
Citation: Limratchatamorn
B, Asakawa K, Mashimo K, Uga S, Ishikawa H. Effects of 0.4% ripasudil
hydrochloride hydrate on morphological changes in rabbit eyes. Int
J Ophthalmol 2019;12(10):1637-1642. DOI:10.18240/ijo.2019.10.18
・Brief Report・
Effects
of 0.4% ripasudil hydrochloride hydrate on morphological changes in rabbit eyes
Bundit Limratchatamorn1,2, Ken Asakawa3,
Kimiyo Mashimo2, Shigekazu Uga2, Hitoshi Ishikawa2,3
1Department of Ophthalmology, Priest
Hospital, Department of Medical Services, Ministry of Public Health, Bangkok
10400, Thailand
2Department of Ophthalmology, School
of Medicine, Kitasato University, Kanagawa 252-0374, Japan
3Department of Orthoptics and Visual
Science, School of Allied Health Sciences, Kitasato University, Kanagawa
252-0373, Japan
Correspondence to: Bundit Limratchatamorn. Department
of Ophthalmology, Priest Hospital, Department of Medical Services, Ministry of
Public Health, 445 Si Ayutthaya Road, Thung Phayathai, Ratchathewi, Bangkok
10400, Thailand. siumra060rey609@gmail.com
Received:
Abstract
We evaluated the cellular
structure changes after continuous use of ripasudil hydrochloride hydrate in
rabbit eyes which might affect its own efficacy and adverse effects. Two
pigmented Dutch rabbits and 1 Japanese white rabbit were instilled with 0.4%
ripasudil hydrochloride hydrate to the left eye twice daily. The right eye was
observed as the control. Both eyes of all 3 rabbits were then enucleated for
histopathologic examination by light and electron microscope at 1mo in 1 of the
pigmented Dutch rabbits, 3mo in the other pigmented Dutch rabbit, and in the
Japanese white rabbit after instillation. Microscopic observations showed
increase intercellular space in trabecular meshwork, ciliary body, and iris
stoma, increase pigmented granule number and size in iris epithelial cells, and
decrease actin filament in iris muscle fiber cells. Consequently, ripasudil
hydrochloride hydrate decreases the intraocular pressure by improving the
conventional outflow and may also facilitate the unconventional outflow via
intercellular space widening without serious side effects.
KEYWORDS: ripasudil
hydrochloride hydrate; actin filament; side effect; rabbit
DOI:10.18240/ijo.2019.10.18
Citation: Limratchatamorn
B, Asakawa K, Mashimo K, Uga S, Ishikawa H. Effects of 0.4% ripasudil
hydrochloride hydrate on morphological changes in rabbit eyes. Int
J Ophthalmol 2019;12(10):1637-1642
INTRODUCTION
Ripasudil (K-115) is a Rho kinase
(ROCK) inhibitor that is commercially available to treat open angle glaucoma in
Japan[1-2]. ROCKs are
serine/threonine kinase that regulates smooth muscle contraction. In mammals,
ROCKs exist in 2 isoforms (ROCK1 and ROCK2) which being expressed in many
tissues, including human trabeculocytes and ciliary muscle cells[3]. ROCK inhibitors inhibit myosin light chain phosphatase
resulting in actomyosin-based cellular relaxation in many types of cells
including trabeculocytes, ciliary muscle cells, and vascular endothelium[4]. The levels of mRNAs for ROCK and ROCK substrates are
higher in trabeculocytes compared to ciliary muscle cells. Trabeculocytes are
also more sensitive to ROCK inhibitors than ciliary muscle cells[4]. The exact molecular mechanisms of the conventional
aqueous outflow improvement are not well understood, but it has been
hypothesized that cellular relaxation and intercellular adhesion relaxation
cause widening empty spaces in the juxtacanalicular region and increased
vacuoles in endothelial cells improve outflow volume[5].
Previous animal model studies also found that ROCK inhibitors may improve blood
flow to the optic nerve[6], increase ganglion cell
survival[7], and reduce bleb scarring in glaucoma
surgery[8].
Ripasudil has a good ocular
penetration property, first described by Isobe et al[9]
using radioactive distribution assay. Ripasudil showed high intraocular
penetration through the transcorneal route within 15min after instillation with
very low transcleral and systemic absorption. The radioactive distribution
assay showed the drug concentration in the anterior chamber and periocular soft
tissue in 15min, but only a little was detected in the posterior retina,
choroid, and extraocular tissue around the optic nerve without contralateral
drug absorption. Due to the drug distribution property, which mainly affects
the anterior chamber structure, myosin light chain containing cell changes in
the anterior segment including trabeculocytes, vascular endothelium,
epithelium, fibroblasts, and smooth muscle in these regions might have been
affected by this medication. Ripasudil can be used to treat open angle glaucoma
as primary or adjuvant therapy[10-11].
However, to our knowledge, no
previous investigators reported ultrastructural changes in an in vivo
study. A morphologic study may not only define the exact molecular mechanisms
of ripasudil, it may also postulate future side effects of this medication. The
purpose of this study is to evaluate the cellular structure changes after
continuous use of ripasudil in rabbit eyes, and to investigate its efficacy and
adverse effects.
MATERIALS AND METHODS
Ethical Approval All experiments were conducted under
proper anesthesia. We considered ethical issues and paid careful attention to
minimizing pain. All experiments were also performed according to the ARVO
Statement for the Use of Animals in Ophthalmic and Vision Research and with the
approval of the animal experiment Ethics Committee of Kitasato University
(No.2015-176).
Animals The experiments were performed on 6
eyes of 3 rabbits (2 male Dutch rabbits and 1 male Japanese white rabbit). They
were purchased at 8 weeks of age and raised in a temperature- and
humidity-controlled environment (25ºC and 60%, respectively) until 40 weeks
old, weight 2.0, 1.9, and
After 1mo of continuous 0.4%
ripasudil instillation to the left eye of all 3 rabbits, the first pigmented
Dutch rabbit was euthanized with an overdose intravenous injection of
pentobarbital sodium. The 2 remaining rabbits were instilled continuously with
ripasudil for 3mo. After 3mo of the experiment, the remaining 2 rabbits were
euthanized in the same manner. The eyeballs were immediately enucleated for the
histological examinations.
Light and Electron Microscopy The enucleated eyes were fixed
overnight in 2.5% glutaraldehyde +0.1 mol/L phosphate-buffered saline (PBS).
The enucleated eyeballs were grossly examined before horizontally incised near
equator. Vitreous body and lens were removed. Corneal ring was excised
circumferentially
RESULTS
Microscopy of Trabecular
Meshwork Comparing the control eyes (Figure
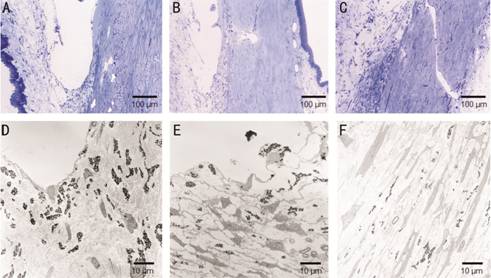
Figure 1 Trabecular meshwork A, D: Control; B, E: 1mo after
instillation; C, F: 3mo after instillation. The trabecular meshwork shows
increase intercellular spaces between the trabeculocytes after ripasudil
instillation.
Microscopy of Ciliary Body and
Iris Comparing the control eyes (Figure
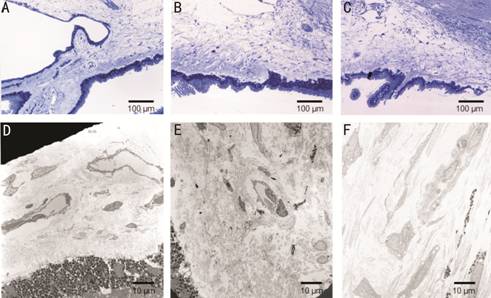
Figure 2 Ciliary body A, D: Control; B, E: 1mo after
instillation; C, F: 3mo after instillation. The interstitial space is widening
after ripasudil instillation.
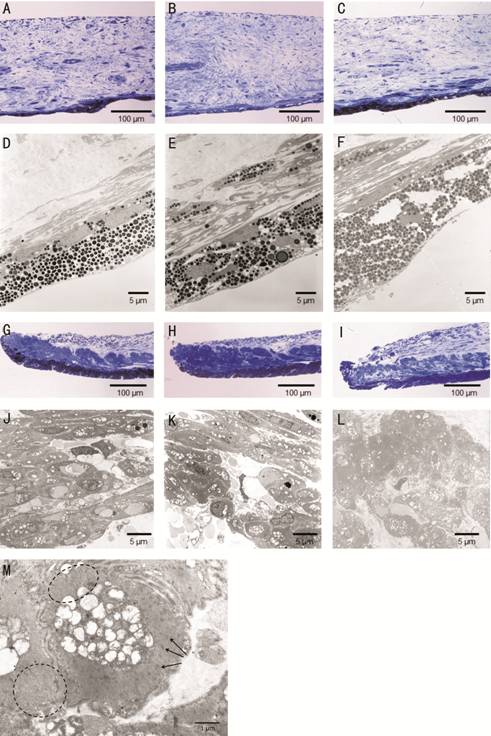
Figure 3 Iris A, D, G, J: Control; B, E, H, K: 1mo
after instillation; C, F, I, L: 3mo after instillation. After ripasudil instillation, the
stroma (B, E, C, F) and muscles (H, K, I, L) show increased
intercellular spaces between the cells. The muscle fiber bundles are thin (K,
L). Actin filaments are diminished (black dot circles). Z-bands (dense bodies)
are observed (black arrows, M).
DISCUSSION
In the microscopic findings of the
trabecular meshwork, the space between cells also widened after prolonged
exposure to ripasudil over the time period of the present study. Moreover,
thinning of the iris muscle fiber and loss of actin filament were detected.
After 3mo of ripasudil instillation, subclinical histological changes were
detected without the functional disturbance.
In rabbit eyes, the anterior chamber
has no true trabecula, and the vascular anatomy of the outflow pathways differs
from that of primates[12]. Rabbits do not have a
true Schlemm’s canal or collecting system as do primates. Many small
intrascleral vessels (the trabecular plexus) may play an important role in the
aqueous drainage system in rabbits. This vascular plexus becomes the densest
adjacent to the trabecular meshwork, and drains the aqueous humor through the
perilimbal blood vessels. In all mammals except primates, an iris pillar which
originates from the anterior surface of the iris to the posterior surface of
the corneoscleral junction, forms the completely enclosed angle called the
space of Fontana which further develops to Schlemm’s canal as a result of the
huge development of the ciliary muscle. The actin cytoskeleton modifying
medication is known to inhibit the myosin light chain phosphorylation processes
causing changes in cell shape, cell-cell adhesion, and cell-matrix adhesion[13].
The vascular plexus in the sclera
also increases in vascular diameter with increased vacuolization in the
vascular endothelium and the trabecular epithelium. These results confirm the
cellular structure changes in the trabecular meshwork, which promotes the
conventional outflow rate. Not only increasing the aqueous flow through the
trabecular system, ripasudil also improves the permeability of the chamber
angle trabecular plexus and the iris and ciliary body vasculature. The outflow
facility increased 2 and 2.2 times from baseline as a result of the Isobe et
al[9] and Honjo et al[14] experiments which were confirmed previously by the
two-level constant-pressure perfusion method.
The unconventional outflow is
comprised of uveoscleral and uveovortex outflows. The uveoscleral outflow
drains the aqueous humor through the iris root and the anterior surface of the
ciliary body then reaches the suprachoroidal space that drains into the episcleral
vein. The uveovortex outflow drains the fluid into the iris vessel and the
vortex veins. Because of the lower hydrostatic pressure in the suprachoroidal
space than that in the anterior chamber, it causes the driving force of the
aqueous to the uveoscleral outflow. These unconventional outflows depend on the
net osmotic resorption of aqueous humor into the uveal venous circulation,
extracellular matrix spaces, and episcleral venous pressure[15].
The results of our study showed obviously widening of the space between cells
in the trabecular meshwork cells, ciliary body, and iris stoma. The relaxation
of the vascular endothelium might improve vascular permeability and blood flow
to the target organ[16].
As previously described, the
unconventional outflow was determined by the fluid flow through the space
between cells, drain into the vessel in the iris stroma and the suprachoroidal
space. Continuous ripasudil instillation which resulted in myosin light chain-based
cellular relaxation, space between cell-cell and cell-matrix become widened,
might cause an increase in the unconventional outflow. The uveoscleral outflow
was measured with a perfusion technique using fluorescein
isothiocyanate-dextran in studies by Isobe et al[9]
and Honjo et al[14] in which they reported
non-significant increases from the baseline of 30% and 15%, respectively.
Because these studies were performed after one instillation of a ROCK
inhibitor, the ultrastructural change, which may prove to increase the
unconventional outflow in the present study, occurred after continuous use of
the ripasudil for at least 1mo. The unconventional outflow measuring method
would be required to confirm this result after 1mo instillation of ripasudil.
The main cause of failure of
glaucoma filtration surgery is postoperative subconjunctival scarring in the
filtration bleb. There is an evident base indicating that human Tenon’s
fibroblasts from subconjunctival space play a key role in the scarring process via
the proliferation, migration, and contraction processes. The
transdifferentiation of fibroblasts into myofibroblasts is a crucial step in
wound healing and scar formation[17]. Myofibroblasts
are responsible for fibrosis via increased extracellular
matrixsynthesis, granulation tissue formation, and wound contraction[18]. A ROCK inhibitor, as an actin cytoskeleton
inhibitor, functions as a potent antiscarring agent by inhibiting the
transdifferentiation of Tenon’s fibroblasts into myofibroblasts, improving
cellular migration, and inhibiting cell contraction during the wound healing
process[8]. The results of the present in vivo
study revealed that the prolonged exposure to ripasudil did not affect the
stromal fibroblast structure. Eventhough the previous studies confirmed the
antiscarring effect of ripasudil, no structural confirmationwas made from this
study.
The other ultrastructural changes
found in the present study might indicate the potentially subclinical adverse
effects in the future after long-term clinical use of ripasudil, such as an
increase in the size of melanin granules in the iris pigment epithelium, iris
stromal cells, and the anterior layer of melanocytes. Latanoprost-induced iris
darkening (LIID), the most common ocular side effect of latanoprost, was
initially speculated to arise from either a proliferation of melanocytes in the
iris stroma or an increase in melanin granule numbers within stromal
melanocytes. The morphological findings in LIID patients’ peripheral iris
tissue obtained after trabeculectomy, showed significant change in the sizes of
the intracellular melanin granules without change in the number of the granules
or cellular proliferations. This effect was largely found in the anterior
border rather than in the deep stroma[19]. As
seen in the present study, continuous use of ripasudil might cause iris
darkening in the future.
Regarding the experimental
procedures, some limitations need to be acknowledged. First, there is 2 eyes of
only 1 animal sample in the group of 1 and 3mo, respectively. Second, we did
not measure intraocular pressure during experiment. Thus, the relationship
between morphological changes and intraocular pressure still unknown. In the
future, these evaluations should be included to confirm this limitation.
However, this limitation does not affect our conclusions, because the
histopathological differences in the trabecular meshwork, the ciliary body, and
the iris were obvious. Inoue et al[20]
showed significantly intraocular pressure lowering effect of ripasudil after 1
and 3mo after treatment which corresponding to morphological change found in
this study.
In conclusion, ripasudil, as a
potent ROCK inhibitor, inhibits myosin light chain containing cells such as
epithelium, endothelium, fibroblasts, and smooth muscle cells in the anterior
segment resulting in cellular relaxation, and increases intercellular space and
cellular permeability.
ACKNOWLEDGEMENTS
The authors thank Mr. Robert E.
Brandt of MedEd Japan, for editing the manuscript.
Foundation: Supported by Kitasato University
Research Grant for Young Researchers (2018).
Conflicts of Interest:
Limratchatamorn B, None; Asakawa K, None; Mashimo K, None; Uga S, None;
Ishikawa H, None.
REFERENCES