.9��20.6 |
74.6��21.1 |
70.9��16.1 |
0.60 |
At 1-year follow-up in group A, 14 patients (29.8%)
were considered a success and 9 patients (19.1%) a qualified success. No
clinical failures were noticed in this group. In group B there was 12 cases of
success (25.5%), 10 cases of qualified success (21.3%) and 2 cases of failure
(4.2%). Both failure patients developed a suprachoroidal hemorrhage that
required drainage and a pars plana vitrectomy. The first case developed late
corneal decompensation and the second case developed chronic hypotony after
vitrectomy. No significant differences were found between groups in terms of
qualified success (P=0.87) or failure (P=0.16).
Kaplan-Meier survival analysis was used to compare
the time to qualified success or to failure between the 2 treatment groups
(Figures 2 and 3).
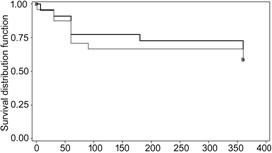
Figure 2 Kaplan Meyer survival curve for qualified
success at 1-year follow-up.
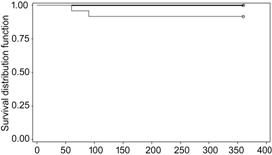
Figure 3 Kaplan Meier survival curve for failure at
1-year follow-up.
DISCUSSION
Glaucoma filtration surgeries that function by
draining aqueous humor to the sub-Tenon��s space, including trabeculectomy, some
varieties of nonpenetrating surgery, and glaucoma drainage devices, rely upon
the external resistance created by wound healing to prevent hypotony. However,
the sustainability of success of the surgical procedure depends on maintaining
the patency of the fistula tract created during surgery. Failure of the
procedure is usually the result of conjunctival scarring to the episclera
leading to aqueous outflow restriction. Histologic studies have demonstrated
that the utmost proliferation of subconjunctival fibroblasts happens between
the third and fifth surgical day[19].
Pharmacologic enhancement of trabeculectomy using
either 5-FU or MMC has improved rates of trabeculectomy success considerably.
However, the nonspecific mechanism of action of those agents might lead to
widespread cell death and thin-walled avascular blebs that are vulnerable to
leak[20-21], infection[24-25], and dysaesthesia[26]. With lower levels of exposure, the frequency of
bleb-related complications is less, however episcleral fibrosis and bleb
failure is more likely to occur.
The search for selective wound modulators that have
an acceptable risk profile and can replace the current antifibrotics that are
notorious for their nonspecific inhibitory activity and continued risk of
failure has been underway for the last few years. VEGF plays a pivotal role in
both physiological and pathological angiogenesis throughout the body. Different
VEGF isoforms may be responsible for different roles in the process of ocular wound healing. van Bergen et
al[27] studied the presence of VEGF in
trabeculectomy blebs and reported VEGF (165) and VEGF (121) to be mainly
involved in angiogenesis, and VEGF (189) to be predominantly responsible for
fibrous tissue proliferation. Lopilly Park et al[28]
examined the levels of VEGF in both the aqueous humor and Tenon��s tissue
in eyes with POAG and compared their levels with the surgical outcome. They
identified a significant correlation between the VEGF level in Tenon��s tissue
at the time of filtration surgery with the 1-year IOP and the final success of
the surgery. Therefore by downgrading angiogenesis, resultant inflammation and
collagen buildup, VEGF inhibition may have a favorable effect on the outcomes
of filtration surgery[29-30].
In 2009, Li et al[31]
reported a rise in aqueous humor VEGF protein concentration post trabeculectomy
in rabbits, which was inhibited partially for up to 6d by a single
subconjunctival and intracameral injection of bevacizumab at the time of
surgery. No substantial advantage was observed in terms of IOP lowering up to
29d post trabeculectomy, however larger bleb area was documented using the
Moorfields bleb classification in eyes receiving bevacizumab. One year later,
Miyake et al[32] reported that
intravitreal injection of 1.25 mg bevacizumab in 3 macaque monkey eyes resulted
in deceased aqueous VEGF concentrations 1 to 28d after injection. They also
noted that this decrease was maintained until 42d after injection. Memarzadeh et
al[33] randomized 42 rabbits to trabeculectomy
using a more intensive regimen of 7 subconjunctival injections of 1.25 mg
bevacizumab, 5 mg 5-FU, or 0.1 mL BSS, given during the first 14 postoperative
days. They documented no differences in mean IOP during the study period, but
bevacizumab significantly improved bleb survival and resulted in less scarring
in comparison with the 5-FU and BSS groups. Similarly, How et al[34] reported 100% bleb survival in the rabbit model at
28d when subconjunctival bevacizumab (2.5 mg) was combined with 5-FU (5 mg),
50% in the bevacizumab only group, 25% in the 5-FU-only group, and
A limitation of animal studies of glaucoma filtration
surgery in general was the difficulty in getting demonstrable IOP differences
as primary outcomes, necessitating the use of bleb morphologic features
instead.
Grewal et al[35]
published a prospective, non-randomized small case series in 2008, analyzing
the postoperative effect of a single subconjunctival dose of bevacizumab in
patients who underwent a primary trabeculectomy without MMC or 5-FU. All eyes
had an IOP reduction of 52% after 6mo. However, the authors noticed increases
in bleb vascularity 3mo after surgery.
Kahook[36] published a
prospective, randomized, case control study in 2010, comparing the use of a
single intravitreal injection of bevacizumab versus placebo in 10 patients who
had a primary trabeculectomy with MMC. At 6mo follow-up, there were no
differences in terms of success or IOP, however, the authors mentioned more
diffuse and less vascularized blebs in the bevacizumab group. In 2011,
Sedghipour et al[37] published a
prospective, randomized, placebo controlled trial comparing the use of single
subconjunctival dose of bevacizumab (0.2 mg) vs placebo (BSS) in 37
patients with glaucoma that had a primary trabeculectomy without MMC, finding
no differences between groups in terms of IOP after 3mo follow-up. During the
same year, Ghanem[38] published a similar study
including 55 patients comparing the single use of subconjunctival bevacizumab
(1.25 mg/0.05 mL) versus placebo (BSS) in patients that had a primary
trabeculectomy with MMC. Similar to the findings of Sedghipour et al[37], they didn��t find differences in IOP or success rates
at 1-year follow up, however the reduction in vascularity of the filtering bleb
was statistically significant in the bevacizumab group. In 2014, Vandewalle et al[39] published a prospective, randomized, placebo
controlled study comparing the use of a single dose of intracameral bevacizumab
(1.25 mg/0.05 mL) versus placebo (BSS) in patients with open angle glaucoma
scheduled for a primary trabeculectomy with or without MMC. A total of 138
patients were analyzed one year after surgery and no statistically significant
differences in IOP were noticed between groups, however absolute success was
reached in more patients in the bevacizumab group and also the need for
needling was lower in this group. Kiddee et al[40]
in October 2015, published a prospective, randomized, placebo-control study
comparing the single use of subconjunctival bevacizumab (1.25 mg/0.05 mL)
versus placebo (BSS) in patients with POAG that had a primary trabeculectomy
with MMC. The authors analyzed a total of 39 patients, finding no statistically
significant differences in IOP reduction or success rates between groups after
1-year follow up. The bleb vascularity score was significantly lower in
bevacizumab during the first month but the difference was not sustained
throughout the follow-up period.
In 2016, Fakhraie et al[41]
published a prospective, randomized, placebo-controlled study using a different
technique. They compared a single dose of intracameral bevacizumab (1.25
mg/0.05 mL) versus placebo (BSS) at the end of a primary trabeculectomy without
MMC in patients with open angle glaucoma who had a primary trabeculectomy. The
authors intentionally included only patients who were 65 years of age or more.
Sixty-five patients completed a six months follow-up and were analyzed. They
observed that bevacizumab significantly improved the short-term success rate in
these patients and the rate of encapsulated bleb formation was clinically more
significant in placebo group but it didn��t reach a statistically significant
difference. On the other hand, early filtering bleb leakage was significantly
more common in the bevacizumab group. The authors concluded that bevacizumab
could be as effective as MMC or 5-FU preventing bleb failure in primary
trabeculectomy; however bleb morphology was not analyzed and long term results
have not been published so far. On the other hand, previous studies that
compared the use of 5-FU or MMC to subconjunctival bevacizumab in primary
trabeculectomy found lower rates of success when bevacizumab was used as single
agent[42-44].
Our study is, so far, the largest prospective,
randomized, placebo-control study published comparing the adjunctive use of a
standard dose of subconjunctival bevacizumab (1.25 mg/0.5 mL) in primary
trabeculectomy surgery using MMC. Previous prospective and randomized studies analyzed
smaller groups of patients[40], included
refractory glaucomas [neovascular, post-penetrating keratoplasty (PKP),
uveitis, post-vitrectomy] tested lower dose of bevacizumab[37], or used different routes of administration[39-41].
In our study no statistically significant differences
were found at 1-year follow up in terms of IOP between bevacizumab and control
group. The bleb aspect was optimal in more patients in the bevacizumab group,
however this was not statistically significant and not related to a lower
intraocular pressure at 1 year-follow-up. Although the bevacizumab group had a
tendency to have more successful results and a lower use of glaucoma
medications after surgery, this group of patients also required more 5-FU
needling procedures. None of these differences, however, were statistically
significant.
The fact that we didn��t find any statistically
significant difference between groups despite the tendency to get a better
quality blebs and more success results in the bevacizumab group could be
explained by our sample size. This study was originally powered for 60 subjects
for a 95% of significance level. Unfortunately although 59 patients were
randomized, for various reasons only 47 patients completed the follow up and
were analyzed. The lower power obtained due to the loss of subjects is a
weakness of our paper. A bigger sample size that fulfilled the requirements for
a fully powered study is needed in order to determine whether the differences
found between groups are in fact statistically significant.
The fact that several other researchers have reported
similar results with little difference between treatment and placebo groups,
however, suggests the possibility that the BSS injected subconjunctivally for
placebo group may have an active effect on surgery outcomes. The saline bolus
may help in bleb formation, either by breaking fibrin adhesions and/or diluting
adverse growth factors within the bleb. We did not include a sham injection
control group, but this might be a good idea for future studies.
Furthermore, the pharmacokinetics of subconjunctival
bevacizumab have not been well studied[45]. A
single dose of 1.25 mg intravitreal bevacizumab is likely to result in complete
intravitreal VEGF blockade for 4 to 6wk[19,46]. The fact that no statistically
significant differences between the bevacizumab and placebo groups were found
with a single dose of sub-conjunctival bevacizumab, may justify future trials
using repeated injections of bevacizumab or a comparison of bevacizumab vs
5-FU in patients with severely vascularized blebs in the early postoperative
period after trabeculectomy.
Of importance, the optimal route of administration
and dosing frequency are still undetermined for bevacizumab[45-49]. Surprisingly, results from animal studies suggest
that there is not a major advantage for intravitreal use over subconjunctival.
Intravitreal administration reaches higher concentrations inside the eye,
although there is some evidence that subconjunctival injection may result in
high tissue levels for periods as long as those associated with intravitreal
injection[45].
The utilization of bevacizumab in trabeculectomy is
an off-label treatment, and several issues need to be addressed, such as the
best administration route (intravitreal, anterior chamber or subconjunctival),
duration of action, dosage and toxicity. In this study, the complication rate
was comparable in both groups for bleb leak, hypotony and choroidal
detachments, and no systemic side effects were reported. The goal of modulating
wound healing to provide safe and effective IOP control in our surgical
patients�� remains highly desirable, and anti-VEGF antibody treatment, such as
with bevacizumab continues to be a possible addition to our armamentarium in
this regard. Further work exploring the options available for treatment is
indicated.
ACKNOWLEDGEMENTS
This study was presented as a poster at the World
Glaucoma Congress 2019.
This study was presented as an abstract at ARVO
annual meeting in April, 2014.
Foundation: Supported by the Glaucoma Research Society of
Conflicts of Interest: Muhsen S, None; Compan J, None; Lai T, None;
Kranemann C, None; Birt C, None.
REFERENCES