
Citation: Altinel MG, Kanra AY, Karadag R, Bayramlar H. Ex-PRESS implantation for different types of glaucoma. almol 2019;12(8):1290-1297. DOI:10.18240/ijo.2019.08.09
¡¤Clinical Research¡¤
Ex-PRESS implantation for different types of glaucoma
Meltem Guzin Altinel1, Ayse Yagmur Kanra2, Remzi Karadag3, Huseyin Bayramlar2
1Department of Ophthalmology, Fatih Sultan Mehmet Training and Research Hospital, Icerenkoy Atasehir 34752, Istanbul, Turkey
2Dunyagoz Hospitals Group, Sultanbeyli 34337, Istanbul, Turkey
3Department of Ophthalmology, Istanbul Medeniyet University School of Medicine, Goztepe Research and Training Hospital, Goztepe 34000, Istanbul, Turkey
Correspondence to: Meltem Guzin Altinel. Department of Ophthalmology, Fatih Sultan Mehmet Training and Research Hospital, Icerenkoy Atasehir 34752, Istanbul, Turkey. meltem.atik@gmail.com
Received:
Abstract
AIM: To evaluate the clinical results, efficacy and safety of Ex-PRESS P200 glaucoma shunt implant in different types of medically uncontrolled glaucoma.
METHODS: The study included 31 eyes of 31 patients that were unresponsive to medical antiglaucomatous therapy in whom Ex-PRESS P200 glaucoma shunt implantation was performed. The demographic characteristics of the patients, type of glaucoma, complete ocular examination results, number of antiglaucomatous drugs before and after surgery, early and late complications of surgery, additional surgical and nonsurgical medical interventions, and success rates were investigated from the patients¡¯ files, retrospectively.
RESULTS: The mean postoperative follow-up time was
16.4¡À7.5mo. The preoperative mean corrected intraocular pressure (IOP) was
28.7¡À
CONCLUSION: Ex-PRESS P200 glaucoma shunt implantation may be an effective procedure for medically uncontrolled glaucoma with significantly lower use of antiglaucomatous medications.
KEYWORDS: glaucoma; Ex-PRESS implant; glaucoma surgery
DOI:10.18240/ijo.2019.08.09
Citation: Altinel MG, Kanra AY, Karadag R, Bayramlar H. Ex-PRESS implantation for different types of glaucoma. almol 2019;12(8):1290-1297
INTRODUCTION
Glaucoma is a progressive optic neuropathy, and intraocular pressure (IOP) is the only known modifiable risk factor. According to the World Health Organization¡¯s (WHO) report, the number of persons estimated to be blind as a result of primary glaucoma is 4.5 million, accounting for slightly more than 12% of all global blindness, and as the majority of glaucoma cases can be effectively managed. When maximal tolerable IOP lowering medication and/or laser surgery fail to lower IOP or fail to prevent optic neuropathy progression, glaucoma filtration surgery comes into prominence[1]. The most widely used procedure for lowering IOP is trabeculectomy[2], which is effective in controlling IOP, but can lead to potential sight-threatening complications such as hyphema, hypotony, choroidal effusion, malignant glaucoma, infection, and cataract progression[3-4].
The Ex-PRESS glaucoma implant (Alcon Laboratories, Fort Worth, Texas, USA) is stainless steel device with an external diameter of 400 ¦̀m that was designed for more optimized filtration surgery. It is available with both 50-¦̀m and 200-¦̀m internal lumen size. The Ex-PRESS implant is a biocompatible, non-valved stainless steel device that was initially designed to be implanted under the conjunctiva to allow drainage of aqueous humor into the subconjunctival space. Implantion under a scleral flap is recommended to limit excessive aqueous flow and to decrease postoperative complications of subconjunctival implantation such as hypotonia and conjunctival erosion[5]. Compared with trabeculectomy, the Ex-PRESS device eliminates the need for peripheral iridectomy and removal of a corneoscleral tissue block. There are reports on Ex-PRESS implantation versus trabeculectomy in primary open angle glaucoma (POAG), but as yet there are no long-term follow up reports on the safety and efficacy of Ex-PRESS implantation in different types of glaucoma.
In this retrospective study, we wanted to report our experience with Ex-PRESS shunt implantation, the efficacy and safety of the device, the success and complication rates in patients with different types of glaucoma.
SUBJECTS AND METHODS
Ethical Approval This retrospective study was approved by the Scientific Research Commission of Istanbul Medeniyet University Goztepe Training and Research Hospital and conducted in accordance with the principles of the Declaration of Helsinki. Informed consent was obtained from the subjects.
Ex-PRESS
mini glaucoma shunt implantation results were evaluated retrospectively in 31
patients who were followed between December 2012 and December
Before the operation, identification of the exact cause of glaucoma, complete evaluation including Snellen best corrected visual acuity (BCVA; converted into logMAR), anterior segment examination, drainage angle evaluation, pachymetry, applanation tonometry, fundus examination, optical coherence tomography (3D-OCT 2000, Topcon Medical Systems) of the optic disc and RNFL, and standard automated perimetry using the Swedish interactive threshold algorithm 30-2 (Humphrey Field Analyzer II; Carl Zeiss Meditec, Dublin, CA, USA) were all performed.
Patients with POAG, pseudoexfoliative glaucoma (PXFG), secondary glaucoma due to congenital aniridia (CA), pigment dispersion syndrome (PDS), Axenfeld-Rieger syndrome (ARS), uveitic glaucoma (UG), primary angle closure glaucoma (PACG), neovascular glaucoma (NVG), post penetrating keratoplasty glaucoma (PKG), and aphakic glaucoma (AG) were included. All subjects had uncontrolled glaucoma despite maximally tolerated topical antiglaucomatous eye drops with progression in visual field defects and reduction in RNFL thickness. Patients with a follow-up less than 6mo were excluded.
All Ex-PRESS glaucoma shunt implantations were performed by two surgeons (Karadag R and Bayramlar H) under retrobulbar anesthesia. The surgical technique consisted of opening the conjunctiva and Tenon¡¯s capsule at the limbus, implanting the Ex-PRESS P200 shunt under a 4¡Á3-mm2 300-400-¦̀m partial thickness limbus-based scleral flap, similar to a standard limbus-based trabeculectomy. The superficial scleral flap was repositioned over the implant and closed using two 10-0 nylon sutures. The conjunctiva and Tenon¡¯s capsule were closed using 8-0 resorbable sutures. Antimetabolites were not used. Topical antibiotics and topical corticosteroids were used immediately after the operation 4 times per day and tapered off at the end of 1st month.
Postoperative examinations were done on the 1st day, 1st week, 1st, 3rd, and 6th month, and every 6mo thereafter. BCVA, anterior and posterior segment evaluations, and IOP were checked at every visit.
Complete surgical
success criteria were IOP¡Ư
Statistical Analysis Data were conducted using the IBM SPSS Statistics 22 software package (IBM SPSS, Turkey). Data were obtained in the forms of mean¡Àstandard deviation (SD) and frequency and percentage. The Wilcoxon signed-rank test and t-test were used to compare preoperative and postoperative data. The Mann-Whitney U test was used to compare two groups and Fisher-Freeman-Halton test was used for qualitative data. A P value <0.05 was considered to reflect significance.
RESULTS
The Ex-PRESS P200 shunt was implanted to 31 eyes of 31 patients (16 males, 15 females). The mean follow-up period was 16.4¡À7.5 (range 6-30) mo. The mean age of the patients at the time of surgery was 67.1¡À17.7 (range 12-88)y (Table 1).
Table 1 Demographic characteristics of all patients
Parameters |
Values |
Gender, n (%) |
|
M |
16 (51.6) |
F |
15 (48.4) |
Age, range (mean¡ÀSD, y) |
12-88 (67.1¡À17.7) |
Preop. medication, range (mean¡ÀSD) |
2-4 (3.90¡À0.39) |
Preop. IOP, range (mean¡ÀSD, mm Hg) |
15-57 (28.71¡À10.31) |
Preop. BCVA, range (mean¡ÀSD, logMAR) |
0-2 (0.73¡À0.66) |
IOP: Intraocular pressure; BCVA: Best corrected visual acuity.
Twenty-one of 31 eyes had previous surgical treatment and laser iridotomy procedures. Three eyes had Nd-YAG laser iridotomy (2 eyes had only Nd-YAG, 1 eye Nd-YAG plus clear lens extraction) before Ex-PRESS surgery. Nine previous Phaco-IOL surgeries, 1 pars plana vitrectomy (PPV), 1 penetrating keratoplasty (PK), 1 combined PK and Phaco-IOL surgery, and 1 corneal perforation surgery combined with PK and Phaco-IOL were performed before Ex-PRESS surgery. Five patients had previous trabeculectomy history; 1 had only trabeculectomy, 3 had combined trabeculectomy and Phaco-IOL, and 1 had Phaco-IOL and two trabeculectomy operations before the Ex-PRESS surgery. This study contains different etiologies of glaucoma. Fifteen of 31 eyes with POAG, 5 with PXFG, 2 with PACG, 1 with UG, 1 with NVG, 1 with CA, 1 with ARS, 1 with PDS, 2 with PKG, 1 with combined PKG and AG together, and 1 with AG were included (Figure 1).
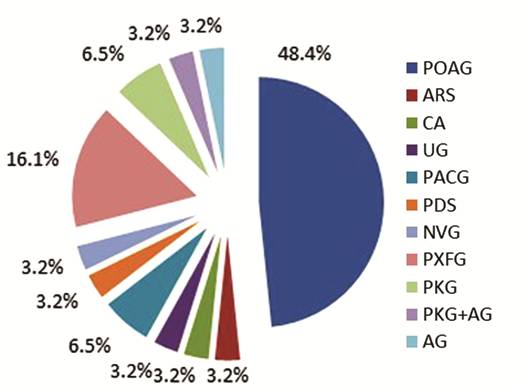
Figure 1 Types of glaucoma POAG: Primary open-angle glaucoma; PXFG: Pseudoexfoliative glaucoma; CA: Congenital aniridia; PDS: Pigment dispersion syndrome; ARS: Axenfeld-Rieger syndrome; UG: Uveitic glaucoma; PACG: Primary angle-closure glaucoma; NVG: Neovascular glaucoma; PKG: Post penetrating keratoplasty glaucoma; AG: Aphakic glaucoma.
The BCVA changed from a mean preoperative value of 0.73¡À0.66 to 1.03¡À0.68 logMAR on the 1st day, 0.68¡À0.52 logMAR at the 1st week, 0.66¡À0.55 logMAR at the 1st month, 0.57¡À0.50 logMAR at the 3rd month, 0.65¡À0.60 logMAR at the 6th month, 0.79¡À0.64 logMAR at the 12th month, 0.92¡À0.78 logMAR at the 18th month, 0.82¡À0.69 logMAR at the 24th month, and 0.34¡À0.10 logMAR at the 30th month (Figure 2). At 7 patients vision worsened but 6 patients gained a significant amount of vision: >2 Snellen lines when comparing pre-operative and last visit¡¯s BCVA values.
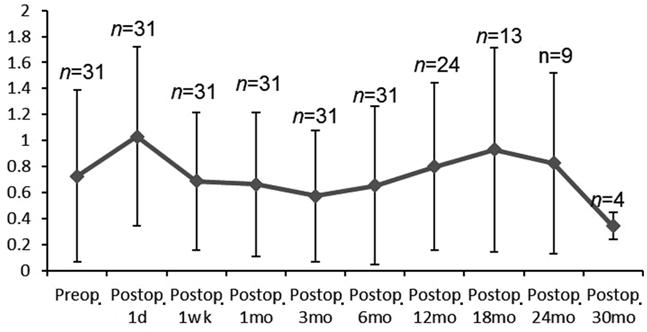
Figure 2 Preoperative and postoperative change of vision BCVA: Best corrected visual acuity; n: No. of patients for each time point.
The IOP
decreased from a mean preoperative pressure of 28.7¡À10.3 to 14.1¡À
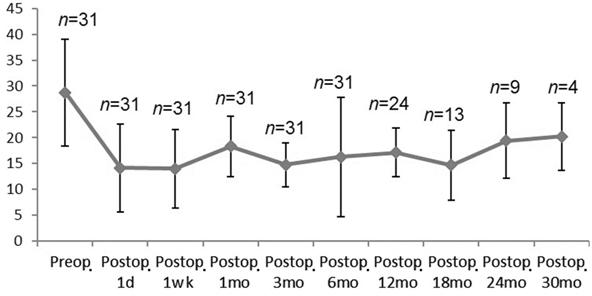
Figure 3 Mean IOP fluctuations at different times before and after surgery n: No. of patients for each time point.
The reduction in anti-glaucomatous medication after the surgery was significant. Twenty-nine (93.5%) of 31 patients were using 4, 1 patient (3.2%) was using 2, and 1 patient (3.2%) was using 3 anti-glaucomatous medications before surgery. The patients who were using 2 and 3 medications before surgery were allergic to other anti-glaucomatous drugs. At the last visit, 13 (41.9%) of 31 patients were not using any anti-glaucomatous medications. At the last visit, the mean number of medications per patient was reduced from a preoperative value of 3.9¡À0.4 to 1.7¡À1.7 (P<0.05; Figure 4).

Figure 4 The medication use rate before and after surgery.
At the last visit, the complete success rate was 32.3%, and the qualified success rate was 77.5% in all patients. Failure occurred in 7 (22.6%) patients (Table 2).
Table 2 Relationship between failure and types of glaucoma
Types of glaucoma |
Failure |
PKG |
1 |
ARS |
1 |
PKG+AG |
1 |
POAG |
3 |
PXFG |
1 |
Total No. of patients |
7 |
PKG: Post penetrating keratoplasty glaucoma; ARS: Axenfeld-Rieger syndrome; AG: Aphakic glaucoma, POAG: Primary open angle glaucoma; PXFG: Pseudoexfoliative glaucoma.
No significant intraoperative complications were noted. The most common post-operative early complication was conjunctival leakage, which was seen in 7 patients. The other early post-operative complications were device iris touch in 1 patient, intraocular hemorrhage, hyphema, device iris touch and choroidal effusion together in 1 patient, transient hypertony in 3 patients, transient hypotony in 2 patients, and corneal edema in 1 patient. Late complications were endophthalmitis at 6th month in 1 patient, and corneal opacification in 1 patient, in whom early corneal edema was seen (Table 3, Figure 5).
Table 3 Postoperative complications at different types of glaucoma n (%)
Postop. complications |
POAG |
PXFG |
UG |
AG |
PKG+AG |
PKG |
ARS |
PACG |
NVG |
PDS |
CA |
Conjunctival leakage |
2 (13.3) |
2 (40.0) |
1 (100) |
1 (100) |
1 (100) |
- |
- |
- |
- |
- |
- |
Iris touch |
- |
- |
- |
- |
1 (50.0) |
- |
- |
- |
- |
- |
|
Transient hypotony |
2 (13.3) |
- |
- |
- |
- |
- |
- |
- |
- |
- |
|
Transient hypertony |
- |
- |
- |
1 (100) |
- |
1 (100) |
1 (50.0) |
- |
- |
- |
|
Corneal edema |
1 (6.6)a |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
Intraocular hemorrhage, hyphema, iris touch, and choroidal effusion |
1 (6.6) |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
Endophthalmitis |
- |
1 (20.0) |
- |
- |
- |
- |
- |
- |
- |
- |
- |
Corneal opacification |
1 (6.6)a |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
PKG: Post-keratoplasty glaucoma; ARS: Axenfeld-Rieger syndrome; AG: Aphakic glaucoma; POAG: Primary open angle glaucoma; PXFG: Pseudoexfoliation glaucoma; UG: Uveitic glaucoma; PACG: Primary angle-closure glaucoma; NVG: Neovascular glaucoma; PDS: Pigment dispersion syndrome; CA: Congenital aniridia. aSame patient.
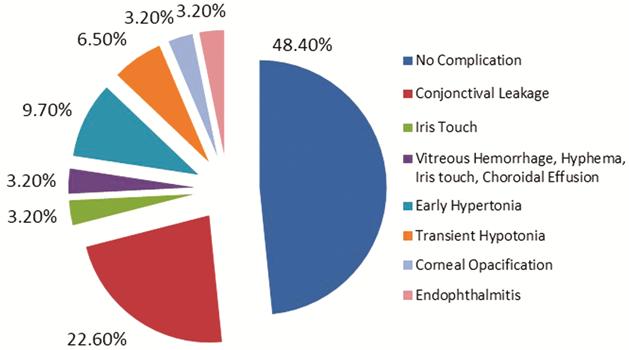
Figure 5 Early and late complications of surgery.
Twelve additional surgical treatments were needed and 4 of these were Phaco-IOL surgery alone. Additionally, to this surgical treatments 3 eyes had bleb needling to control high IOP. One of the patients who needed bleb needling had UG and required needling at the 19th month, the second patient had NVG and required needling at the 17th month, the third patient had POAG and required needling at the 2nd month. Fibrotic membranes over tubes were removed in 2 eyes with additional surgery. One of these patients had PKG and blockage occurred at the 6th month, the other patient had ARS and tube blockage occurred at the 6th day. Bleb revisions were performed in 3 eyes and 5-FU was used in 2 of these eyes. One of these patients had PXFG and required revision at the 1st month. The second patient had POAG and needling was performed, which required revision at the 12th month. The third patient, in whom fibrotic membranes had previously been removed, had PKG and required revision at the 24th month. Ex-Press implant was removed from 1 patient at the 6th month because of endophthalmitis and Phaco-IOL surgery was performed. Phaco-IOL surgery was performed in another 4 patients. PK was performed in 1 patient at the 18th month because of corneal opacification. Because of grefon failure in 1 patient in whom fibrotic membrane removal and revision had been performed previously, rekeratoplasty was performed at 8th month (Table 4).
Table 4 Additional surgical treatments at different types of glaucoma n=31
Additional Surgical Treatments |
POAG (n=15, phakic:9) |
PXFG (n=5, phakic:2) |
UG (n=1, phakic:1) |
AG (n=1, phakic:0) |
PKG (n=2, phakic:1) |
PKG+AG (n=1, phakic:0) |
ARS (n=1, phakic:0) |
PACG (n=2, phakic:1) |
NVG (n=1, phakic:0) |
PDS (n=1, phakic:0) |
CA (n=1, phakic:1) |
Phaco-IOL |
3 |
- |
1 |
- |
- |
- |
- |
- |
- |
- |
- |
Fibrotic membrane removal |
- |
- |
- |
- |
|
- |
1 |
- |
- |
- |
- |
Bleb revision |
1b |
1 |
- |
- |
|
- |
- |
- |
- |
- |
- |
Ex-PRESS removal+ Phaco-IOL |
- |
1 |
- |
- |
- |
- |
- |
- |
- |
- |
- |
PK |
1 |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
Re-keratoplasty |
- |
- |
- |
- |
|
- |
- |
- |
- |
- |
- |
Bleb needling |
1b |
- |
1 |
- |
- |
- |
- |
- |
1 |
- |
- |
POAG: Primary open angle glaucoma; PXFG: Pseudoexfoliation glaucoma; UG: Uveitic glaucoma; AG: Aphakic glaucoma; PKG: Post-keratoplasty glaucoma; ARS: Axenfeld-Rieger syndrome; PACG: Primary angle-closure glaucoma; NVG: Neovascular glaucoma; PDS: Pigment dispersion syndrome; CA: Congenital aniridia; Phaco-IOL: Phacoemulsification and intraocular lens implantation; PK: Penetrating keratoplasty. a,bSame patient.
When we
compared patients with POAG and others within the two groups, complete success
rates were 46.7% in POAG group and 18.8% in the others group. The qualified
success rates were 90% in the POAG group and 75% in other groups. The mean
preoperative IOP was 23.8¡À
Table 5 Comparison of BCVA (logMAR) and IOP values of the POAG and other groups mean¡ÀSD (median)
Prameters |
POAG group |
Other groups |
aP |
BCVA (logMAR) |
|||
Preop. |
0.4¡À0.21 (0.3) |
1.03¡À0.8 (0.9) |
0.075 |
At the last visit |
0.53¡À0.48 (0.4) |
1.05¡À0.73 (1) |
0.036 |
bP |
0.530 |
0.205 |
|
IOP |
|||
Preop. |
23.8¡À7.98 (21) |
33.31¡À10.31 (30) |
0.006 |
At the last visit |
15.67¡À5.64 (15) |
14.44¡À5.67 (14) |
0.311 |
bP |
0.004 |
0.001 |
BCVA: Best corrected visual acuity; POAG: Primary open angle glaucoma. aMann-Whitney U test; bWilcoxon signed-rank test.
DISCUSSION
Since 1968, the most widely used procedure for glaucoma surgery has been trabeculectomy[2]. Trabeculectomy is an effective surgery for controlling medically uncontrolled glaucoma but has severe complications. The potential complications of trabeculectomy include early hypotony, choroidal detachment, and bleb-related problems[3]. In recent years, there has been increasing interest in finding an alternative to trabeculectomy in glaucoma surgery, which explains the rising popularity of this field in ophthalmic surgery. The Ex-PRESS glaucoma shunt device was created to mimic the effect of trabeculectomy and improve its safety. Compared with trabeculectomy, the Ex-PRESS device eliminates the need for both peripheral iridectomy and removal of a deep corneoscleral tissue block. The initial tube was designed for subconjunctival implantation. Complications such as conjunctival erosion, tube obstruction, and device dislocation were seen with this technique[6]. In 2005, Dahan and Carmichael described an alternative surgical technique in which a device was implanted under a scleral flap[5]. Maris et al[7] reported satisfactory IOP control and reduced postoperative complication rates with this new technique.
The Ex-PRESS shunt device has been in widespread use but there is a paucity of data regarding its results. In this retrospective study, we investigated the efficacy and safety of the Ex-PRESS P200 device in a group of patients with different types of medically uncontrolled glaucoma.
In our
study, the mean IOP reduction was 39.9%¡À30.6% when comparing the last visit
with pre-operative values in all patients. Our results for all patients are
similar to other studies in the literature. Dahan and Carmichael reported
>35% mean IOP reduction at all time points in 24 eyes after implantation of
Ex-PRESS devices under scleral flaps[5]. De Jong et
al[8] reported 42%, and Good and Kahook[9] reported a 45% mean IOP reduction. In the others group,
the mean IOP reduction was -51.91¡À
In 7 patients vision worsened: >2 Snellen lines but 6 patients gained a significant amount of vision: >2 Snellen lines when comparing pre-operative BCVA values and those of the last visit. The reasons of vision reduction were failure of the operation, post-operative complications or having complicated or secondary glaucoma. One of the reasons for vision improvement was having cataract surgery after Ex-PRESS surgery, the other reason might be the reduction of anti-glaucomatous medication, which are mostly toxic to the corneal epithelium and cause of dry eye syndrome. In the POAG group, the preoperative and postoperative BCVA values were higher than in the others group because others¡¯ group contained complicated or secondary glaucoma cases.
In our study, the most common early complication was leakage, which was seen in 7 patients (22.6%). In Dahan et al¡¯s[10] study, the bleb leakage ratio was 7%, in de Jong¡¯s[11] study it was 2.5%, in Netland et al¡¯s[12] study, the early wound leak ratio was 3.3% and late bleb leakage ratio was 1.7%, and it was 6% in the device group in Maris et al¡¯s[7] study. Only in Marzette and Herndon¡¯s[13] study was the bleb leakage ratio higher than in our study (29%).
Choroidal hemorrhage, hyphema, and choroidal effusion were seen in 1 patient. Choroidal effusion was reported in 8% of cases in Maris et al¡¯s[7] study, 4% in Marzette and Herndons[13] study, and 6.8% in Netland et al¡¯s[12] study. Choroidal detachment was reported in 20% of cases in Sugiyama et al¡¯s[14] study, and 7.5% in de Jong¡¯s[11] study. In Wang et al¡¯s[15] study, choroidal effusion was reported in 4.2% of cases in the Ex-PRESS group and 29.2% in the trabeculectomy group.
Corneal edema was seen in 1 patient on the first day after surgery, and corneal opacification was seen in the same patient at 18mo. In this study endothelial cell counting was not a routine procedure so we cannot comment as to whether it was because of surgery or endothelial cell insufficiency. Implantation surgery is considered to be a safer procedure but it can cause more corneal complications than trabeculectomy[16-17]. Lee et al[18] reported statistically significant differences between trabeculectomy and Ex-PRESS shunt implantation, particularly in IOP fluctuation and the rate of endothelial cell loss. In their analysis, endothelial cell loss was significantly higher in the trabeculectomy group[18]. The mechanism underlying the damage to corneal endothelial cells following Ex-PRESS implantation remains unknown. There are as yet no long-term follow-up reports on the safety of ExPress implantation in the corneal endothelium.
Twelve additive surgical interventions and 3 bleb needling procedures were performed. The bleb needling ratio was similar to other studies in the literature. In the study of Maris et al[7], the bleb needling with 5-FU ratio was 20% in the device group, and in Sugiyama and Gallego-Pinazo¡¯s study the bleb needling ratios were also 20% in the device group[14,19]. The Ex-PRESS implant was removed at the 6th month in 1 patient because of endophthalmitis. In Maris et al¡¯s[7] study, endophthalmitis was reported at a rate of 2% in the device group. In our study, medical and surgical interventions were performed more than in other studies because our study included complicated glaucoma cases. In our study, number of phakic patients was 15, 4 of whom needed cataract extraction surgery after Ex-PRESS implantation. In a recent randomized clinical trial for postoperative complications after Ex-PRESS implantation versus trabeculectomy, Arimura et al[20] reported that although Ex-PRESS implantation might be associated with an increased rate of corneal endothelial cell loss compared with trabeculectomy, it is beneficial for preventing cataract progression. The reason for cataract progression after glaucoma surgery is not clearly understood.
Our study included 1 patient with NVG. We observed no early or late complications in this patient. This patient required bleb needling at the 17th month and qualified success was observed at the end of the follow-up even though this was a complicated glaucoma case. In a recent retrospective comparative study with 1-year follow-up, Ex-PRESS shunt surgery was found less effective but safer than trabeculectomy for the treatment of patients with NVG[21].
In the literature,
complete and qualified success criteria differ between studies. In our study,
complete success was defined as IOP¡Ư5 and ¡Ü
In our study, there was a significant reduction in the number of glaucoma medications after implantation of Ex-Press devices. The mean number of medications at the start was 3.9¡À0.4, which lowered to a mean of 1.7¡À1.7 (P<0.05) at the last visit. Coupin et al[26] indicated that the number of topical medications went from a mean of 1.9¡À1.0 down to 1¡À1.0 after surgery. Maris et al. reported an important reduction went from 3.7¡À0.9 to 0.7¡À1.2, similar to Couplin et al¡¯s[26] report. Lee et al[18] showed a significant reduction from 2.75¡À0.45 to 0.53¡À0.80 over a 12-month evaluation period in the Ex-PRESS group. We think that the final number of medications was higher in our study than in other studies because of group heterogeneity and the long duration of follow-up.
In conclusion, although this study included complicated glaucoma cases, IOP and anti-glaucoma medications were significantly reduced at the last visit. Overall, the complication rate was high. The major limitation of our study is its small sample size (31 eyes), the retrospective design of the study, and lack of anti-fibrotics. Studies including a larger number of complicated glaucoma cases with long-term follow-up will provide further information on the efficacy and safety of Ex-PRESS implantation.
ACKNOWLEDGEMENTS
Conflicts of Interest: Altinel MG, None; Kanra AY, None; Karadag R, None; Bayramlar H, None.
REFERENCES