
Citation: Ní Gabhann-Dromgoole J, De Chaumont C, Shahnazaryan D,
Smith S, Malone C, Hassan J, De Gascun CF, Jefferies CA, Murphy CC. Systemic
IL-1β production as a consequence of corneal HSV-1
infection-contribution to the development of herpes simplex keratitis. Int J
Ophthalmol 2019;12(9):1493-1497
DOI:10.18240/ijo.2019.09.19
・Brief Report・
Systemic
IL-1β
production as a consequence of corneal HSV-1 infection-contribution to the
development of herpes simplex keratitis
Joan Ní Gabhann-Dromgoole1,2, Ciaran de
Chaumont1,2, David Shahnazaryan2,3, Siobhán Smith1,
Conor Malone2,3, Jaythoon Hassan4, Cillian F. De Gascun4,
Caroline A. Jefferies1,5, Conor C. Murphy2,3
1Molecular
and Cellular Therapeutics and RSCI Research Institute, Royal College of
Surgeons in Ireland, Dublin 2, Ireland
2Department
of Ophthalmology, Royal College of Surgeons in Ireland, Dublin 2, Ireland
3Department
of Ophthalmology, Royal Victoria Eye and Ear Hospital, Dublin 2, Ireland
4National
Virus Reference Laboratory, University College Dublin, Belfield, Dublin 4,
Ireland
5Department
of Medicine, Division of Rheumatology and Department of Biomedical Sciences,
Cedars-Sinai Medical Centre, Los Angeles, CA 90048, USA
Co-first
authors: Joan Ní
Gabhann-Dromgoole and Ciaran de Chaumont
Correspondence to: Joan Ní Gabhann-Dromgoole. Royal College of Surgeons in
Ireland, Molecular & Cellular Therapeutics (MCT) and Dept of Ophthalmology,
Royal College of Surgeons in Ireland, 123 St. Stephen’s Green, Dublin 2,
Ireland. joannigabhann@rcsi.ie
Received:
Abstract
This study sought to identify
potential therapeutic targets in herpes simplex keratitis (HSK) patients with
active and inactive infection by investigating peripheral cytokine production.
Peripheral blood mononuclear cells (PBMCs) and serum were prepared from healthy
controls and HSK patients during active infection or following treatment
(inactive infection). Serum antibody titres were determined by ELISA. Protein
expression levels were analysed by Western blot. Cytokine levels were determined
by multiplex ELISA. Active corneal herpes simplex virus type 1 (HSV-1)
infection resulted in significantly elevated peripheral levels of IL-1β in HSK patients compared to
healthy controls, and remained significantly increased following treatment.
Elevated production of IL-1β in inactive patients was associated with significantly increased levels
of IRF3 and STAT1, key proteins involved in promoting anti-viral immune
responses. Our data suggest that inflammation persists beyond the period that
it is clinically evident and that enhanced peripheral production of IL-1β may have implications for
HSV-1 viral clearance in active and inactive HSK patients.
KEYWORDS: herpes simplex virus type 1; herpes
simplex keratitis; inflammation; peripheral immune response; pathogenesis
DOI:10.18240/ijo.2019.09.19
Citation:
Ní Gabhann-Dromgoole J, De Chaumont C, Shahnazaryan D, Smith S, Malone C,
Hassan J, De Gascun CF, Jefferies CA, Murphy CC. Systemic IL-1β production as a consequence of corneal HSV-1
infection-contribution to the development of herpes simplex keratitis. Int J
Ophthalmol 2019;12(9):1493-1497
INTRODUCTION
Herpes
simplex keratitis (HSK), caused by herpes simplex virus type 1 (HSV-1), is a
sight-threatening infection and is the commonest cause of infectious blindness
in the developed world, affecting up to 90% of adult populations in certain
countries[1]. A hallmark of HSV-1 infection―as
with all members of the Herpesvirus family―is that following primary infection,
the virus remains latent for the life of the host. In the case of HSV-1, the
site of latency is the trigeminal ganglion. The virus may then undergo cycles
of reactivation from latency causing inflammation and scarring that can
permanently damage the cornea. Recurrent episodes of HSK, can lead to corneal
damage, visual morbidity and even corneal melting and perforation in
necrotising stromal keratitis, one of the most severe manifestations of the
disease. Corneal transplantation for visual or tectonic indications in HSK is
associated with a high risk of HSK recurrence in the transplant as well as
graft rejection and failure. Therefore, there is a need to develop better
treatments that can both control the infection quickly to limit the damage caused
by replicating virus and prevent reactivation of the disease by keeping it in
its latent state. HSV-1 is a ubiquitous human pathogen, with remarkably high
prevalence of HSV-1 infection that increases with age: autopsy studies have
revealed HSV-1 DNA in the trigeminal ganglia in 93% of adults, and 92% of
individuals with no reported history of herpes infection have been shown to
periodically shed HSV-1 DNA in their tears[2].
Despite being highly prevalent among the general population less than 1% of
people who are infected with HSV-1 develop ocular infection. Murine studies
suggest that the elevated levels of cytokines including IL-6, TGF-β, IL-1β,
TNF-α and more recently IL-17, detected within corneas following HSV-1
infection are important contributors to the development of HSK pathogenesis[3]. While current evidence supports a role for cytokines
acting locally in the cornea during HSK pathogenesis, peripheral cytokines have
not yet been characterised. To investigate the effect of HSV-1 infection
peripherally, serum cytokines and expression of key signalling molecules were
evaluated in healthy controls and patients with active and inactive HSV-1
infection.
SUBJECTS AND METHODS
Ethical
Approval This study was conducted in
accordance with the Helsinki Declaration. The study was approved by the
Research and Ethics Committee of the Royal Victoria Eye and Ear Hospital and
written informed consent was obtained from all participants.
Patient
Recruitment Six consecutive patients attending
the emergency department who met the inclusion criteria for this study and were
willing to participate were recruited. Of 1 female and 5 female patients with
an average age of 36-66y (mean age 48.3±10.7y) were recruited. Inclusion
criteria were recurrent acute epithelial or stromal HSK, age over 18y, and
ability to provide informed consent. Exclusion criteria were ocular or systemic
infection or inflammation other than HSK and a history of autoimmune disease.
All patients recruited to this study had presented with a sore, red, watery eye
and blurring of vision. Active HSK was defined by the presence of a distinctive
(usually dendritic) ulcer or inflammation in the cornea which was clinically
consistent with HSK. Upon diagnosis, suitable patients provided a blood sample
on the day of the diagnosis. Patients commenced standard treatment with topical
acyclovir +/- topical corticosteroids and oral acyclovir where clinically
indicated and were followed up at regular intervals, i.e. every 2-6wk
until the inflammation had resolved. Resolution of inflammation was determined
by an ophthalmologist in the course of follow up appointments, following a
clinical exam of the eye, skin, conjunctiva, anterior chamber, iris, retina, etc.
Specifically for keratitis, resolution of inflammation and healing of the
corneal wound was determined by clinical examination and negative fluorescein
staining. A second blood sample was obtained when the disease was determined to
be inactive or resolved based on clinical examination and for the purpose of
the study these patients were designated inactive HSK patients. None of the
active or inactive HSK patients recruited for the study suffered from recurrent
orolabial herpes. Five healthy controls who did not have a history of, and were
not suffering from, either HSK or recurrent orolabial herpes were recruited for
comparison.
Sample
Preparation Peripheral blood mononuclear cells
(PBMCs) were isolated from whole blood using a Ficoll density gradient
centrifugation and cultured in phenol red-free RPMI-1640 medium supplemented
with 10% fetal calf serum and 100
μg/mL penicillin-streptomycin. Antibody titres were determined in serum samples
from HSK patients and healthy controls using HerpeSelect® 1 ELISA
IgG and HerpeSelect® 2 ELISA IgG ELISA kits (Focus Diagnostics).
Index values were then calculated according to kit instructions. Samples with
index values >1.1 were recorded as positive, index values <0.9 were
recorded as negative. Values between 0.9 and 1.1 were recorded as equivocal.
Serum samples of HSK patients and healthy controls were further analysed by
multiplex ELISA (Meso Scale Dicovery) for the following cytokines: IL-1β
(0.57-10 000 pg/mL), IL-12/p70 (0.23-10 000 pg/mL), IL-10 (0.32-10 000 pg/mL),
TNF-α (0.69-10 000 pg/mL), IL-6 (0.13-10 000 pg/mL), IL-8 (0.15-10 000 pg/mL),
and IFN-γ (3.2-10 000 pg/mL).
Lysates were prepared and changes in the expression of STAT1 (Santa Cruz
Biotechnology #sc-592), and IRF3 (Santa Cruz Biotechnology #sc-15991), were
determined by Western blot followed by optical densitometry.
Statistics Student’s
paired t-tests were performed to examine differences in antibody titres,
cytokine levels and protein expression between active and inactive HSK
patients. Differences in antibody titres, cytokine levels and protein
expression between HSK patients and controls were examined using the
non-parametric Mann-Whitney test.
RESULTS
Levels of anti-HSV-1 and 2 antibodies were determined
in serum samples for all study participants (Figure 1). All study participants
were negative for HSV-2 antibodies. All HSK patients were positive for
anti-HSV-1 IgG antibodies during the active and inactive stages of the disease
and all healthy controls were negative for anti-HSV-1 antibodies. As expected,
we observed no significant difference in antibody titres between patients with
active or inactive infection (Figure 1).
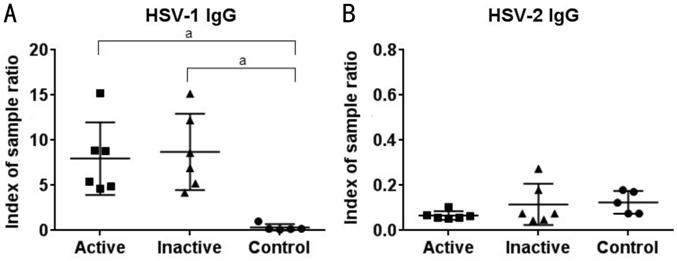
Figure 1 Detection of anti-HSV-1 and 2 antibody titers in
patients with active or inactive HSK and healthy controls Levels of
anti-HSV-1 and 2 antibodies in serum samples for patients with active or
inactive HSK and healthy controls were determined by ELISA as indicated. Each
symbol represents individual samples where numerical values denote index of
sample ratio. All analyses were performed using GraphPad Prism 6.0 for Windows
(GraphPad Software, La Jolla, CA, USA). aP<0.005 versus
healthy controls.
Serum samples of HSK patients and healthy controls were
analysed by multiplex ELISA in order to determine whether peripheral cytokines
are differentially expressed in HSK patients compared with healthy controls
(Figure 2). We observed a significant increase in IL-1β levels between patients
with active HSV-1 infection and healthy controls (P=0.004).
Additionally, IL-1β levels remained significantly elevated (P=0.01) in
these patients following treatment compared to healthy controls (Figure
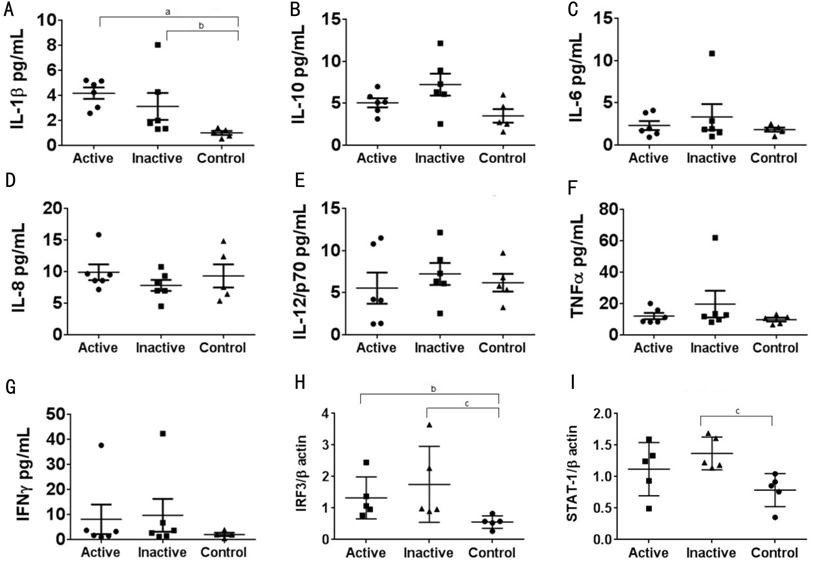
Figure 2 Comparison of peripheral cytokine levels and
transcription factor expression between patients with active or inactive HSK
and healthy controls A-G: Levels
of cytokines in serum samples were simultaneously measured using a multiplex
electrochemiluminescence assay (Meso Scale Discovery, Gaithersburg, MD, USA)
and read by an Imager2400 plate reader (Meso Scale Discovery, Gaithersburg, MD,
USA; n=5-6). H-I: PBMCs were isolated from HSK patients with active and
inactive disease and healthy controls. Endogenous expression levels of
indicated proteins were determined by Western blot (n=5). Results are
presented as mean±SD. Data were deemed to be significantly different at P values
less than 0.05. All analyses were performed using GraphPad Prism 6.0 for
Windows (GraphPad Software, La Jolla, CA, USA). aP<0.00, bP<0.05
and cP<0.008 versus healthy controls.
DISCUSSION
Overall our data suggests that localised HSV-1 infection
in the cornea results in potent IL-1β production and increased STAT-1 and IRF-3
expression in peripheral cells. These increases were shown to persist beyond
the period that was clinically evident, suggesting that enhanced peripheral
production of the pro-inflammatory cytokine IL-1β may have implications for
HSV-1 viral clearance. Our findings are consistent with studies showing that
prolonged use of topical steroids is required in the treatment of HSK to
promote resolution and reduce recurrence[7]. Murine studies suggest that
cytokines may be important contributors to the development of HSK
pathogenesis. These studies have shown
that following HSV-1 infection of the cornea the most prominent cytokines
produced are IL-1β, IL-6, IL-10, IL-12, and IFN-γ[3], typically several days after the
development of stromal keratitis. Elevated levels of IL-1β and TNF-α are
associated with corneal inflammation, while IL-6 and TGF-β are thought to exert
antiviral and inflammation regulatory activities in HSV-1 corneal infection[8]. Recent studies have confirmed that elevated levels of
IL-1β and TNF-α are not important for inhibiting viral replication, but instead
play a role in pathogenesis of HSV-1 infection[3].
IL-1β promotes the production of IL-17 and recent studies found that
following HSV-1 infection IL-17 was detected in infected corneas and its
suppression reduced the severity of the HSK[9].
Our study suggests that in addition to acting locally in the cornea during HSV
infection, peripheral cytokines may also contribute to disease pathogenesis. In
support of corneal HSV infection effecting peripheral cytokine production
previous studies have demonstrated a role for the innate system in the
pathogenesis of HSK, finding that prior to T cell mediated responses viral
infection leads to the production of pro-inflammatory cytokines and chemokines
and invasion of the cornea by polymorphonuclear leukocyte (PMN) initially
thought to promote viral clearance[10]. However, subsequent studies have demonstrated that
PMN invasion contributes to the pathology of HSK as these cells are a major
source of angiogenesis and tissue damaging factors, including nitric oxide[11]. Thus, events occurring at the ocular surface during
HSV infection can potentially contribute to the altered peripheral levels of
IL-1β observed in this study, potentially resulting in increased numbers of
lymphocytes being recruited to the cornea, while reduced induction of
pro-inflammatory cytokines and anti-viral factors might fail to limit viral
replication. Investigations into autoimmune conditions like Systemic Lupus
Erythematosus have shown the utility of measuring cytokines as indicators of
potential flares in these patients[12]. Thus,
monitoring serum samples of patients with a history of HSV infection might
prove to be a useful diagnostic tool as increased levels of IL-1β (and
consequently IL-17) in the periphery may predict relapses of HSK. Current
treatments for HSK include topical and systemic antiviral drugs such as
acyclovir and trifluorothymidine[7]. Thus, in
addition to current treatment options for HSK targeting IL-1β in the periphery
may have a beneficial outcome for localised keratitis induced by HSV-1, as a
means to reduce neutrophil and Th17 cell infiltration. Of note mice
transgenic for the IL-1 receptor antagonist protein are resistant to HSK[13]. Further studies are required to determine if
therapies targeting overproduction of IL-1β, such as anakinra, a recombinant
IL1-Ra antibody, and canakinumab, an anti-IL-1β monoclonal antibody hold potential
for the treatment of HSK[14].
Given the central role of IL-1β to the pathology of HSV-1 infection recent studies have
investigated the role of inflammasome activation following HSV-1 infection. In
human fibroblasts HSV-1 was shown to induce the activation of the IFI16 and
NLRP3 inflammasomes and promote the maturation of IL-1β during the early phase of infection. Furthermore ICP0
was shown to target IFI16 for rapid proteasomal degradation at later times
postinfection[5]. It has been suggested that IL-1β is secreted in a continuum which is dependent upon the
extracellular requirement for IL-1β[15]. Given that IL-1β has a very short half-live in
plasma[15],our data supports the view that
peripheral IL-1β levels observed in our study are as a result of
increased production in response to ocular HSK infection. However further
studies in PBMCs are required to determine if HSV-1 proteins (including ICP0)
play a role in modulating the expression of IRF3 and STAT
ACKNOWLEDGEMENTS
Foundation: Supported by the Health Research
Board and the Royal Victoria Eye and Ear Hospital Research Foundation through
the Medical Research Charities Group (No.1409).
Conflicts of
Interest: Ní
Gabhann-Dromgoole J, None; De Chaumont C, None; Shahnazaryan D,
None; Smith S, None; Malone C, None; Hassan J, None; De
Gascun CF, None; Jefferies CA, None; Murphy CC, None.
References