IF
in JCR CiteScore Rank About IJO Current Issue Featured
Articles Articles
In Press Recent
Accepted
 International Journal
of Ophthalmology
International Journal
of Ophthalmology
2017; 10(9): 1349-1353
��Basic Research��
MMP-2 Rs24386 (C-->T) gene polymorphism and the phenotype of age-related
macular degeneration
Rasa Liutkeviciene1,2, Vaiva
Lesauskaite3, Giedre Sinkunaite-Marsalkiene3, Sandrita
Simonyte3, Reda Zemaitiene1, Loresa Kriauciuniene1,2,
Dalia Zaliuniene1
¹Department
of Ophthalmology, Lithuanian University of Health Sciences, Medical Academy,
Kaunas LT-50161, Lithuania
²Neuroscience
Institute, Lithuanian University of Health Sciences, Medical Academy, Kaunas
LT-50161, Lithuania
3Intitute of
Cardiology, Lithuanian University of Health Sciences, Medical Academy, Kaunas
LT-50161, Lithuania
Correspondence
to: Sandrita Simonyte. Intitute of Cardiology, Medical Academy,
Lithuanian University of Health Sciences, Sukileliu 17, Kaunas LT-50161,
Lithuania. sandrita.simonyte@lsmuni.lt
Received: 2016-03-24
Accepted: 2017-06-23
Abstract
AIM:
To examine the MMP-2 (-1306 C/T) gene polymorphism and the phenotype characterized
by soft and hard drusen of early age-related macular degeneration (AMD) and
geographic atrophy of late AMD form.
METHODS: The
study enrolled 850 investigations (290 AMD patients with soft and hard drusen, 34
with geographic atrophy and a random sample of the population n=526).
Early AMD was classified according to the International Classification and
Grading System. For geographic atrophy diagnosis the Age-Related Eye Disease
Study classification was used. The potential association with single nucleotide
polymorphisms on MMP-2 Rs243865 was evaluated for all patients, adjusted
for age and sex. The genotyping test of MMP-2 Rs243865 (C-->T) was
conducted using the real-time polymerase chain reaction method.
RESULTS:
MMP-2 (-1306 C/T) C/C genotype was more frequently detected in AMD
patients with hard drusen than the soft drusen or control group (66.43% vs 53.74%,
vs 54.94%, P=0.047). Logistic regression analysis showed that the
MMP-2 (-1306) C/C genotype increased the likelihood to develop hard
drusen in AMD patients (OR=1.7, 95% CI: 1.06-2.74; P=0.028). No
association between MMP-2 (-1306 C/T) gene polymorphism in patients with
atrophic AMD and control group was found (54.94%, 37.64%, 7.41% vs 50%,
38.24%, 11.76%; P=0.6).
CONCLUSION:
The MMP-2 Rs24386 (C-->T) polymorphism is found to be associated with
the development of hard drusen in patients with AMD.
KEYWORDS: age-related macular degeneration; phenotype; matrix metalloproteinases;
gene polymorphism
Citation: Liutkeviciene
R, Lesauskaite V, Sinkunaite-Marsalkiene G, Simonyte S, Zemaitiene R,
Kriauciuniene L, Zaliuniene D. MMP-2 Rs24386 (C-->T) gene
polymorphism and the phenotype of age-related macular degeneration. Int J Ophthalmol 2017;10(9): 1349-1353
INTRODUCTION
Age-related
macular degeneration (AMD) causes significant and irreversible loss of central
vision. In developed countries, AMD is the most common cause of visual loss in
persons aged 60y and older[1].
Macular
degenerative lesions in case of AMD include drusen formation, changes in the
retinal pigment epithelium, atrophy of the retinal pigment epithelium and the
choroidal choriocapillary layer, lesion of Bruch��s membrane, geographic atrophy
of the central fovea, exudative AMD with choroidal neovascularization,
detachment of the retinal pigment epithelium, or submacular disciform scarring.
Drusen are
defined as outgrowth of colloidal material, similar to hyaline, that
accumulates in the retina, in Bruch��s membrane underlying the retinal pigment
epithelium. Drusen formation causes progressive degeneration of the retinal
pigment epithelium and photoreceptors[2] by disturbing oxygen metabolism and leading to the
degeneration of photoreceptors, while visual function impairment is associated
with the quantity of damaged photoreceptors. The fovea, with the largest
quantity of photoreceptors, is dominated by cones, whereas in the parafoveal
region, surrounding the fovea, rods dominate. In the early stages, mostly
photoreceptors in the parafovea are damaged.
Drusen are
divided into hard and soft. Hard drusen can induce atrophy of the retinal
pigment epithelium and choriocapillary layer. Soft drusen may confluent and
cause exudative AMD and can later induce the detachment of the neuroepithelium[3].
Recent
studies have demonstrate that angiogenesis is the most important mechanism of
AMD development and is associated with important extracellular remodeling
involving different proteolytic systems, among which matrix metalloproteinases
(MMPs) play an essential role[4].
It has been suggested that the decrease in MMP-2 activity correlates to drusen
formation[5].
In the MMP-2 (�C1306) gene promoter transcription region, a mutation
(rs243865), which causes an increase in promoter activity, has been discovered.
The coding gene for MMP-2 is located in 16q13-q21 locus. The C-to-T allelic
variation located at nucleotide -1306 disrupts the Sp1-binding site in the
promoter region and leads to low transcriptional activity; and when the T
allele has a markedly lower promoter activity than the C allele where promoter
loses 50% of activity[6].
Assuming the
fact that MMP-2 could have an influence on subretinal deposit formation[4-5], we hypothesized that the MMP-2
gene might be associated with soft/hard or atrophic AMD.
SUBJECTS AND
METHODS
Permission
to undertake the study was obtained from Kaunas Regional Ethics Committee for
Biomedical Research (No BE-2-14). All the subjects provided written, informed consent in accordance with the
Declaration of Helsinki. The study was conducted in the Department of
Ophthalmology, Hospital of the Lithuanian Health Sciences University and
Institute of Cardiology, Medical Academy, Lithuanian University of Health
Sciences.
The control
group comprised 526 persons, selected from persons participating in the
International Health, Alcohol and Psychosocial Factors in Eastern Europe
Project[7],
the International Countrywide Integrated Non-communicable Disease Intervention
Project[8],
and the Kaunas Healthy Ageing Study[9].
Of the 400
patients with early AMD, 290 (580 eyes) were recruited into the study according
to the inclusion criteria. The early AMD patients and the control persons were matched
by age and gender (Table 1). The atrophic AMD group matched the control group
only by gender.
Table 1
Demographic characteristics of patients with AMD and reference group
subjects
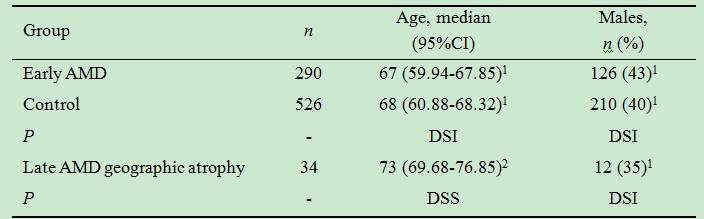
1DSI: Difference statistically insignificant; 2DSS:
Difference statistically significant.
Ophthalmological
Evaluation Ophthalmological evaluation for all subjects in our study
was carried out as described previously[10]. Morphologic
fundus changes were classified in our study as follows. Hard drusen were
defined as discrete whitish-yellow spots with a diameter of less than 63 ��m.
Soft distinct drusen were pale-yellow spots with uniform density and discrete
edges with a diameter larger than 125 ��m. Soft indistinct drusen were
pale-yellow spots with decreasing density from the center outwards and fuzzy
edges with a diameter greater than 125 ��m. The latter two groups were
integrated into one group. For the diagnosis of geographic atrophy, the
classification system of AMD formulated by the Age Related Eye Disease Study
(AREDS) was used[10].
Geographic atrophy was characterized by well seen separate areas of retinal
pigment epithelium atrophy with visible choroidal blood vessels, involving the
fovea and any of the features of neovascular AMD[11].
The fundus
images were graded according to the digital analysis method ��A Drusen Volume
Quantification Method based on a Segmentation algorithm in VIP Image��[12]. Where
disagreement occurred between the two graders, the results were adjudicated by
a senior retinal specialist (when there was a disagreement rate of over 4.5%, patients
were excluded from further research).
Optical
coherence tomography (OCT) was performed on all AMD patients, and fluorescence
angiography was performed on the patients with suspected exudative stage AMD
after the OCT examination.
The
exclusion criteria for subjects were as described previously[10]. Exudative
AMD was diagnosed in two patients excluding them from further analysis. Five
patients were excluded from the study because of suspected inherited macular
dystrophies.
The
inclusion criteria for patients with AMD were as follows: 1) patients of both
genders, with diagnosis of early mild or early intermediate AMD and without
other eye disorders, found on detailed ophthalmologic examination; 2) patients
with AMD but only with soft and hard drusen classified according to the
International Classification and Grading System; 3) patients with the diagnosis
of geographic atrophy according to the AREDS classification; 4) participation
consent.
DNA
Extraction and Genotyping DNA extraction and analysis of MMP-2 Rs243865 (C-->T) gene polymorphism were carried out in the Laboratory of Ophthalmology,
Institute of Cardiology, Lithuanian University of Health Sciences, as described
previously[10].
Statistical
Analysis Statistical
analysis was performed using the computer program SPSS/W 13.0 (Statistical
Package for Social Science for Windows, Inc., Chicago, Illinois, USA). To
compare the observed and expected MMP��s genotype frequencies Hardy-Weinberg
analysis was performed using the ��2 test in all groups. The
distribution of MMP-2 Rs243865 single-nucleotide polymorphism in the
early and atrophic AMD and reference group was compared using the ��2
test or the Fisher exact test. A difference was considered statistically
significant at P<0.05.
RESULTS
A total of
190 patients with early AMD and 34 patients with atrophic AMD were enrolled in
this study following to the inclusion and exclusion criteria. The control
comprised 526 persons. The age of the patients with early AMD ranged from 50 to
93y (median 67); the age of atrophic AMD group varied from 50 to 93y (median
73). The age of the control patients ranged from 50 to 93y (median 68) (Table
1).
The genotyping
of MMP-2 (�C1306) C/T was performed on patients with soft and hard drusen
in early AMD and control group (Table 2). The distribution of the analyzed MMP
genotypes and allele frequencies in patients with AMD with soft and hard
drusen and in the control group matched the Hardy-Weinberg equilibrium.
Table 2 MMP-2 (�C1306 C/T) genotype
frequency in patients with AMD with soft and hard drusen, in geographic atrophy
and in the control group
n
(%)
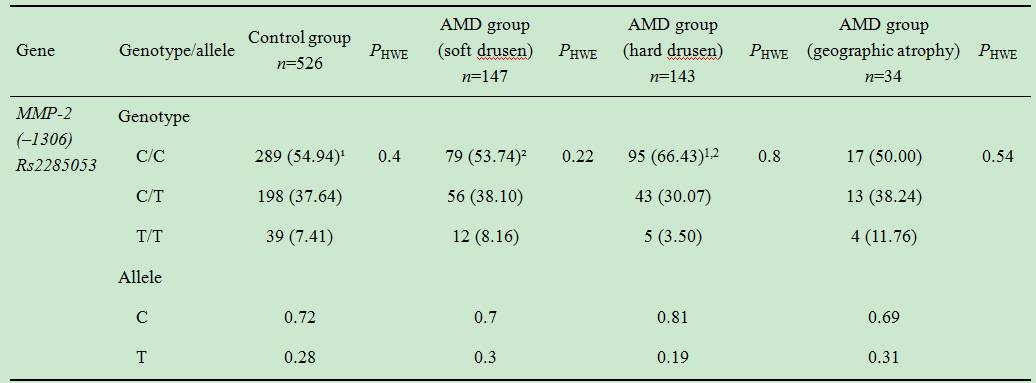
PHWE: Significance level by Hardy-Weinberg
equilibrium. ¹P=0.017; ²P=0.03.
AMD patients
with hard drusen had MMP-2 (�C1306) C/C genotype detected more frequently
than the control group and those with soft drusen (66.43% vs 54.94% and
53.74%, P=0.017 and P=0.03, respectively) (Table 2). Logistic
regression analysis revealed increased likelihood of developing hard drusen in
AMD patients with MMP-2 (�C1306) C/C genotype (OR=1.7, 95%CI:
1.06-2.74; P=0.028).
Analysis of MMP-2
Rs24386 (C-->T) gene polymorphism did not reveal any differences in the
distribution of genotype (T/T, C/T, and C/C)
between men with soft and hard drusen (14.06%, 35.94%, and 50% vs 4.84%,
29.03%, and 66.13%, P=0.95, respectively) and between women with soft
and hard drusen (3.61%, 39.76%, and 56.63% vs 2.47%, 30.86%, and 66.67%,
P=0.41).
To reduce the possibility of type I error due to multiple testing, we
employed the Bonferroni correction, and a P value >0.05 (since we analyzed 4
different groups) was considered to be statistically insignificant.
There were
no statistical significant differences in distribution of MMP-2 (�C1306 C/T) genotypes
comparing atrophic AMD with soft and hard drusen, and control group as well
(Table 2).
DISCUSSION
Studies analyzing the influence of MMP-2 on AMD
development are scarce. Some studies analyzed the MMP-2 concentration in the
blood[13], MMP-2
expression[14]
and MMP-2 polymorphism in different promoter regions[15-16] and results of these studies are inconsistent.
However, to our knowledge, there are no studies analyzing associations between
MMP-2 gene polymorphism and AMD phenotype. Price et al[6] determined a
mutation which causes an increase in promoter activity. The C-to-T allelic
variation located at nucleotide -1306 disrupts the Sp1-binding site in
the promoter region leading to significantly lower transcriptional activity;
therefore, T allele has a markedly lower promoter activity than the C
allele, where promoter loses 50% of activity of the gene. So it was
hypothesized that C/C genotype, which causes higher gene expression,
might participate in exudative AMD development, and T/T genotype, which
causes lower expression, might participate in geographic atrophy AMD
development. Mostly, exudative AMD develops from soft drusen, and geographic
atrophy from hard drusen. These results demonstrate that MMP-2 (�C1306) C/C genotype
was more frequent in AMD patients with hard drusen than the control group
(66.43% vs 54.94%, P=0.017), and in AMD patients with hard drusen
than soft drusen (66.43% vs 53.74%, P=0.03). Logistic regression
analysis revealed that the MMP-2 (�C1306) C/C genotype was associated
with 1.7-fold greater possibility to develop hard drusen in AMD patients. These
results proved that MMP-2 (-1306 C/T) gene polymorphism C/C genotype
was associated with AMD phenotype with hard drusen. Our study supports
the role of matrix MMPs and their polymorphisms in hard drusen
formation, and we are in agreement with Hyman et al[5] which suggested that MMP-2
activity correlates to the increase in collagen deposition and, potentially,
subretinal deposit formation.
We
hypothesized that T/T genotype, which causes lower expression, might be
associated with hard drusen development; however, the results were opposite.
Maybe it can be explained by drusen phenotype changing over time as the disease
progresses, thereby confounding the phenotype and stage of the disease.
To detect
the phenotypical influence of genetic polymorphisms a greater number of patients
may be needed.
To our
knowledge, currently there are only three studies analyzing MMP-2 (�C1306)
C/T gene influence on AMD development[10,15-16]
but not to AMD phenotype. Two studies found no association between AMD[15-16]. Seitzman et al[15] analyzed MMP-2
(-1306) C/T gene polymorphism in females with AMD and did not find any association
between MMP-2 and early or late stage AMD in older women. Ortak et al[16] also analyzed
genotype distributions and allelic frequencies of MMP2 (-1306 C>T).
They did not find significant differences in either genotype distribution or
allelic frequencies of MMP2 (-1306 C>T) among the patients with dry AMD, wet AMD
and the control group[13].
However, the latter study did not analyze MMP2 (-1306 C>T) association
with AMD phenotype, but analyzed the association between dry AMD, wet AMD and
the control group[16],
and, is in disagreement with our study, we found association with MMP-2
(-1306) C/C genotype and hard drusen. We hypothesized that this genotype
might be associated with only hard drusen development but not with exudative or
atrophic AMD, because others studies did not find any association with this
genotype and latest forms of AMD[15-16].
The third study found a significantly more frequent distribution of the C/C and
C/T genotypes in the patients with AMD younger than 65y and those aged ��65y,
respectively. Moreover, the AMD women aged <65y had the C/C genotype
significantly more frequently than the controls of that age[10]. There are studies analyzing
MMP-2 concentration in blood[13,17], and the
choroidal neovascular membrane[18], or another genotype (MMP-2, rs2287074)[15]. In short, these
studies found no association between circulating MMP-2 and AMD development,
although low expression of MMP-2 in the corneal neovascularization (CNV)
membrane of mice and Bruch's membrane-choroid preparations in human donors eyes
was detected in the other studies[14].
Summarizing
gene polymorphism data on MMP-2 (-1306) C/T gene polymorphism, it was
found to have a significant part in the development of hard drusen in patients
with early AMD.
Further
advances in research about the MMP genotype-phenotype correlation in AMD may
prove valuable for better understanding of the genetic influence on the
pathogenesis of AMD, developing new prophylactic and therapeutic measures, and,
ultimately, careing for patients in a more cost-effective individualized, way.
In
conclusion, the MMP-2 Rs24386 (C-->T) polymorphism was found to be
associated with the development of hard drusen in patients with AMD but no
association was found in the group of geographic atrophy.
This study
had strengths. To our knowledge, this is the first study to have looked at the
association between MMP-2 (-1306 C/T) genotype and phenotype
characterized by soft and hard drusen and geographic atrophy.
This study had
several limitations. In future, these results need to be replicated in larger
studies, with bigger sample sizes in particular, to confirm the association
with late AMD and to determine whether the association might differ among wet
AMD and early AMD, and to include classification into 12 subgroups based upon
the International Classification and Grading System. The second of our study
limitations is absence of fundus autofluorescence (FAF) imaging, because fundus
autofluorescence is valuable for AMD progression detecting. It is known that
fundus autofluorescence imaging allows in vivo analysis of the dynamics
of accumulation and degradation of lipofuscin in the retinal pigment epithelium
(RPE) in eyes with AMD and documentation of metabolic activity of the RPE[19]. Domalpally et
al[20]
reported that geographic atrophy may be detected earlier by the use of FAF
images. Progression of geographic atriphy area is comparable between color
photographs and FAF images, but evaluating involvement of the center of the
macula may differ, probably because of macular pigmentation
blocking autofluorescence.
FAF is relevant for imaging in diagnosis and monitoring of inherited retinal
dystrophies, early AMD, geographic atrophy and central serous
chorioretinopathy[21].
In
conclusion, the MMP-2 Rs24386 (C-->T) polymorphism was found to be
associated with the development of hard drusen in patients with AMD and
logistic regression analysis revealed that MMP-2 (�C1306) C/C
genotype increased the likelihood of developing hard drusen in AMD patients
(OR=1.7, 95% CI: 1.06-2.74; P=0.028) but no association was found
in the group of geographic atrophy.
ACKNOWLEDGEMENTS
Foundation: Supported by
Lithuanian Science Council (No.MIP-10330).
Conflicts of
Interest: Liutkeviciene R, None; Lesauskaite V, None; Sinkunaite-Marsalkiene
G, None; Simonyte S, None; Zemaitiene R, None; Kriauciuniene
L, None; Zaliuniene D, None.
REFERENCES
1 Klein R, Peto T, Bird A, Vannewkirk MR. The epidemiology of age-related
macular degeneration. Am J Ophthalmology
2004;137(3):486-495. [CrossRef] [PubMed]
2 Young RW. Pathophysiology of age-related macular degeneration. Surv Ophthalmol 1987;31(5):291-306. [CrossRef]
3 Davis MD, Gangnon RE, Lee LY, Hubbard LD, Klein BE, Klein R, Ferris FL,
Bressler SB, Milton RC. The Age-Related Eye Disease Study severity scale for
agerelated macular degeneration: AREDS Report No. 17. Arch Ophthalmol 2005;123(11):1484-1498.
[CrossRef] [PMC free article] [PubMed]
4 Tatar O, Adam A, Shinoda K, Eckert T, Scharioth GB, Klein M, Yoeruek E,
Bartz-Schmidt KU, Grisanti S. Matrix metalloproteinases in human choroidal
neovascular membranes excised following verteporfin photodynamic therapy. Br J Ophthalmol
2007;91(9):1183-1189. [CrossRef] [PMC free article] [PubMed]
5 Hyman L, Neborsky R. Risk factors for age-related macular degeneration:
an update. Curr Opin Ophthalmol 2002;13(3):171-175. [CrossRef] [PubMed]
6 Price SJ, Greaves DR, Watkins H. Identification of novel, functional
genetic variants in the human matrix metalloproteinase-2 gene: role of Sp1 in allele-specific transcriptional
regulation. J Biol Chem 2001;276(10):
7549-7558. [CrossRef] [PubMed]
7 Peasey A, Bobak M, Kubinova R, Malyutina S, Pajak A, Tamosiunas A,
Pikhart H, Nicholson A, Marmot M. Determinants of cardiovascular disease and
other non-communicable diseases in Central and Eastern Europe: rationale and
design of the HAPIEE study. BMC Public
Health 2006;6:255. [CrossRef] [PMC free article] [PubMed]
8 Grabauskas V, Miseviciene I, Klumbiene J, Petkeviciene J, Milasauskiene
Z, Plieskiene A, Margeviciene L. Prevalence of dyslipidemias among Lithuanian
rural population (CINDI program). Medicina
(Kaunas) 2003; 39(12):1215-1222.
9 Lesauskaite V, Macijauskiene J, Rader E. Challenges and opportunities
of health care for the aging community in Lithuania. Gerontology 2006; 52(1):40-44. [CrossRef] [PubMed]
10 Liutkeviciene R, Lesauskaite V, Sinkunaite-Marsalkiene G, Zaliuniene
D, Zaliaduonyte-Peksiene D, Mizariene V, Gustiene O, Jasinskas V, Jariene G,
Tamosiunas A. The role of matrix metalloproteinases polymorphisms in
age-related macular degeneration. Ophthalmic
Genet 2015;36(2):149-155. [CrossRef] [PubMed]
11 The Age-Related Eye Disease Study Research Group. The Age-Related Eye
Disease Study system for classifying age-related macular degeneration from
stereoscopic color fundus photographs: the Age-Related Eye Disease Study Report
Number 6. Am J Ophthalmol 2001; 132(5):668-681. [CrossRef]
12 Moitinho F, Mora A, Vieira P, Fonseca J. A drusen volume
quantification method based on a segmentation algorithm in VIP image-thematic
conference on computational vision and medical image processing. Porto,
Portugal, 2007. [PubMed]
13 Zeng R, Wen F, Zhang X, Su Y. Serum levels of matrix metalloproteinase
2 and matrix metalloproteinase 9 elevated in polypoidal choroidal vasculopathy
but not in age-related macular degeneration.
Mol Vis 2013;19:729-736 [PMC free article] [PubMed]
14 Berglin L, Sarman S, van der Ploeg I, Steen B, Ming Y, Itohara S,
Seregard S, Kvanta A. Reduced choroidal neovascular membrane formation in
matrix metalloproteinase-2-deficient mice.
Invest Ophthalmol Vis Sci 2003;44(1):403-408. [CrossRef]
15 Seitzman RL, Mahajan VB, Mangione C, Cauley JA, Ensrud KE, Stone KL,
Cummings SR, Hochberg MC, Hillier TA, Sinsheimer JS, Yu F, Coleman AL. Estrogen
receptor alpha and matrix metalloproteinase 2 polymorphisms and age-related
maculopathy in older women. Am J
Epidemiol 2008;167(10):1217-1225. [CrossRef] [PubMed]
16 Ortak H, Demir S, Ates O, Benli I, Soğut E, Sahin M. The role of MMP2
(-1306C>T) and TIMP2 (-418 G>C) promoter variants in age-related macular
degeneration. Ophthalmic Genet 2013;34(4):217-222.
[CrossRef] [PubMed]
17 Chau KY, Sivaprasad S, Patel N, Donaldson TA, Luthert PJ, Chong NV.
Plasma levels of matrix metalloproteinase-2 and -9 (MMP-2 and MMP-9) in
age-related macular degeneration. Eye
(Lond) 2008;22(6): 855-859. [CrossRef] [PubMed]
18 Steen B, Sejersen S, Berglin L, Seregard S, Kvanta A. Matrix
metalloproteinases and metalloproteinase inhibitors in choroidal neovascular
membranes. Invest Ophthalmol Vis Sci 1998;39(11):2194-2200.
[PubMed]
19 von R��ckmann A, Schmidt KG, Fitzke FW, Bird AC, Jacobi KW.
Dynamics of accumulation and degradation of lipofuscin in retinal pigment
epithelium in senile macular degeneration. Klin
Monbl Augenheilkd 1998;213(1):32-37. [PubMed]
20 Domalpally A, Danis R, Agr��n E, Blodi B, Clemons T, Chew E.
Age-Related Eye Disease Study 2 Research Group. Evaluation of geographic
atrophy from color photographs and fundus autofluorescence images: Age-Related
Eye Disease Study 2 Report Number 11. Ophthalmology
2016;123(11):2401-2407. [CrossRef] [PubMed]
21 Frampton GK, Kalita N, Payne L, Colquitt J, Loveman E. Accuracy of
fundus autofluorescence imaging for the diagnosis and monitoring of retinal
conditions: a systematic review. Health
Technol Assess 2016; 20(31):1-108. [CrossRef]
[PMC free article] [PubMed]
--------------------------------------------------------------------------------------------------------------------------------
All rights reserved by Press of International Journal of Ophthalmology (IJO
PRESS)