
Citation: Krysik K,
Lyssek-Boron A, Janiszewska-Bil D, Wylegala E, Dobrowolski D. Impact of
ultrasound and optical biometry on refractive outcomes of cataract surgery
after penetrating keratoplasty in keratoconus. Int J Ophthalmol
2019;12(6):949-953
DOI:10.18240/ijo.2019.06.11
・Clinical Research・
Impact of ultrasound and optical biometry on refractive
outcomes of cataract surgery after penetrating keratoplasty in keratoconus
Katarzyna
Krysik1, Anita Lyssek-Boron1, Dominika Janiszewska-Bil1,2,
Edward Wylegala2,3,4, Dariusz Dobrowolski1,2,3
1Department of Ophthalmology with
Pediatric Unit, St. Barbara Hospital, Trauma Centre, Sosnowiec 41-200, Poland
2Department of Ophthalmology,
District Railway Hospital, Katowice 40-760, Poland
3Chair and Clinical Department of
Ophthalmology, School of Medicine with the Division of Dentistry in Zabrze,
Medical University of Silesia in Katowice, Katowice 40-760, Poland
4Hebei Provincial Eye Hospital,
Xingtai 054001, Hebei
Province, China
Correspondence to: Dariusz
Dobrowolski. Chair and
Clinical Department of Ophthalmology, School of Medicine with the Division of
Dentistry in Zabrze, Medical University of Silesia, District Railway Hospital,
Panewnicka 65 str., Katowice 40760, Poland.
dardobmd@wp.pl
Received:
Abstract
AIM: To
analyse the impact of ultrasound and optical intraocular lens (IOL) calculation
methods on refractive outcomes of cataract phacoemulsification performed after
penetrating keratoplasty (PK) in keratoconus.
METHODS:
Phacoemulsification cataract surgery was performed on 42 eyes of 34 patients
with keratoconus who had previously undergone PK. The IOL power was determined
by using both standard and corneal topography-derived keratometry using the
SRK/T formula. We used two independent methods-ultrasound
biometry (UB) and
interferometry [optical
biometry (OB)] for IOL
calculation. The analysed data from medical records included demographics,
medical history, best corrected visual acuity (BCVA) on Snellen
charts, technique of IOL calculation and calculation formula and its impact on
final refractive result.
RESULTS: BCVA
ranged from 0.01 to 0.4 (mean 0.09±0.19) before surgery and ranged
from 0.2 to 0.7 (mean 0.38±0.14) at 1mo and from 0.2 to 1.0 (mean 0.56±0.16) (P<0.05)
at 3mo, postoperatively. The refractive aim differed significantly from the
refractive outcome in both the UB and OB groups (P<0.05). There was no statistically significant
difference in the accuracy of the two biometry methods.
CONCLUSION:
The refractive aim in keratoconus eyes post-PK is
not achieved with either ultrasound or OB.
KEYWORDS: ultrasound
biometry; optical biometry; cataract surgery; penetrating keratoplasty;
keratoconus
DOI:10.18240/ijo.2019.06.11
Citation: Krysik K, Lyssek-Boron A, Janiszewska-Bil D, Wylegala E,
Dobrowolski D. Impact of ultrasound and optical biometry on refractive outcomes
of cataract surgery after penetrating keratoplasty in keratoconus. Int J
Ophthalmol 2019;12(6):949-953
Outline
Keratoconus is an ectatic non-inflammatory corneal
disorder characterized by central or paracentral thinning and protrusion of the
cornea, resulting in irregular astigmatism[1].
This condition is usually bilateral and diagnosed in the second or third decade
of life[2]. Because of its progressive character, correction
of the variable refractive error and irreversible scarring of the corneal
tissue often demand lamellar or penetrating keratoplasty (PK) as refractive
treatment. PK for advanced keratoconus remains the surgery of choice, but the
type of keratoplasty depends on the corneal structure, the individual patient’s
needs and the surgeon’s experience[3]. In eyes
without stromal scars, the lamellar approach is a method of choice.
Knowledge of cornea stromal ultrastructure and its
biomechanics can help explain and predict post-cataract wound healing. The
anterior and peripheral stroma in keratoconus are more rigid than the posterior
ones, and the interlamellar strength profile of the collagen lamellae is
significantly weaker inferiorly and centrally. Recurrent keratoconus is related
to the incomplete excision of the cone. Healing of the wounds in the peripheral
cornea, where surgical cuts are performed during cataract surgery, may be
difficult and unpredictable[4].
Cataract surgery in keratoconic eyes, especially
after keratoplasty, is frequently challenging. Posterior chamber intraocular
lens (PC IOL) power calculation is less predictable than in eyes without prior
corneal surgery[5]. The choice of an accurate
calculation formula is more difficult and demanding, especially when
considering a low postoperative refractive error and rising patient
expectations. With the improvement in surgical techniques and biometry devices,
cataract surgery is now considered a form of refractive surgery. Accurate
preoperative intraocular lens (IOL) power calculation is crucial in achieving satisfactory
results[6]. IOL power calculation formulae are
good for predicting the postoperative refractive status in eyes with normal
axial length and with no prior ocular surgery[7].
The sequential procedure seems to be more accurate when calculating the IOL
power compared to the triple procedure (cataract removal, IOL implantation and
PK). The triple procedure allows for faster visual rehabilitation but may pose
a higher risk of postoperative intraocular infection[8-9].
The accuracy of biometric measurements is higher for
optical methods than for ultrasonic methods. In ultrasound biometry (UB), there are
more operator-dependent factors that are not present with optical methods[10]. The development of optical devices indicates that UB
will be used only given specific indications. Unfortunately, most authors of
the available papers analysing post-keratoplasty procedures have focused on the
refractive result and visual acuity rather than comparing the planned and
obtained results.
UB remains the preferred method for IOL calculation
in dense cataracts. In regular corneas, standard keratometry and computed
corneal topography accurately measure central corneal power. In
post-keratoplasty corneas, the average central corneal power is more secure and
stable than in topography-derived keratometry. This may improve the accuracy of
the IOL calculation. In our study, keratometry was achieved using swept-source
optical coherence tomography (Casia SS-1000, Tomey, Nagoya, Japan). For axial
length measurement in the first group, we used an A-scan ultrasonic biometer
(Quantel Medical, Bozeman, MT USA) with an applanation technique under topical
anaesthesia (group A: UB).
Optical biometry (OB) is the most commonly used
method for IOL calculation; it uses keratometry measurements and thus obviates
the need for a second instrument. The advantages of OB over applanation are the
lack of risk of trauma or infection, increased patient comfort and improved
accuracy and repeatability of measurements[11].
In the second group, we used an AL-Scan Optical Biometer (Nidek Co., Ltd.,
Japan) for the IOL calculation (group B: OB). The goal for IOL power selection
was a postoperative refraction of ±1.00 D.
The present study’s aim was to evaluate refractive
outcomes in keratoconic patients who underwent cataract surgery after PK and to
analyse the impact of different devices (ultrasonography and interferometry)
for IOL calculation within this group.
Ethical Approval This
retrospective research study was carried out in the Ophthalmology Department of
Saint Barbara Hospital, Trauma Centre in Sosnowiec, Poland. It presents the
surgical treatment of 42 eyes in 34 patients with keratoconus who primarily
underwent PK with consecutive phacoemulsification. The data analysed from
medical records included demographics, medical history, corrected distance
visual acuity, technique of IOL calculation and calculation formula. All parts
of the data analysis were conducted under the tenets of the Declaration of
Helsinki, and all patients signed an individual informed consent form before
every surgical procedure. All surgeries, as routine treatments, did not require
bioethical committee approval.
All qualified patients underwent a complete
ophthalmic examination, including best corrected distance visual acuity test
(BCVA), an intraocular pressure (IOP) measurement by Goldmann applanation
tonometry, a slit-lamp biomicroscopy and a fundus examination (if possible).
Exclusion criteria were other corneal ectasias, other ocular surgery, previous
trauma and high astigmatism (>8.0 D) that could affect the final refractive
treatment. The keratoplasties were performed between 2009 and 2015, and the
phacoemulsifications were performed between 2011 and 2017. The mean interval
between the keratoplasty and the phacoemulsification was 32mo. All corneal
sutures were removed at least one year before cataract surgery. Eight patients
underwent PK and phacoemulsification in both eyes.
Keratoplasties were performed under general
anaesthesia. The donor corneas originated from domestic tissue banks. For
trephination, we used the Hanna vacuum trephine system (Moria Inc., Antony,
France) and a femtosecond laser (VisuMax, Carl Zeiss, USA) or Barron radial
vacuum trephines (Katena Products Inc. Denville, NJ, USA). The
phacoemulsification procedure with PC IOL implantation (Acrysof IQ, Alcon, USA)
was performed under topical anaesthesia (Alcaine, Alcon, Fort Worth, TX, USA)
with the Infiniti or Centurion Vision Systems (Alcon, Fort Worth, TX, USA). IOL
power was determined by using both standard and corneal topography-derived
keratometry using the SRK/T formula[12]. We used
two independent methods, UB and OB, for IOL calculation. In 16 eyes with dense
cataracts, IOL power was based on UB, while for 26 eyes OB was the basis for
choosing the IOL power. Target refraction, including refractive error of the
contralateral eye, was evaluated to reach refractive errors between 0 and -1.0
D.
Statistical Analysis Statistical
analysis was performed using Statistica v.3.1 (StatSoft, Tulsa, OK, USA). The
Wilcoxon rank-sum test was used to compare numerical variables between the two
groups. The results are presented as a mean±standard deviation (SD). In a
Bland-Altman plot, the difference between the measurements with different
methods is plotted against their mean. The 95% limits of agreement (mean
difference ±1.96SD) give the
distance between the measurements of the methods with 95% confidence. The
Bland-Altman plot also shows the proportional bias in the measurements, which
is the relationship of the difference between the measurements and the true
value. A P value of less than 0.05 was considered statistically
significant.
The BCVA before cataract surgery ranged from 0.01
to 0.4 (mean 0.09±0.19) on Snellen charts. The BCVA ranged from 0.2 to 0.7
(mean 0.38±0.14) at 1mo after surgery and from 0.2 to 1.0 (mean 0.56±0.16; P<0.05)
at 3mo postoperatively.
All 42 eyes underwent both methods of IOL
calculation. Table 1 presents the expected and achieved refractions 3mo after
phacoemulsification and PC IOL implantation (final refraction). Method 1
presents refraction dependent on UB, while Method 2 presents refraction
dependent on OB. IOL power was calculated to reach final refractive errors
between 0 and -1.0 D.
Table 1 Expected and achieved refractions
|
Refraction |
Method 1, UB |
Method 2, OB |
P value |
|
Expected refraction |
|
|
0.47 |
|
Mean±SD |
-0.69±0.40 D |
-0.72±0.40 D |
|
|
Range in diopters (spherical
equivalent) |
-1.55 to -0.1 |
-1.43 to -0.20 |
|
|
Achieved refraction |
|
|
0.16 |
|
Mean±SD |
-1.02±0.54 D |
-0.86±0.53 D |
|
|
Range in diopters
(spherical equivalent) |
-1.78 to -0.25 |
-1.54 to -0.3 |
|
|
P value |
0.016 |
0.045 |
- |
UB: Ultrasound biometry; OB: Optical biometry.
The expected and achieved refractions were not
statistically significantly different in either Methods 1 or 2 (P>0.05),
and there was no statistically significant difference when comparing the two
different methods of IOL measurement. The distribution of final refractive
errors in both groups is summarized in Table 2. A majority of patients in both
groups did not meet the target refraction (below ±1.0 D): 64% with UB and 55%
with OB.
Table 2 Distribution of final achieved spherical
equivalent 3mo after surgery n (%)
|
Final refraction (spherical equivalent) |
Method 1, UB |
Method 2, OB |
|
≤0.25 D |
5 (12) |
4 (10) |
|
0.25 to ≤0.50 D |
4 (10) |
9 (21) |
|
0.50 to ≤0.75 D |
6 (14) |
1 (2) |
|
0.75 to ≤1.00 D |
0 |
5 (12) |
|
>1.00 D |
27 (64) |
23 (55) |
UB: Ultrasound biometry; OB: Optical biometry.
The Bland-Altman test shows the difference between
expected and achieved refractions using IOL measurement methods. The dotted lines
represent the mean thickness differences between the two methods, and the
interline zones represent the area of 95% limits of agreement (Figure 1). There
were no statistically significant differences between expected and achieved
refractions using IOL measurement methods (P>0.05).
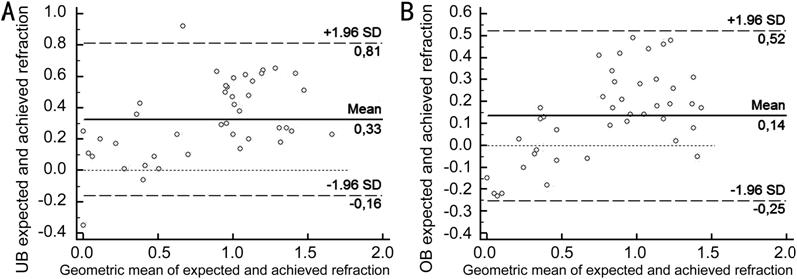
Figure 1 Results of the Bland-Altman test,
presenting the mean difference between achieved and expected refractions and
mean refractions in the UB measurement group (A) and the OB measurement group (B).
The differences between expected and achieved
refractions 3mo post-cataract surgery with myopic shift and the comparison of
expected and achieved refractions are presented in Figures 2 and 3,
respectively.
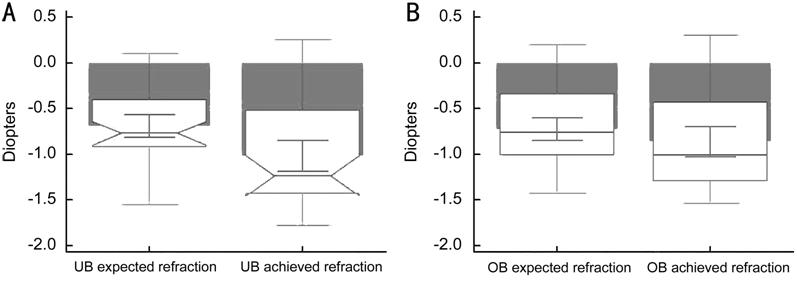
Figure 2 The difference between expected and
achieved refractions in both measurement methods with myopic shift in UB measurement
group (A) and OB measurement group (B); P<0.05.
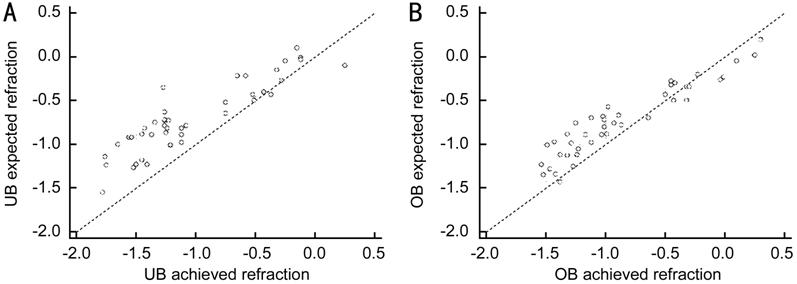
Figure 3 Comparison of expected and achieved
refractions in UB measurement group (A) and OB measurement group (B). The
significance level for both groups was P<0.0001.
To achieve expected refraction after cataract
surgery, precise IOL power calculation is crucial. Patients, especially younger
ones, have higher expectations of and demands regarding the final optical
result. As a group, our keratoconus patients are younger than typical cataract
patients. One of the most important sources of refractive surprise in UB is the
pressure on the cornea during measurement. Even when a single doctor takes all
the measurements, its result is myopic shift, which is compatible with the findings
of Karabela et al[13] and which contrasts
with the results reported by Fontes et al[14].
Despite the common usage of UB, OB is now
considered the gold standard in IOL calculation. Our results showed that it is
difficult to judge which method is preferable in keratoconus patients who have
undergone PK. Comparisons do not reveal significant differences between the
approaches. Keratoplasty in keratoconus patients interferes not only with
keratometry but also with axial length. In eyes that have undergone any kind of
refractive surgery, preoperative data could be included in the IOL calculation
formulae. In post-PK keratoconus patients, previous data are not applicable to
IOL power evaluation[15]. The changes in axial
length are high, as well as decrease of K-value. Additionally, after PK,
refractive error is not stable, and many patients require changes in spectacle
or contact lens correction[16]. Many patients
expect additional correction such as soft contact lenses or RGP lenses to
obtain better visual acuity and comfort.
In the present study, expected refraction did not
accord with the final result; several reasons may account for this discrepancy.
One is the presence of fluctuations of refractive error in a keratoconic cornea[17]. Such changes depend on the structure of a graft-host
interface and on changes in the remaining peripheral stroma. A second reason
may involve the clear corneal cut, and there is no data about the potential
influence of this cut on the final refraction in keratoconus. We know that
arcuate cuts in the peripheral cornea can be beneficial when correcting high
astigmatism, but in keratoconus we cannot precisely predict the final influence
of the cut. In such cases, we should consider a scleral tunnel for surgery or
microincision techniques to decrease the cut’s impact on postoperative
refraction[18]. In specific and demanding situations,
as in eyes with prior corneal surgery, especially post-PK, the standard
IOL calculation remains insufficient[19]. New
mathematical algorithms are necessary that take into account the specificity of
corneal shape, the anterior chamber depth and the clear corneal cut location in
keratoconus.
In the most challenging cases of high astigmatism,
in patients with ectatic corneal disorders like pellucid marginal degeneration
or keratoconus, cataract surgery with toric lens implantation is helpful in
reducing refractive error[20-22]. This method is also applied to eyes with prior
corneal refractive surgery with residual or induced astigmatism. In our group,
10 patients had astigmatism over 5 D; however, during the analysis of
topographic values and previous medical history, we decided to apply a
monofocal lens. These patients were offered, before cataract onset, spectacle
astigmatism correction lower then keratometric values, with satisfactory
results.
Cataract surgery after keratoplasty in keratoconus
presents a significant challenge. While the surgeon must include all available
data, including corneal shape and anterior chamber configuration, the surgeon’s
professional experience remains crucial to choosing the correct IOL power[23].
Conflicts of Interest: Krysik
K, None; Lyssek-Boron A, None; Janiszewska-Bil D, None; Wylegala
E, None; Dobrowolski D, None.